From Surf Wiki (app.surf) — the open knowledge base
Hydrogen

the chemical element
Hydrogen is a chemical element; it has the symbolH and atomic number1. It is the lightest and most abundant chemical element in the universe, constituting about 75% of all normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the formula, called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecules, such as in water and organic compounds. The most common isotope of hydrogen, H, consists of one proton, one electron, and no neutrons.
Hydrogen gas was first produced artificially in the 17thcentury by the reaction of acids with metals. Henry Cavendish, in1766–1781, identified hydrogen gas as a distinct substance and discovered its property of producing water when burned: this is the origin of hydrogen's name, which means (from , and ). Understanding the colors of light absorbed and emitted by hydrogen was a crucial part of the development of quantum mechanics.
Hydrogen, typically nonmetallic except under extreme pressure, readily forms covalent bonds with most nonmetals, contributing to the formation of compounds like water and various organic substances. Its role is crucial in acid–base reactions, which mainly involve proton exchange among soluble molecules. In ionic compounds, hydrogen can take the form of either a negatively-charged anion, where it is known as hydride, or as a positively-charged cation, , hydron. Although tightly bonded to water molecules, hydrons strongly affect the behavior of aqueous solutions, as reflected in the importance of pH. Hydride, on the other hand, is rarely observed because it tends to deprotonate solvents, yielding.
In the early universe, neutral hydrogen atoms formed about 370,000 years after the Big Bang as the universe expanded and plasma had cooled enough for electrons to remain bound to protons. After stars began to form, most of the hydrogen in the intergalactic medium was re-ionized.
Nearly all hydrogen production is done by transforming fossil fuels, particularly steam reforming of natural gas. It can also be produced from water or saline by electrolysis, but this process is more expensive. Its main industrial uses include fossil fuel processing and ammonia production for fertilizer. Emerging uses for hydrogen include the use of fuel cells to generate electricity.
Properties
Atomic hydrogen
Electron energy levels
Main article: Hydrogen atom
The ground state energy level of the electron in a hydrogen atom is −13.6electronvolts(eV), equivalent to an ultraviolet photon of roughly 91nanometers wavelength. The energy levels of hydrogen are referred to by consecutive quantum numbers, with n=1 being the ground state. The hydrogen spectral series corresponds to emission of light due to transitions from higher to lower energy levels. Each energy level is further split by spin interactions between the electron and proton into four hyperfine levels.
High-precision values for the hydrogen atom energy levels are required for definitions of physical constants. Quantum calculations have identified nine contributions to the energy levels. The eigenvalue from the Dirac equation is the largest contribution. Other terms include relativistic recoil, the self-energy, and the vacuum polarization terms.
Nomenclature
The standards organization for chemical names, IUPAC, gives general names when the context assumes natural isotope abundance or ignores the isotope. These general names are hydrogen for the neutral atom, hydron for the positive cation, H+, hydride for the negative cation, H-. The name proton is often used for the positive cation but this strictly correct only for the cation of the dominant isotope .
Isotopes
Main article: Isotopes of hydrogen

Hydrogen has three naturally-occurring isotopes, denoted , and. Other, highly-unstable nuclides( to ) have been synthesized in laboratories but not observed in nature.{{cite journal |display-authors=8
**** is the most common hydrogen isotope, with an abundance of 99.98%. Because the nucleus of this isotope consists of only a single proton, it is given the descriptive but rarely used formal name protium.{{cite journal
****, the other stable hydrogen isotope, is known as deuterium and contains one proton and one neutron in the nucleus. Nearly all deuterium nuclei in the universe are thought to have been produced in Big Bang nucleosynthesis, and have endured since then. Deuterium is not radioactive, and is not a significant toxicity hazard. Water enriched in molecules that include deuterium instead of normal hydrogen is called heavy water. Deuterium and its compounds are used as a non-radioactive label in chemical experiments and in solvents for -NMR spectroscopy.{{cite journal |access-date=12 February 2008 |archive-date=29 January 2021 |archive-url=https://web.archive.org/web/20210129015717/https://www.nytimes.com/1991/11/11/us/breakthrough-in-nuclear-fusion-offers-hope-for-power-of-future.html |url-status=live
**** is known as tritium and contains one proton and two neutrons in its nucleus. It is radioactive, decaying into helium-3 through beta decay with a half-life of 12.32years. It is radioactive enough to be used in luminous paint to enhance the visibility of data displays, such as for painting the hands and dial-markers of watches. The watch glass prevents the small amount of radiation from escaping the case. Small amounts of tritium are produced naturally by cosmic rays striking atmospheric gases; tritium has also been released in nuclear weapons tests. It is used in nuclear fusion, as a tracer in isotope geochemistry, and in specialized self-powered lighting devices. Tritium has also been used in chemical and biological labeling experiments as a radiolabel.
Unique among the elements, distinct names are assigned to hydrogen's isotopes in common use. During the early study of radioactivity, heavy radioisotopes were given their own names, but these are mostly no longer used. The symbols D andT (instead of and ) are sometimes used for deuterium and tritium, but the symbolP was already used for phosphorus and thus was not available for protium. In its nomenclatural guidelines, the International Union of Pure and Applied Chemistry(IUPAC) allows any of D, T, , and to be used, though and are preferred.
Antihydrogen () is the antimatter counterpart to hydrogen. It consists of an with a positron. The exotic atom muonium (symbol Mu), composed of an antimuon and an electron, is the analogue of hydrogen; IUPACnomenclature incorporates such hypothetical compounds as muonium chloride(MuCl) and sodium muonide(NaMu), analogous to hydrogen chloride and sodium hydride respectively.{{cite journal |author2-link=International Union of Pure and Applied Chemistry |access-date=15 November 2016 |archive-url=https://web.archive.org/web/20110514000319/http://www.iupac.org/publications/pac/2001/pdf/7302x0377.pdf |archive-date=14 May 2011 |url-status=live
Dihydrogen
Under standard conditions, hydrogen is a gas of diatomic molecules with the formula, officially called "dihydrogen", but also called "molecular hydrogen", or simply hydrogen. Dihydrogen is a colorless, odorless, flammable gas.
Combustion
Hydrogen gas is highly flammable, reacting with oxygen in air to produce liquid water: The amount of heat released per mole of hydrogen is (kJ), or (MJ) for a 1 kilogram mass.{{cite book |access-date=3 September 2020 |archive-date=29 January 2021 |archive-url=https://web.archive.org/web/20210129015745/https://books.google.com/books?id=ugniowznToAC&pg=PA240 |url-status=live
Hydrogen gas forms explosive mixtures with air in concentrations from {{cite journal |access-date=3 September 2020 |archive-date=26 January 2021 |archive-url=https://web.archive.org/web/20210126131413/https://books.google.com/books?id=-CRRJBVv5d0C&pg=PA402 |url-status=live
Hydrogen flames emit faint blue and ultraviolet light. Flame detectors are used to detect hydrogen fires as they are nearly invisible to the naked eye in daylight.
