From Surf Wiki (app.surf) — the open knowledge base
X-ray diffraction
Elastic interaction of x-rays with electrons
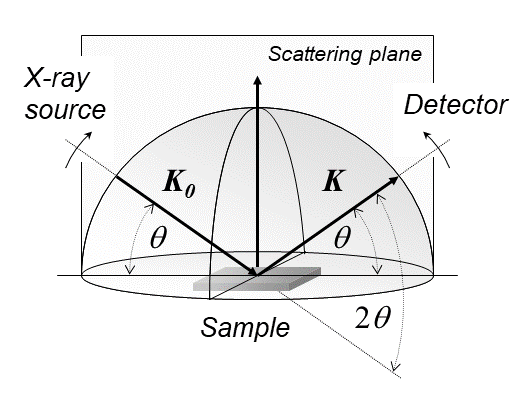
Elastic interaction of x-rays with electrons

X-ray diffraction is a generic term for phenomena associated with changes in the direction of X-ray beams due to interactions with the electrons around atoms. It occurs due to elastic scattering, when there is no change in the energy of the waves. The resulting map of the directions of the X-rays far from the sample is called a diffraction pattern. It is different from X-ray crystallography which exploits X-ray diffraction to determine the arrangement of atoms in materials, and also has other components such as ways to map from experimental diffraction measurements to the positions of atoms.
This article provides an overview of X-ray diffraction, starting with the early history of x-rays and the discovery that they have the right spacings to be diffracted by crystals. In many cases these diffraction patterns can be Interpreted using a single scattering or kinematical theory with conservation of energy (wave vector). Many different types of X-ray sources exist, ranging from ones used in laboratories to higher brightness synchrotron light sources. Similar diffraction patterns can be produced by related scattering techniques such as electron diffraction or neutron diffraction. If single crystals of sufficient size cannot be obtained, various other X-ray methods can be applied to obtain less detailed information; such methods include fiber diffraction, powder diffraction and (if the sample is not crystallized) small-angle X-ray scattering (SAXS).
History
When Wilhelm Röntgen discovered X-rays in 1895 physicists were uncertain of the nature of X-rays, but suspected that they were waves of electromagnetic radiation. The Maxwell theory of electromagnetic radiation was well accepted, and experiments by Charles Glover Barkla showed that X-rays exhibited phenomena associated with electromagnetic waves, including transverse polarization and spectral lines akin to those observed in the visible wavelengths. Barkla created the x-ray notation for sharp spectral lines, noting in 1909 two separate energies, at first, naming them "A" and "B" and, supposing that there may be lines prior to "A", he started an alphabet numbering beginning with "K." Single-slit experiments in the laboratory of Arnold Sommerfeld suggested that X-rays had a wavelength of about 1 angstrom. X-rays are not only waves but also have particle properties causing Sommerfeld to coin the name Bremsstrahlung for the continuous spectra when they were formed when electrons bombarded a material. Albert Einstein introduced the photon concept in 1905, but it was not broadly accepted until 1922, when Arthur Compton confirmed it by the scattering of X-rays from electrons. The particle-like properties of X-rays, such as their ionization of gases, had prompted William Henry Bragg to argue in 1907 that X-rays were not electromagnetic radiation. Bragg's view proved unpopular and the observation of X-ray diffraction by Max von Laue in 1912 confirmed that X-rays are a form of electromagnetic radiation. [[File:Interferenz-Erscheinungen_bei_Röntgenstrahlen_Tafel_II_Fig._5.jpg|thumb|One of the [[zinc blende]] X-ray interference patterns published in Von Laue's 1912 paper.]]
