From Surf Wiki (app.surf) — the open knowledge base
Zincke aldehyde
Zincke aldehydes, or 5-aminopenta-2,4-dienals, are the product of the reaction of a pyridinium salt with two equivalents of any secondary amine, followed by basic hydrolysis. Using secondary amines (as opposed to primary amines) the Zincke reaction takes on a different shape forming Zincke aldehydes in which the pyridine ring is ring-opened with the terminal iminium group hydrolyzed to an aldehyde. The use of the dinitrophenyl group for pyridine activation was first reported by Theodor Zincke.{{cite journal
The synthesis and utility of Zincke aldehydes has been reviewed.{{cite journal
A variation of the Zincke reaction has been applied in the synthesis of novel indoles:
with cyanogen bromide mediated pyridine activation (König method).
More recently, an interesting rearrangement of Zincke aldehydes to Z-unsaturated amides was discovered serendipitously while trying to do an intramolecular Diels-Alder reaction.{{cite journal


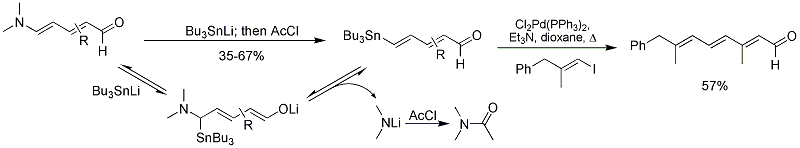
The Vanderwal group has also reported the synthesis of 4-stannyldienals from Zincke aldehydes by addition of tributylstannyl anion and quenching with acetyl chloride.{{cite journal
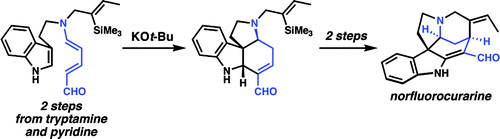
In 2009, the Vanderwal group reported another interesting rearrangement of Zincke aldehydes. Tryptamine-derived Zincke aldehydes are heated with strong base to give the rearranged enal as shown below. This reaction was the key step in their total synthesis of norfluorocurarine, a Strychnos alkaloid.{{cite journal

Also in 2009, the first reports of Zincke aldehydes undergoing a Pictet-Spengler reaction appeared from the group of Christian Marazano.{{cite journal
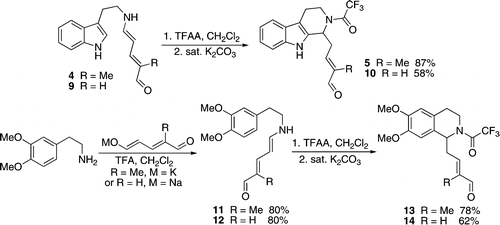
One drawback of the Zincke Aldehyde synthesis is the need for 2 equivalents of the amine in the initial pyridine ring opening reaction. This is of particular concern for the case of complex secondary amines required for natural product synthesis. The group of Marazano recently found an alternative synthesis by condensation onto a variety of glutaconaldehyde derivatives using TFA. This solution has greatly simplified the production and purification of complex Zincke aldehydes.
References
References
- (2006). "Synthesis of Nitrogen Heterocycles by the Ring Opening of Pyridinium Salts". [[Angew. Chem. Int. Ed.]].
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Zincke aldehyde — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report