Spin isomers
Main article: Spin isomers of hydrogen
Molecular exists as two nuclear isomers that differ in the spin states of their nuclei. In the **** form, the spins of the two nuclei are parallel, forming a spin triplet state having a total molecular spin S = 1; in the **** form the spins are and form a spin singlet state having spin S = 0. The equilibrium ratio of ortho- to para-hydrogen depends on temperature. At room temperature or warmer, equilibrium hydrogen gas contains about 25% of the para form and 75% of the ortho form. The ortho form is an excited state, having higher energy than the para form by , and it converts to the para form over the course of several minutes when cooled to low temperature.{{cite journal|last1=Milenko|first1=Yu. Ya.|last2=Sibileva|first2=R. M.|last3=Strzhemechny|first3=M. A.|title=Natural ortho-para conversion rate in liquid and gaseous hydrogen|journal=Journal of Low Temperature Physics|date=1997|volume=107|issue=1–2|pages=77–92
The ortho-to-para ratio in is an important consideration in the liquefaction and storage of liquid hydrogen: the conversion from ortho to para is exothermic, and produces sufficient heat to evaporate most of the liquid if the conversion to does not occur during the cooling process. Catalysts for the ortho-para , such as ferric oxide and activated carbon compounds, are therefore used during hydrogen cooling to avoid this loss of liquid.
Phases

Liquid hydrogen can exist at temperatures below hydrogen's critical point of 33 K. However, for it to be in a fully liquid state at atmospheric pressure, H2 needs to be cooled to 20.28 K. Hydrogen was liquefied by James Dewar in1898 by using regenerative cooling and his invention, the vacuum flask.
Liquid hydrogen becomes solid hydrogen at standard pressure below hydrogen's melting point of 14.01 K. Distinct solid phases exist, known as PhaseI through PhaseV, each exhibiting a characteristic molecular arrangement. Liquid and solid phases can exist in combination at the triple point; this mixture is known as slush hydrogen.
Metallic hydrogen, a phase obtained at extremely high pressures (in excess of 400 e6Pa), is an electrical conductor. It is believed to exist deep within giant planets like Jupiter.
When ionized, hydrogen becomes a plasma. This is the form in which hydrogen exists within stars.
Thermal and physical properties
| Temperature (K) | Density (kg/m3) | Specific heat (kJ/kg K) | Dynamic viscosity (kg/m s) | Kinematic viscosity (m2/s) | Thermal conductivity (W/m K) | Thermal diffusivity (m2/s) | Prandtl Number |
|---|---|---|---|---|---|---|---|
| 100 | 0.24255 | 11.23 | 4.21E-06 | 1.74E-05 | 6.70E-02 | 2.46E-05 | 0.707 |
| 150 | 0.16371 | 12.602 | 5.60E-06 | 3.42E-05 | 0.0981 | 4.75E-05 | 0.718 |
| 200 | 0.1227 | 13.54 | 6.81E-06 | 5.55E-05 | 0.1282 | 7.72E-05 | 0.719 |
| 250 | 0.09819 | 14.059 | 7.92E-06 | 8.06E-05 | 0.1561 | 1.13E-04 | 0.713 |
| 300 | 0.08185 | 14.314 | 8.96E-06 | 1.10E-04 | 0.182 | 1.55E-04 | 0.706 |
| 350 | 0.07016 | 14.436 | 9.95E-06 | 1.42E-04 | 0.206 | 2.03E-04 | 0.697 |
| 400 | 0.06135 | 14.491 | 1.09E-05 | 1.77E-04 | 0.228 | 2.57E-04 | 0.69 |
| 450 | 0.05462 | 14.499 | 1.18E-05 | 2.16E-04 | 0.251 | 3.16E-04 | 0.682 |
| 500 | 0.04918 | 14.507 | 1.26E-05 | 2.57E-04 | 0.272 | 3.82E-04 | 0.675 |
| 550 | 0.04469 | 14.532 | 1.35E-05 | 3.02E-04 | 0.292 | 4.52E-04 | 0.668 |
| 600 | 0.04085 | 14.537 | 1.43E-05 | 3.50E-04 | 0.315 | 5.31E-04 | 0.664 |
| 700 | 0.03492 | 14.574 | 1.59E-05 | 4.55E-04 | 0.351 | 6.90E-04 | 0.659 |
| 800 | 0.0306 | 14.675 | 1.74E-05 | 5.69E-04 | 0.384 | 8.56E-04 | 0.664 |
| 900 | 0.02723 | 14.821 | 1.88E-05 | 6.90E-04 | 0.412 | 1.02E-03 | 0.676 |
| 1000 | 0.02424 | 14.99 | 2.01E-05 | 8.30E-04 | 0.448 | 1.23E-03 | 0.673 |
| 1100 | 0.02204 | 15.17 | 2.13E-05 | 9.66E-04 | 0.488 | 1.46E-03 | 0.662 |
| 1200 | 0.0202 | 15.37 | 2.26E-05 | 1.12E-03 | 0.528 | 1.70E-03 | 0.659 |
| 1300 | 0.01865 | 15.59 | 2.39E-05 | 1.28E-03 | 0.568 | 1.96E-03 | 0.655 |
| 1400 | 0.01732 | 15.81 | 2.51E-05 | 1.45E-03 | 0.61 | 2.23E-03 | 0.65 |
| 1500 | 0.01616 | 16.02 | 2.63E-05 | 1.63E-03 | 0.655 | 2.53E-03 | 0.643 |
| 1600 | 0.0152 | 16.28 | 2.74E-05 | 1.80E-03 | 0.697 | 2.82E-03 | 0.639 |
| 1700 | 0.0143 | 16.58 | 2.85E-05 | 1.99E-03 | 0.742 | 3.13E-03 | 0.637 |
| 1800 | 0.0135 | 16.96 | 2.96E-05 | 2.19E-03 | 0.786 | 3.44E-03 | 0.639 |
| 1900 | 0.0128 | 17.49 | 3.07E-05 | 2.40E-03 | 0.835 | 3.73E-03 | 0.643 |
| 2000 | 0.0121 | 18.25 | 3.18E-05 | 2.63E-03 | 0.878 | 3.98E-03 | 0.661 |
History
Main article: Timeline of hydrogen technologies
18th century
In 1671, Irish scientist Robert Boyle discovered and described the reaction between iron filings and dilute acids, which results in the production of hydrogen gas.{{cite web |access-date=5 February 2008 |archive-url=https://web.archive.org/web/20080410102154/http://education.jlab.org/itselemental/ele001.html |archive-date=10 April 2008 Boyle did not note that the gas was flammable, but hydrogen would play a key role in overturning the phlogiston theory of combustion.
In 1766, Henry Cavendish was the first to recognize hydrogen gas as a discrete substance, by naming the gas from a metal-acid reaction "inflammable air". He speculated that "inflammable air" was in fact identical to the hypothetical substance "phlogiston"{{cite book |last = Musgrave |access-date = 22 October 2011 |chapter-url = http://ebooks.cambridge.org/chapter.jsf?bid=CBO9780511760013&cid=CBO9780511760013A009 |url-access = registration
In 1783, identified the element that came to be known as hydrogen when he and reproduced Cavendish's finding that water is produced when hydrogen is burned. produced hydrogen for his experiments on mass conservation by treating metallic iron with a stream of water through an incandescent iron tube heated in a fire. Anaerobic oxidation of iron by the protons of water at high temperature can be schematically represented by the set of following reactions:
Many metals react similarly with water, leading to the production of hydrogen. In some situations, this H2-producing process is problematic, for instance in the case of zirconium cladding on nuclear fuel rods.