The idea that crystals could be used as a diffraction grating for X-rays arose in 1912 in a conversation between Paul Peter Ewald and Max von Laue in the English Garden in Munich. Ewald had proposed a resonator model of crystals for his thesis, but this model could not be validated using visible light, since the wavelength was much larger than the spacing between the resonators. Von Laue realized that electromagnetic radiation of a shorter wavelength was needed, and suggested that X-rays might have a wavelength comparable to the spacing in crystals. Von Laue worked with two technicians, Walter Friedrich and his assistant Paul Knipping, to shine a beam of X-rays through a copper sulfate crystal and record its diffraction pattern on a photographic plate. After being developed, the plate showed rings of fuzzy spots of roughly elliptical shape. Despite the crude and unclear image, the image confirmed the diffraction concept. The results were presented to the Bavarian Academy of Sciences and Humanities in June 1912 as "Interferenz-Erscheinungen bei Röntgenstrahlen" (Interference phenomena in X-rays).
After seeing the initial results, Laue was walking home and suddenly conceived of the physical laws describing the effect. Laue developed a law that connects the scattering angles and the size and orientation of the unit-cell spacings in the crystal, for which he was awarded the Nobel Prize in Physics in 1914.
After Von Laue's pioneering research the field developed rapidly, most notably by physicists William Lawrence Bragg and his father William Henry Bragg. In 1912–1913, the younger Bragg developed Bragg's law, which connects the scattering with evenly spaced planes within a crystal. The Braggs, father and son, shared the 1915 Nobel Prize in Physics for their work in crystallography. The earliest structures were generally simple; as computational and experimental methods improved over the next decades, it became feasible to deduce reliable atomic positions for more complicated arrangements of atoms; see X-ray crystallography for more details.
Introduction to x-ray diffraction theory
Basics
Crystals are regular arrays of atoms, and X-rays are electromagnetic waves. Atoms scatter X-ray waves, primarily through the atoms' electrons. Just as an ocean wave striking a lighthouse produces secondary circular waves emanating from the lighthouse, so an X-ray striking an electron produces secondary spherical waves emanating from the electron. This phenomenon is known as elastic scattering, and the electron (or lighthouse) is known as the scatterer. A regular array of scatterers produces a regular array of spherical waves. Although these waves cancel one another out in most directions through destructive interference, they add constructively in a few specific directions.
An intuitive understanding of X-ray diffraction can be obtained from the Bragg model of diffraction. In this model, a given reflection is associated with a set of evenly spaced sheets running through the crystal, usually passing through the centers of the atoms of the crystal lattice. The orientation of a particular set of sheets is identified by its three Miller indices (h, k, l), and their spacing by d. William Lawrence Bragg proposed a model where the incoming X-rays are scattered specularly (mirror-like) from each plane; from that assumption, X-rays scattered from adjacent planes will combine constructively (constructive interference) when the angle θ between the plane and the X-ray results in a path-length difference that is an integer multiple n of the X-ray wavelength λ. : 2 d\sin\theta = n\lambda. A reflection is said to be indexed when its Miller indices (or, more correctly, its reciprocal lattice vector components) have been identified from the known wavelength and the scattering angle 2θ. Such indexing gives the unit-cell parameters, the lengths and angles of the unit-cell, as well as its space group.