19th century
By 1806 hydrogen was used to fill balloons. built the first engine, an internal combustion engine powered by a mixture of hydrogen and oxygen, in1806. Edward Daniel Clarke invented the hydrogen gas blowpipe in1819. The 's lamp and limelight were invented in1823. Hydrogen was liquefied for the first time by James Dewar in1898 by using regenerative cooling and his invention, the vacuum flask. He produced solid hydrogen the next year.
One of the first quantum effects to be explicitly noticed, although not understood at the time, was James Clerk Maxwell's observation that the specific heat capacity of unaccountably departs from that of a diatomic gas below room temperature, and begins to increasingly resemble that of a monatomic gas at cryogenic temperatures. According to quantum theory, this behavior arises from the spacing of the (quantized) rotational energy levels, which are particularly wide-spaced in because of its low mass. These widely-spaced levels inhibit equal partition of heat energy into rotational motion in hydrogen at low temperatures. Diatomic gases composed of heavier atoms do not have such widely spaced levels and do not exhibit the same effect.{{cite journal
20th century
The existence of the hydride anion was suggested by GilbertN. Lewis in1916 for group1 and group2 salt-like compounds. In1920, electrolyzed molten lithium hydride(LiH), producing a stoichiometric quantity of hydrogen at the anode.
Because of its simple atomic structure, consisting only of a proton and an electron, the hydrogen atom, together with the spectrum of light produced from it or absorbed by it, has been central to the development of the theory of atomic structure.{{cite book |last=Crepeau |first=R. |access-date=20 December 2007 |archive-url=https://web.archive.org/web/20081017073826/http://www.iki.rssi.ru/mirrors/stern/stargaze/Q5.htm |archive-date=17 October 2008
Hydrogen's unique position as the only neutral atom for which the equation can be directly solved, has significantly contributed to the understanding of quantum mechanics through the exploration of its energetics. Furthermore, study of the corresponding simplicity of the hydrogen molecule and the corresponding cation, , brought understanding of the nature of the chemical bond, which followed shortly after the quantum mechanical treatment of the hydrogen atom had been developed in the mid-1920s.
Hydrogen-lifted airship
Because has only 7% the density of air, it was once widely used as a lifting gas in balloons and airships. The first hydrogen-filled balloon was invented by in1783. Hydrogen provided the lift for the first reliable form of air-travel following the1852 invention of the first hydrogen-lifted airship by . German count promoted the idea of rigid airships lifted by hydrogen that later were called , the first of which had its maiden flight in1900. Regularly-scheduled flights started in1910 and by the outbreak of World WarI in August1914, they had carried 35,000 passengers without a serious incident. Hydrogen-lifted airships in the form of blimps were used as observation platforms and bombers during World WarII, especially on the USEastern seaboard.
The first non-stop transatlantic crossing was made by the British airshipR34 in1919 and regular passenger service resumed in the1920s. Hydrogen was used in the Hindenburg, which caught fire over New Jersey on 6May 1937. The hydrogen that filled the airship was ignited, possibly by static electricity, and burst into flames. Following this disaster, commercial hydrogen airship travel ceased. Hydrogen is still used, in preference to non-flammable but more expensive helium, as a lifting gas for weather balloons.
Deuterium and tritium
Deuterium was discovered in December1931 by Harold Urey, and tritium was prepared in1934 by Ernest Rutherford, Mark Oliphant, and Paul Harteck. Heavy water, which consists of deuterium in the place of regular hydrogen, was discovered by Urey's group in1932.
Chemistry
Reactions of H2
is relatively unreactive. The thermodynamic basis of this low reactivity is the very strong , with a bond dissociation energy of . It does form coordination complexes called dihydrogen complexes. These species provide insights into the early steps in the interactions of hydrogen with metal catalysts. According to neutron diffraction, the metal and two Hatoms form a triangle in these complexes. The remains intact but is elongated. They are acidic.{{Cite book
Although exotic on Earth, the ion is common in the universe. It is a triangular species, like the aforementioned dihydrogen complexes. It is known as protonated molecular hydrogen or the trihydrogen cation.{{cite journal
Hydrogen reacts with chlorine to produceHCl, and with bromine to produceHBr, via a chain reaction. The reaction requires initiation. For example, in the case of Br2, the dibromine molecule is split apart: . Propagating reactions consume hydrogen molecules and produceHBr, as well as Brand Hatoms: Finally the terminating reaction: consumes the remaining atoms.
The addition of H2 to unsaturated organic compounds, such as alkenes and alkynes, is called hydrogenation. Even if the reaction is energetically favorable, it does not occur spontaneously even at higher temperatures. In the presence of a catalyst like finely divided platinum or nickel, the reaction proceeds at room temperature.
Hydrogen-containing compounds
Main article: Hydrogen compounds
Hydrogen can exist in both +1 and −1oxidation states, forming compounds through ionic and covalent bonding. The element is part of a wide range of substances, including water, hydrocarbons, and numerous other organic compounds. The H+ion—commonly referred to as a proton due to its single proton and absence of electrons—is central to acid–base chemistry, although the proton does not move freely. In the –Lowry framework, acids are defined by their ability to donate H+ions to bases.
Hydrogen forms a vast variety of compounds with carbon, known as hydrocarbons, and an even greater diversity with other elements (heteroatoms), giving rise to the broad class of organic compounds often associated with living organisms.
Hydrogen compounds with hydrogen in the oxidation state−1 are known as hydrides, which are usually formed between hydrogen and metals. The hydrides can be ionic (aka saline), covalent, or metallic. With heating, H2 reacts efficiently with the alkali and alkaline earth metals to give the ionic hydrides of the formulasMH and MH2, respectively. These salt-like crystalline compounds have high melting points and all react with water to liberate hydrogen. Covalent hydrides include boranes and polymeric aluminium hydride. Transition metals form metal hydrides via continuous dissolution of hydrogen into the metal. Perhaps the most extensive series of hydrides are the boranes, compounds consisting only of boron and hydrogen.{{cite journal
Hydrides can bond to these electropositive elements not only as a terminal ligand but also as bridging ligands. In diborane(), four hydrogen atoms are terminal, while two bridge between the two boron atoms.
Hydrogen bonding
Main article: Hydrogen bond
When bonded to a more electronegative element, particularly fluorine, oxygen, or nitrogen, hydrogen can participate in a form of medium-strength noncovalent bonding with another electronegative element with a lone pair like oxygen or nitrogen. This phenomenon, called hydrogen bonding, is critical to the stability of many biological molecules. Hydrogen bonding alters molecule structures, viscosity, solubility, melting and boiling points, and even protein folding dynamics.
Protons and acids
In water, hydrogen bonding plays an important role in reaction thermodynamics. A hydrogen bond can shift over to proton transfer. Under the –Lowry acid–base theory, acids are proton donors, while bases are proton acceptors. A bare proton() essentially cannot exist in anything other than a vacuum. Otherwise it attaches to other atoms, ions, or molecules. Even chemical species as inert as methane can be protonated. The term "proton" is used loosely and metaphorically to refer to solvated hydrogen cations attached to other solvated chemical species; it is denoted"" without any implication that any single protons exist freely in solution as a species. To avoid the implication of the naked proton in solution, acidic aqueous solutions are sometimes considered to contain the "hydronium ion"(), or still more accurately, .{{cite journal
The concentration of these solvated protons determines the pH of a solution, a logarithmic scale that reflects its acidity or basicity. Lower pHvalues indicate higher concentrations of hydronium ions, corresponding to more acidic conditions.
Occurrence
Cosmic
Hydrogen, as atomic H, is the most abundant chemical element in the universe, making up 75% of normal matter by mass.{{cite web |access-date=5 February 2008 |archive-url=https://web.archive.org/web/20150115074556/http://imagine.gsfc.nasa.gov/ask_astro/stars.html#961112a |archive-date=15 January 2015 |url-status=live
In astrophysics, neutral hydrogen in the interstellar medium is called HI and ionized hydrogen is called HII. Radiation from stars ionizes HI to HII, creating spheres of ionized HII around stars. In the chronology of the universe neutral hydrogen dominated until the birth of stars during the era of reionization, which then produced bubbles of ionized hydrogen that grew and merged over hundreds of millions of years. These are the source of the 21-centimeter hydrogen line, at , that is detected in order to probe primordial hydrogen. The large amount of neutral hydrogen found in the damped Lyman-alpha systems is thought to dominate the cosmological baryonic density of the universe up to a redshift of .{{cite journal
Hydrogen is found in great abundance in stars and gas giant planets. Molecular clouds of are associated with star formation. Hydrogen plays a vital role in powering stars through the proton-proton reaction in lower-mass stars, and through the CNOcycle of nuclear fusion in stars more massive than the Sun.{{cite web |archive-date=11 December 2011 |title=Solar Thermonuclear Energy Generation
Protonated molecular hydrogen() is found in the interstellar medium, where it is generated by ionization of molecular hydrogen by cosmic rays. This ion has also been observed in the upper atmosphere of Jupiter. The ion is long-lived in outer space due to the low temperature and density. is one of the most abundant ions in the universe, and it plays a notable role in the chemistry of the interstellar medium. Neutral triatomic hydrogen can exist only in an excited form and is unstable.
Terrestrial
Hydrogen is the third most abundant element on the Earth's surface,{{cite journal |author-link=Mildred Dresselhaus |display-authors=etal |access-date=5 February 2008 |archive-url=https://web.archive.org/web/20080213144956/http://www.sc.doe.gov/bes/hydrogen.pdf |archive-date=13 February 2008
Large underground deposits of hydrogen gas have been discovered in several countries including Mali, France and Australia. As of 2024, it is uncertain how much underground hydrogen can be extracted economically.
Production and storage
Main article: Hydrogen production
Industrial routes
Nearly all of the world's current supply of hydrogen gas() is produced from fossil fuels. Many methods exist for producing H2, but three dominate commercially: steam reforming often coupled to water-gas shift, partial oxidation of hydrocarbons, and water electrolysis.
Steam reforming

Hydrogen is mainly produced by steam methane reforming(SMR), the reaction of water and methane.{{cite book Producing one tonne of hydrogen through this process emits tonnes of carbon dioxide. The production of natural gas feedstock also produces emissions such as vented and fugitive methane, which further contributes to the overall carbon footprint of hydrogen.
This reaction is favored at low pressures but is nonetheless conducted at high pressures(2.0 MPa) because high-pressure is the most marketable product, and pressure swing adsorption(PSA) purification systems work better at higher pressures. The product mixture is known as "synthesis gas" because it is often used directly for the production of methanol and many other compounds. Hydrocarbons other than methane can be used to produce synthesis gas with varying product ratios. One of the many complications to this highly-optimized technology is the formation of coke or carbon:
Therefore, steam reforming typically employs an excess of. Additional hydrogen can be recovered from the steam by using carbon monoxide through the water gas shift reaction(WGS). This process requires an iron oxide catalyst:
Hydrogen is sometimes produced and consumed in the same industrial process, without being separated. In the process for ammonia production, hydrogen is generated from natural gas.
Partial oxidation of hydrocarbons
Other methods for CO and production include partial oxidation of hydrocarbons:
Although less important commercially, coal can serve as a prelude to the above shift reaction:
Olefin production units may produce substantial quantities of byproduct hydrogen, particularly from cracking light feedstocks like ethane or propane.
Water electrolysis
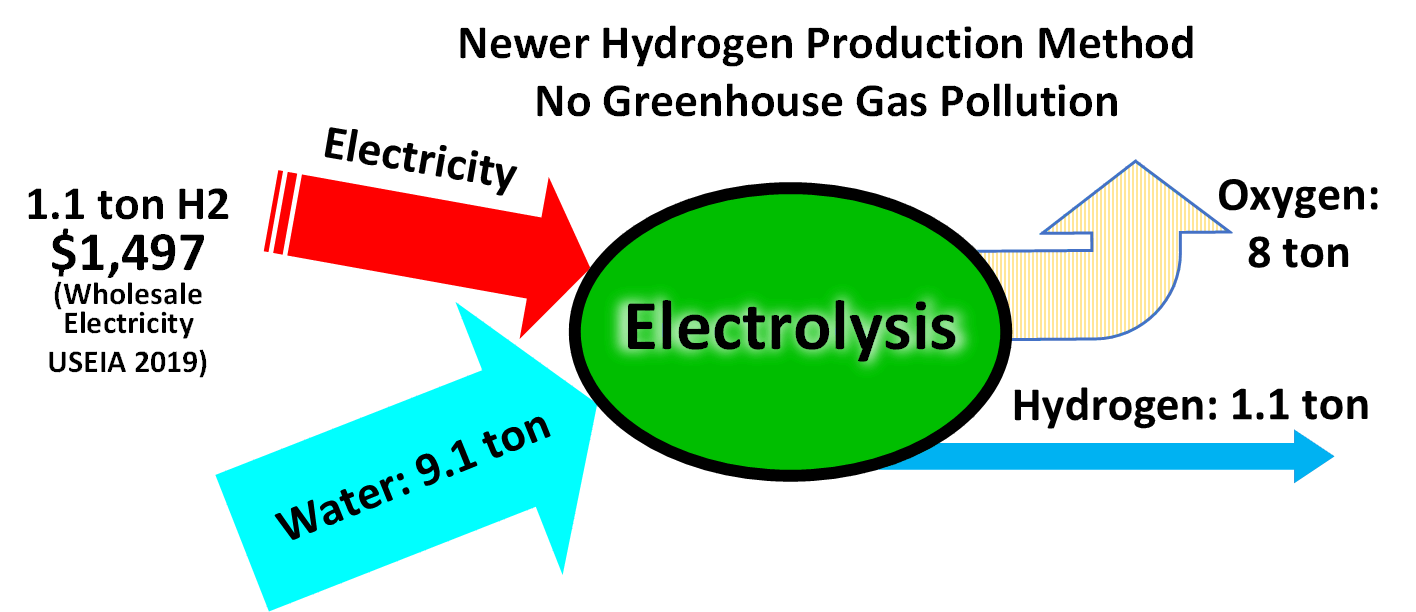
Electrolysis of water is a conceptually simple method of producing hydrogen. Commercial electrolyzers use nickel-based catalysts in strongly alkaline solution. Platinum is a better catalyst but is expensive. The hydrogen created through electrolysis using renewable energy is commonly referred to as "green hydrogen".
Electrolysis of brine to yield chlorine also produces high-purity hydrogen as a co-product, which is used for a variety of transformations such as hydrogenations.