Ewald's sphere
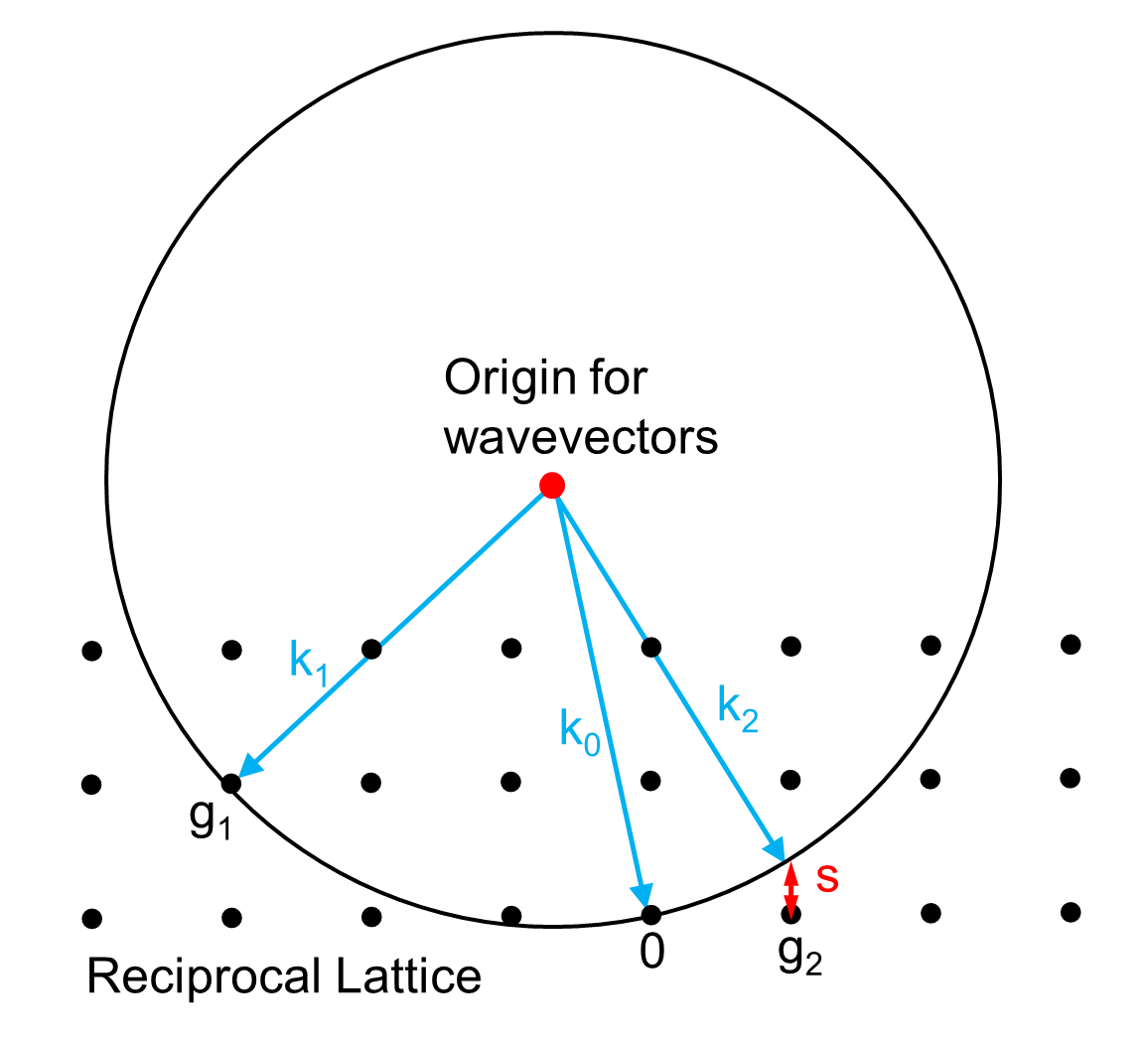
Each X-ray diffraction pattern represents a spherical slice of reciprocal space, as may be seen by the Ewald sphere construction. For a given incident wavevector k0 the only wavevectors with the same energy lie on the surface of a sphere. In the diagram, the wavevector k1 lies on the Ewald sphere and also is at a reciprocal lattice vector g1 so satisfies Bragg's law. In contrast the wavevector k2 differs from the reciprocal lattice point and g2 by the vector s which is called the excitation error. For large single crystals primarily used in crystallography only the Bragg's law case matters; for electron diffraction and some other types of x-ray diffraction non-zero values of the excitation error also matter.
Scattering amplitudes
X-ray scattering is determined by the density of electrons within the crystal. Since the energy of an X-ray is much greater than that of a valence electron, the scattering may be modeled as Thomson scattering, the elastic interaction of an electromagnetic ray with a charged particle.
The intensity of Thomson scattering for one particle with mass m and elementary charge q is: : I_o = I_e \left(\frac{q^4}{m^2c^4}\right)\frac{1+\cos^22\theta}{2} = I_e7.94\times10^{-26}\frac{1+\cos^22\theta}{2} = I_ef
Hence the atomic nuclei, which are much heavier than an electron, contribute negligibly to the scattered X-rays. Consequently, the coherent scattering detected from an atom can be accurately approximated by analyzing the collective scattering from the electrons in the system.