The electrolysis process is more expensive than producing hydrogen from methane without carbon capture and storage.
Innovation in hydrogen electrolyzers could make large-scale production of hydrogen from electricity more cost-competitive.{{Cite book|author1-link=International Energy Agency|last1=IEA|title=Net Zero by 2050: A Roadmap for the Global Energy Sector|year=2021|url=https://iea.blob.core.windows.net/assets/ad0d4830-bd7e-47b6-838c-40d115733c13/NetZeroby2050-ARoadmapfortheGlobalEnergySector.pdf|archive-date=23 May 2021|archive-url=https://web.archive.org/web/20210523155010/https://iea.blob.core.windows.net/assets/ad0d4830-bd7e-47b6-838c-40d115733c13/NetZeroby2050-ARoadmapfortheGlobalEnergySector.pdf|url-status=live
Methane pyrolysis
Hydrogen can be produced by pyrolysis of natural gas (methane), producing hydrogen gas and solid carbon with the aid of a catalyst and input heat: The carbon may be sold as a manufacturing feedstock or fuel, or landfilled. This route could have a lower carbon footprint than existing hydrogen production processes, but mechanisms for removing the carbon and preventing it from reacting with the catalyst remain obstacles for industrial-scale use.
Thermochemical
Water splitting is the process by which water is decomposed into its components. Relevant to the biological scenario is this equation: The reaction occurs in the light-dependent reactions in all photosynthetic organisms. A few organisms, including the alga Chlamydomonas reinhardtii and cyanobacteria, have evolved a second step in the dark reactions in which protons and electrons are reduced to form gas by specialized hydrogenases in the chloroplast.
Efforts have been undertaken to genetically modify cyanobacterial hydrogenases to more efficiently generate gas even in the presence of oxygen.{{cite web |access-date= 6 August 2016 |archive-url= https://web.archive.org/web/20161229231756/https://www.hydrogen.energy.gov/pdfs/progress05/iv_e_6_smith.pdf |archive-date= 29 December 2016 |url-status= live
Relevant to the thermal water-splitting scenario is this simple equation: Over 200 thermochemical cycles can be used for water splitting. Many of these cycles such as the iron oxide cycle, cerium(IV) oxide–cerium(III) oxide cycle, zinc–zinc oxide cycle, sulfur–iodine cycle, copper–chlorine cycle and hybrid sulfur cycle have been evaluated for their commercial potential to produce hydrogen and oxygen from water and heat without using electricity. A number of labs (including in France, Germany, Greece, Japan, and the United States) are developing thermochemical methods to produce hydrogen from solar energy and water.
Natural routes
Biohydrogen
is produced in organisms by enzymes called hydrogenases. This process allows the host organism to use fermentation as a source of energy. These same enzymes also can oxidizeH2, such that the host organisms can subsist by reducing oxidized substrates using electrons extracted fromH2.
Hydrogenase enzymes feature iron or iron–nickel centers at their active sites. The natural cycle of hydrogen production and consumption by organisms is called the hydrogen cycle.
Some bacteria such as Mycobacterium smegmatis can use the small amount of hydrogen in the atmosphere as a source of energy when other sources are lacking. Their hydrogenases feature small channels that exclude oxygen from the active site, permitting the reaction to occur even though the hydrogen concentration is very low and the oxygen concentration is as in normal air.
Confirming the existence of hydrogenase‐employing microbes in the human gut, occurs in human breath. The concentration in the breath of fasting people at rest is typically under (ppm), but can reach when people with intestinal disorders consume molecules they cannot absorb during diagnostic hydrogen breath tests.
Serpentinization
Serpentinization is a geological mechanism which produces highly-reducing conditions. Under these conditions, water is capable of oxidizing ferrous() ions in fayalite, generating hydrogen gas:
Closely related to this geological process is the reaction: This process also is relevant to the corrosion of iron and steel in oxygen-free groundwater and in reducing soils below the water table.
Laboratory syntheses
is produced in laboratory settings, such as in the small-scale electrolysis of water using metal electrodes and water containing an electrolyte, which liberates hydrogen gas at the cathode: Hydrogen is also often a by-product of other reactions. Many metals react with water to produce, but the rate of hydrogen evolution depends on the metal, the pH, and the presence of alloying agents. Most often, hydrogen evolution is induced by acids. The alkali and alkaline earth metals as well as aluminium, zinc, manganese, and iron, react readily with aqueous acids.
Many metals, such as aluminium, are slow to react with water because they form passivated oxide coatings. An alloy of aluminium and gallium, however, does react with water. In high-pH solutions, aluminium can react with :
Storage
If H2 is to be used as an energy source, its storage is important. It dissolves only poorly in solvents. For example, at room temperature and 0.1 mPa, of hydrogen dissolve into 1 kg of diethyl ether. H2 can be stored in compressed form, although compressing costs energy. Liquefaction is impractical given hydrogen's low critical temperature. In contrast, ammonia and many hydrocarbons can be liquified at room temperature under pressure. For these reasons, hydrogen carriers—materials that reversibly bindH2—have attracted much attention. The key question is then the weight percent of H2-equivalents within the carrier material. For example, hydrogen can be reversibly absorbed into many rare earths and transition metals{{cite journal |access-date=19 May 2015 |archive-url=https://web.archive.org/web/20150521085421/http://news.mongabay.com/2005/0921-hydrogen_tablet.html |archive-date=21 May 2015 |url-status=live
The most problematic aspect of metal hydrides for storage is their modest H2content, often on the order of1%. For this reason, there is interest in storage of H2 in compounds of low molecular weight. For example, ammonia borane () contains 19.8weight percent ofH2. The problem with this material is that after release of H2, the resulting boron nitride does not re-add H2: i.e., ammonia borane is an irreversible hydrogen carrier. More attractive are hydrocarbons such as tetrahydroquinoline, which reversibly release someH2 when heated in the presence of a catalyst:
Applications

Petrochemical industry
Large quantities of are used in the "upgrading" of fossil fuels. Key consumers of include hydrodesulfurization and hydrocracking. Many of these reactions can be classified as hydrogenolysis, i.e., the cleavage of bonds by hydrogen. Illustrative is the separation of sulfur from liquid fossil fuels:
Hydrogenation
Hydrogenation, the addition of to various substrates, is done on a large scale. Hydrogenation of produces ammonia by the process: This process consumes a few percent of the energy budget in the entire industry and is the biggest consumer of hydrogen. The resulting ammonia is used extensively in fertilizer production; these fertilizers have become essential feedstocks in modern agriculture. Hydrogenation is also used to convert unsaturated fats and oils to saturated fats and oils. The major application is the production of margarine. Methanol is produced by hydrogenation of carbon dioxide; the mixture of hydrogen and carbon dioxide used for this process is known as syngas. It is similarly the source of hydrogen in the manufacture of hydrochloric acid. is also used as a reducing agent for the conversion of some ores to the metals.
Fuel
The potential for using hydrogen(H2) as a fuel has been widely discussed. Hydrogen can be used in fuel cells to produce electricity, or burned to generate heat. When hydrogen is consumed in fuel cells, the only emission at the point of use is water vapor. When burned, hydrogen produces relatively little pollution at the point of combustion, but can lead to thermal formation of harmful nitrogen oxides.
If hydrogen is produced with low or zero greenhouse gas emissions (green hydrogen), it can play a significant role in decarbonizing energy systems where there are challenges and limitations to replacing fossil fuels with direct use of electricity.
Hydrogen fuel can produce the intense heat required for industrial production of steel, cement, glass, and chemicals, thus contributing to the decarbonization of industry alongside other technologies, such as electric arc furnaces for steelmaking. However, it is likely to play a larger role in providing industrial feedstock for cleaner production of ammonia and organic chemicals. For example, in steelmaking, hydrogen could function as a clean fuel and also as a low-carbon catalyst, replacing coal-derived coke (carbon): Hydrogen used to decarbonize transportation is likely to find its largest applications in shipping, aviation and, to a lesser extent, heavy goods vehicles, through the use of hydrogen-derived synthetic fuels such as ammonia and methanol and fuel cell technology. For light-duty vehicles including cars, hydrogen is far behind other alternative fuel vehicles, especially compared with the rate of adoption of battery electric vehicles, and may not play a significant role in future.
Liquid hydrogen and liquid oxygen together serve as cryogenic propellants in liquid-propellant rockets, as in the Space Shuttle main engines. NASA has investigated the use of rocket propellant made from atomic hydrogen, boron or carbon that is frozen into solid molecular hydrogen particles suspended in liquid helium. Upon warming, the mixture vaporizes to allow the atomic species to recombine, heating the mixture to high temperature.
Hydrogen produced when there is a surplus of variable renewable electricity could in principle be stored and later used to generate heat or to re-generate electricity. It can be further transformed into synthetic fuels such as ammonia and methanol. Disadvantages of hydrogen fuel include high costs of storage and distribution due to hydrogen's explosivity, its large volume compared to other fuels, and its tendency to embrittle materials.
Nickel–hydrogen battery
The very long-lived, rechargeable nickel–hydrogen battery developed for satellite power systems uses pressurized gaseousH2. The International Space Station, Mars Odyssey and the Mars Global Surveyor are equipped with nickel-hydrogen batteries. In the dark part of its orbit, the Hubble Space Telescope is also powered by nickel-hydrogen batteries, which were finally replaced in May2009, more than 19years after launch and 13years beyond their design life.
Semiconductor industry
Hydrogen is employed in semiconductor manufacturing to saturate broken ("dangling") bonds of amorphous silicon and amorphous carbon, which helps in stabilizing the materials' properties.{{cite journal The doping behavior of hydrogen varies with material.{{cite journal
Niche and evolving uses
Beyond than the uses mentioned above, hydrogen is used in smaller scales in the following applications:
-
Shielding gas: Hydrogen is used as a shielding gas in welding methods such as atomic hydrogen welding.{{cite journal
-
Coolant: Hydrogen is used as a coolant in large electrical generators due to its high thermal conductivity and low density. The first hydrogen-cooled turbogenerator went into service using gaseous hydrogen as a coolant in the rotor and the stator in1937 in Dayton, Ohio.
-
Cryogenic research: Liquid is used in cryogenic research, including superconductivity studies.{{cite journal
-
Food industry: Hydrogen is an authorized food additive (E949) that is used as a packaging gas, and also has antioxidant properties.
-
Leak detection: Pure or mixed with nitrogen (sometimes called forming gas), hydrogen is a tracer gas for detection of minute leaks. Applications can be found in the automotive, chemical, power generation, aerospace, and telecommunications industries;{{cite conference |access-date=25 March 2008 |archive-url=https://web.archive.org/web/20090108102521/http://www.ndt.net/abstract/wcndt2004/523.htm |archive-date=8 January 2009
-
Neutron moderation: Deuterium (hydrogen-2) is used in nuclear fission applications as a moderator to slow neutrons.
-
Nuclear fusion fuel: Deuterium is used in nuclear fusion reactions.
-
Isotopic labeling: Deuterium compounds have applications in chemistry and biology in studies of isotope effects on reaction rates.{{cite journal|last1=Reinsch| first1=J.|first2=A. |last2=Katz|first3=J.|last3=Wean|first4=G.|last4=Aprahamian|first5=J. T.|last5=MacFarland
-
Tritium uses: Tritium (hydrogen-3), produced in nuclear reactors, is used in the production of hydrogen bombs, as an isotopic label in the biosciences, and as a source of beta radiation in radioluminescent paint for instrument dials and emergency signage.
Safety and precautions
Main article: Hydrogen safety
| NFPA-H = 0 | NFPA-F = 4 | NFPA-R = 0 | NFPA-S = In hydrogen pipelines and steel storage vessels, hydrogen molecules are prone to reacting with metals, causing hydrogen embrittlement and leaks in the pipeline or storage vessel. Since it is lighter than air, hydrogen does not easily accumulate to form a combustible gas mixture. However, even without ignition sources, high-pressure hydrogen leakage may cause spontaneous combustion and detonation.
Hydrogen is flammable when mixed even in small amounts with air. Ignition can occur at a volumetric ratio of hydrogen to air as low as 4%. In approximately 70% of hydrogen ignition accidents, the ignition source cannot be found, and it is widely believed by scholars that spontaneous ignition of hydrogen occurs.
Hydrogen fire, while being extremely hot, is almost invisible to the human eye, and thus can lead to accidental burns. Hydrogen is non-toxic, but like most gases it can cause asphyxiation in the absence of adequate ventilation.
References
References
- "Element: Hydrogen".
- (2009). "Energy Levels". University of Nebraska Lincoln.
- (20 May 2015). "photon wavelength 13.6 eV". Wolfram Alpha.
- Levine, Ira N.. (1970). "Quantum chemistry". Pearson.
- (2011). "The Feynman lectures on physics". Basic Books.
- (2021-09-23). "CODATA Recommended Values of the Fundamental Physical Constants: 2018*". Journal of Physical and Chemical Reference Data.
- Bunnet, J.F.. (1968). "Names for hydrogen atoms, ions, and groups, and for reactions involving them (Recommendations 1988)". [[Pure Appl. Chem.]].
- {{NUBASE2020
- (2022-08-08). "Review of Particle Physics". Progress of Theoretical and Experimental Physics.
- (June 1995). "Tritium radioluminescent devices, Health and Safety Manual". International Atomic Energy Agency.
- Staff. (15 November 2007). "Tritium". U.S. Environmental Protection Agency.
- Nave, C. R.. (2006). "Deuterium-Tritium Fusion". Georgia State University.
- (1998). "Chapter 2: Fundamentals of Isotope Geochemistry". US Geological Survey.
- (2008). "The Tritium Laboratory". University of Miami.
- (2004). "Potential Role of Parasitism in the Evolution of Mutualism in Astigmatid Mites". Experimental and Applied Acarology.
- van der Krogt, P.. (5 May 2005). "Hydrogen". Elementymology & Elements Multidict.
- § IR-3.3.2, [http://old.iupac.org/reports/provisional/abstract04/RB-prs310804/Chap3-3.04.pdf Provisional Recommendations] {{Webarchive. link. (9 February 2016 , Nomenclature of Inorganic Chemistry, Chemical Nomenclature and Structure Representation Division, IUPAC. Accessed on line 3 October 2007.)
- (1 March 2015). "Advances in antihydrogen physics". Science Progress.
- (29 January 2015). "Why Antimatter Matters". European Review.
- [http://old.iupac.org/publications/books/rbook/Red_Book_2005.pdf Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005] - Full text (PDF)
2004 version with separate chapters as pdf: [http://www.iupac.org/reports/provisional/abstract04/connelly_310804.html IUPAC Provisional Recommendations for the Nomenclature of Inorganic Chemistry (2004)] {{webarchive. link. (2008-02-19) - "Hydrogen".
- (2011-02-01). "Mechanism of high-pressure hydrogen auto-ignition when spouting into air". International Journal of Hydrogen Energy.
- (June 2009). "Visible emission of hydrogen flames". Combustion and Flame.
- "Making Visible the Invisible {{!}} NASA Spinoff".
- Staff. (2003). "Hydrogen (H2) Properties, Uses, Applications: Hydrogen Gas and Liquid Hydrogen". Universal Industrial Gases, Inc..
- (2012). "The theory and practice of hyperpolarization in magnetic resonance using ''para''hydrogen". Prog. Nucl. Magn. Reson. Spectrosc..
- "Die Entdeckung des para-Wasserstoffs (The discovery of para-hydrogen)".
- Hritz, J.. (March 2006). "CH. 6 – Hydrogen". NASA.
- (1 November 1998). "Costs of Storing and Transporting Hydrogen". National Renewable Energy Laboratory.
- (1957). "The Conversion of Ortho- to Parahydrogen on Iron Oxide-Zinc Oxide Catalysts". Journal of the American Chemical Society.
- Stevenson, D J. (May 1982). "Interiors of the Giant Planets". Annual Review of Earth and Planetary Sciences.
- (2023). "Hydrogen". [[National Institute of Standards and Technology]].
- (1900). "Liquid Hydrogen". Science.
- (2020-09-01). "Understanding dense hydrogen at planetary conditions". Nature Reviews Physics.
- Ohira, K.. (2016). "Compendium of Hydrogen Energy". Elsevier.
- (2022). "Astronomy 2e". OpenStax.
- Phillips, K. J. H.. (1995). "Guide to the Sun". [[Cambridge University Press]].
- Holman, Jack P.. (2002). "Heat transfer". McGraw-Hill.
- (2007). "Fundamentals of heat and mass transfer". John Wiley and Sons, Inc.
- Boyle, R.. (1672). "Tracts written by the Honourable Robert Boyle containing new experiments, touching the relation betwixt flame and air, and about explosions, an hydrostatical discourse occasion'd by some objections of Dr. Henry More against some explications of new experiments made by the author of these tracts: To which is annex't, an hydrostatical letter, dilucidating an experiment about a way of weighing water in water, new experiments, of the positive or relative levity of bodies under water, of the air's spring on bodies under water, about the differing pressure of heavy solids and fluids". Printed for Richard Davis.
- Ramsay, W.. (1896). "The gases of the atmosphere: The history of their discovery". Macmillan.
- (12 May 1766). "Three Papers, Containing Experiments on Factitious Air, by the Hon. Henry Cavendish, F. R. S.". Philosophical Transactions.
- (2005). "Hydrogen". Wylie-Interscience.
- Emsley, John. (2001). "Nature's Building Blocks". Oxford University Press.
- Stwertka, Albert. (1996). "A Guide to the Elements". Oxford University Press.
- (1983). "Hydrides and delayed hydrogen cracking in zirconium and its alloys". International Metals Reviews.
- (2019). "Hydrogen in zirconium alloys: A review". Journal of Nuclear Materials.
- Szydło, Z. A.. (2020). "Hydrogen - Some Historical Highlights". Chemistry-Didactics-Ecology-Metrology.
- Moers, K.. (1920). "Investigations on the Salt Character of Lithium Hydride". Zeitschrift für Anorganische und Allgemeine Chemie.
- (27 July 2004). "An extremely brief introduction to computational quantum chemistry". University of Michigan.
- Wilson, E. Bright. (1977). "Impact of the Heitler-London hydrogen molecule paper on chemistry". International Journal of Quantum Chemistry.
- (2003). ["History of industrial gases"]({{Google books). Kluwer Academic/Plenum Publishers.
- Kratz, Jessie. (2017-10-27). "Beyond the Hindenburg: Airships Throughout History".
- Follows, Mike. (July 2, 2015). "What ignited the Hindenburg?".
- Rappe, Mollie. (May 9, 2023). "Researchers switch from helium to hydrogen weather balloons".
- {{RubberBible87th
- Laidler, Keith J.. (1998). "Chemical kinetics". HarperCollins.
- (2003). "Organic chemistry: structure and function". W.H. Freeman and Co.
- "Structure and Nomenclature of Hydrocarbons". Purdue University.
- Laurence, Christian. (2010). "Lewis basicity and affinity scales: data and measurement". Wiley.
- A well-known hydride is [[lithium aluminium hydride]]: the {{chem2. [AlH4]-{{nbspanion carries hydridic centers firmly attached to the Al(III).{{Greenwood&Earnshaw2nd
- (October 1971). "Hydrogen Bonding". Annual Review of Physical Chemistry.
- IUPAC Compendium of Chemical Terminology, Electronic version, [http://goldbook.iupac.org/H02899.html Hydrogen Bond] {{Webarchive. link. (19 March 2008)
- (November 2024). "Dynamic-to-static switch of hydrogen bonds induces a metal–insulator transition in an organic–inorganic superlattice". Nature Chemistry.
- Punekar, Narayan S.. (2025). "ENZYMES: Catalysis, Kinetics and Mechanisms". Springer Nature Singapore.
- Tanabashi, M.. (2018). "Big-Bang Cosmology". [[Physical Review D]].
- (1970-12-31). "The Interstellar Medium". Harvard University Press.
- Dijkstra, Mark. (January 2014). "Lyα Emitting Galaxies as a Probe of Reionisation". Publications of the Astronomical Society of Australia.
- McCall Group. (22 April 2005). "H3+ Resource Center". Universities of Illinois and Chicago.
- Helm, H.. (2003). "Dissociative Recombination of Molecular Ions with Electrons". Department of Molecular and Optical Physics, University of Freiburg, Germany.
- (2003). "Inorganic Chemistry". Prentice Hall.
- (2023). "Structural basis for bacterial energy extraction from atmospheric hydrogen". Nature.
- (2023). "Developing high-affinity, oxygen-insensitive [NiFe]-hydrogenases as biocatalysts for energy conversion". Biochemical Society Transactions.
- Pearce, Fred. (January 25, 2024). "Natural Hydrogen: A Potential Clean Energy Source Beneath Our Feet".
- (13 July 2021). "Hydrogen Is One Answer to Climate Change. Getting It Is the Hard Part". The New York Times.
- (27 September 2022). "Is heating homes with hydrogen all but a pipe dream? An evidence review". Joule.
- (2008). "Introduction to Hydrogen Technology". John Wiley & Sons.
- (March 2021). "Dream or Reality? Electrification of the Chemical Process Industries". [[American Institute of Chemical Engineers]].
- (2021). "Industrial decarbonization via hydrogen: A critical and systematic review of developments, socio-technical systems and policy options". [[Energy Research & Social Science]].
- Funderburg, E.. (2008). "Why Are Nitrogen Prices So High?". The Samuel Roberts Noble Foundation.
- Hannula, Ilkka. (2015). "Co-production of synthetic fuels and district heat from biomass residues, carbon dioxide and electricity: Performance and cost analysis". Biomass and Bioenergy.
- (2014). "Nanoscale nickel oxide/Nickel heterostructures for active hydrogen evolution electrocatalysis". Nature Communications.
- (June 2021). "The role of hydrogen and ammonia in meeting the net zero challenge".
- Lees, A.. (2007). "Chemicals from salt". BBC.
- (2006-01-15). "Ullmann's Encyclopedia of Industrial Chemistry". Wiley-VCH Verlag GmbH & Co. KGaA.
- (30 November 2020). "In-depth Q&A: Does the world need hydrogen to solve climate change?".
- (2022-11-15). "The future of hydrogen: Challenges on production, storage and applications". Energy Conversion and Management.
- (2020). "State of the Art of Hydrogen Production via Pyrolysis of Natural Gas". Wiley Online Library.
- (2005). "Improved photobiological H2 production in engineered green algal cells". The Journal of Biological Chemistry.
- Williams, C.. (24 February 2006). "Pond life: the future of energy". The Register.
- (25 May 2005). "Development of solar-powered thermochemical production of hydrogen from water". Solar Thermochemical Hydrogen Generation Project.
- Perret, R.. "Development of Solar-Powered Thermochemical Production of Hydrogen from Water, DOE Hydrogen Program, 2007".
- (2007). "[NiFe] and [FeFe] Hydrogenases Studied by Advanced Magnetic Resonance Techniques". Chemical Reviews.
- (2016). "Genomic and metagenomic surveys of hydrogenase distribution indicate H2 is a widely utilised energy source for microbial growth and survival". The ISME Journal.
- (2001). "Hydrogen as a Fuel: Learning from Nature". Taylor & Francis Ltd.
- (19 May 2006). "The overwhelming role of soils in the global atmospheric hydrogen cycle". Atmospheric Chemistry and Physics.
- (Mar 8, 2023). "Soil bacteria enzyme generates electricity from hydrogen in the air". New Scientist.
- (2008). "Implementation and interpretation of hydrogen breath tests". Journal of Breath Research.
- (3 April 2007). "On Silica Activity and Serpentinization". Journal of Petrology.
- (14 September 2015). "Review and evaluation of hydrogen production methods for better sustainability". International Journal of Hydrogen Energy.
- (2004). "H2-rich fluids from serpentinization: Geochemical and biotic implications". Proceedings of the National Academy of Sciences of the United States of America.
- (2020). "About the Corrosion Mechanism of Metal Iron in Contact with Bentonite". ACS Earth and Space Chemistry.
- (2018). "Inorganic Chemistry". Prentice Hall.
- (2013). "Ullmann's Encyclopedia of Industrial Chemistry".
- (1988). "Hydrogen in amorphous and nanocrystalline metals". Materials Science and Engineering.
- Rogers, H. C.. (1999). "Hydrogen Embrittlement of Metals". [[Science (journal).
- (2023). "Ammonia borane-enabled hydrogen transfer processes: Insights into catalytic strategies and mechanisms". Green Energy & Environment.
- (2018). "NHC-Based Iridium Catalysts for Hydrogenation and Dehydrogenation of N-Heteroarenes in Water under Mild Conditions". ACS Catalysis.
- Barnard, Michael. (2023-10-22). "What's New On The Rungs Of Liebreich's Hydrogen Ladder?".
- (2001). "Kirk-Othmer Encyclopedia of Chemical Technology".
- (2011). "Ullmann's Encyclopedia of Industrial Chemistry". Wiley.
- (2004). "Enriching the Earth: Fritz Haber, Carl Bosch, and the Transformation of World Food Production". MIT.
- Chemistry Operations. (15 December 2003). "Hydrogen". Los Alamos National Laboratory.
- (2024). "A Recent Comprehensive Review of Fuel Cells: History, Types, and Applications". International Journal of Energy Research.
- Lewis, Alastair C.. (10 June 2021). "Optimising air quality co-benefits in a hydrogen economy: a case for hydrogen-specific standards for NO x emissions". Environmental Science: Atmospheres.
- Kjellberg-Motton, Brendan. (2022-02-07). "Steel decarbonisation gathers speed {{!}} Argus Media".
- IPCC. (2022). "Climate Change 2022: Mitigation of Climate Change". Cambridge University Press (In Press).
- (January 2020). "Hydrogen's Decarbonization Impact for Industry". [[Rocky Mountain Institute]].
- Plötz, Patrick. (2022-01-31). "Hydrogen technology is unlikely to play a major role in sustainable road transport". Nature Electronics.
- "NASA/TM—2002-211915: Solid Hydrogen Experiments for Atomic Propellants".
- (2020). "Using hydrogen and ammonia for renewable energy storage: A geographically comprehensive techno-economic study". [[Computers & Chemical Engineering]].
- [[IRENA]]. (2021). "World Energy Transitions Outlook: 1.5°C Pathway".
- Zimmerman, Albert H.. (2009). "Nickel-hydrogen batteries: principles and practice". Aerospace press.
- (July 2002). "Validation of international space station electrical performance model via on-orbit telemetry". IECEC '02. 2002 37th Intersociety Energy Conversion Engineering Conference, 2002.
- (2002). "Proceedings, IEEE Aerospace Conference".
- "Mars Global Surveyor". Astronautix.com.
- (7 May 2009). "Hubble servicing mission 4 essentials". NASA.
- (25 November 2008). "Extending Hubble's mission life with new batteries". NASA.
- Van de Walle, C. G.. (2000). "Hydrogen as a cause of doping in zinc oxide". Physical Review Letters.
- (2022-03-04). "A review of band structure and material properties of transparent conducting and semiconducting oxides: Ga2O3, Al2O3, In2O3, ZnO, SnO2, CdO, NiO, CuO, and Sc2O3". Applied Physics Reviews.
- (2002). "n-type doping of oxides by hydrogen". Applied Physics Letters.
- (2025-05-05). "Advanced Welding Technologies". Wiley.
- (June 2015). "Assessment of impact of hydrogen cooled generator on power system loadability enhancement". IEEE.
- National Electrical Manufacturers Association. (1946). "A chronological history of electrical development from 600 B.C". New York, N.Y., National Electrical Manufacturers Association.
- "Report from the Commission on Dietary Food Additive Intake". [[European Union]].
- (2025). "Re-evaluation of oxygen (E 948) and hydrogen (E 949) as food additives". EFSA Journal.
- (2025-03-01). "A comprehensive review of molecular hydrogen as a novel nutrition therapy in relieving oxidative stress and diseases: Mechanisms and perspectives". Biochemistry and Biophysics Reports.
- Bergeron, K. D.. (2004). "The Death of no-dual-use". Bulletin of the Atomic Scientists.
- "MyChem: Chemical".
- (2022-11-01). "Safety of hydrogen storage and transportation: An overview on mechanisms, techniques, and challenges". Energy Reports.
- (2021-09-03). "Review on hydrogen safety issues: Incident statistics, hydrogen diffusion, and detonation process". International Journal of Hydrogen Energy.
- (2016). "Hydrogen Detection Tape Saves Time and Lives {{!}} NASA Spinoff".
- (2021-09-01). "Review of hydrogen safety during storage, transmission, and applications processes". Journal of Loss Prevention in the Process Industries.
- U.S. Department of Energy. "Current Safe Operating Practices".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Hydrogen — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report