The incoming X-ray beam has a polarization and should be represented as a vector wave; however, for simplicity, it will be represented here as a scalar wave. We will ignore the time dependence of the wave and just concentrate on the wave's spatial dependence. Plane waves can be represented by a wave vector kin, and so the incoming wave at time t = 0 is given by : A \mathrm{e}^{2\pi \mathrm{i}\mathbf{k}_{\mathrm{in}} \cdot \mathbf{r}}.
At a position r within the sample, consider a density of scatterers f(r); these scatterers produce a scattered spherical wave of amplitude proportional to the local amplitude of the incoming wave times the number of scatterers in a small volume dV about r : \text{amplitude of scattered wave} = A \mathrm{e}^{2\pi \mathrm{i}\mathbf{k} \cdot \mathbf{r}} S f(\mathbf{r}) , \mathrm{d}V, where S is the proportionality constant.
Consider the fraction of scattered waves that leave with an outgoing wave-vector of kout and strike a screen (detector) at rscreen. Since no energy is lost (elastic, not inelastic scattering), the wavelengths are the same as are the magnitudes of the wave-vectors |kin| = |kout|. From the time that the photon is scattered at r until it is absorbed at rscreen, the photon undergoes a change in phase : e^{2\pi i \mathbf{k}\text{out} \cdot \left( \mathbf{r}\text{screen} - \mathbf{r} \right)}.
The net radiation arriving at rscreen is the sum of all the scattered waves throughout the crystal : A S \int \mathrm{d}\mathbf{r} , f(\mathbf{r}) \mathrm{e}^{2\pi \mathrm{i} \mathbf{k}\text{in} \cdot \mathbf{r}} e^{2\pi i \mathbf{k}\text{out} \cdot \left( \mathbf{r}\text{screen} - \mathbf{r} \right)} = A S e^{2\pi i \mathbf{k}\text{out} \cdot \mathbf{r}\text{screen}} \int \mathrm{d}\mathbf{r} , f(\mathbf{r}) \mathrm{e}^{2\pi \mathrm{i} \left( \mathbf{k}\text{in} - \mathbf{k}\text{out} \right) \cdot \mathbf{r}}, which may be written as a Fourier transform : A S \mathrm{e}^{2\pi \mathrm{i} \mathbf{k}\text{out} \cdot \mathbf{r}\text{screen}} \int d\mathbf{r} f(\mathbf{r}) \mathrm{e}^{-2\pi \mathrm{i} \mathbf{g} \cdot \mathbf{r}} = A S \mathrm{e}^{2\pi \mathrm{i} \mathbf{k}\text{out} \cdot \mathbf{r}_\text{screen}} F(\mathbf{g}), where g = kout – kin is a reciprocal lattice vector that satisfies Bragg's law and the Ewald construction mentioned above. The measured intensity of the reflection will be square of this amplitude : A^2 S^2 \left|F(\mathbf{g}) \right|^2. The above assumes that the crystalline regions are somewhat large, for instance microns across, but also not so large that the X-rays are scattered more than once. If either of these is not the case then the diffracted intensities will be more complicated.
X-ray sources
Rotating anode
Small scale diffraction experiments can be done with a local X-ray tube source, typically coupled with an image plate detector. These have the advantage of being relatively inexpensive and easy to maintain, and allow for quick screening and collection of samples. However, the wavelength of the X-rays produced is limited by the availability of different anode materials. Furthermore, the intensity is limited by the power applied and cooling capacity available to avoid melting the anode. In such systems, electrons are boiled off of a cathode and accelerated through a strong electric potential of ~50 kV; having reached a high speed, the electrons collide with a metal plate, emitting bremsstrahlung and some strong spectral lines corresponding to the excitation of inner-shell electrons of the metal. The most common metal used is copper, which can be kept cool easily due to its high thermal conductivity, and which produces strong Kα and Kβ lines. The Kβ line is sometimes suppressed with a thin (~10 μm) nickel foil. The simplest and cheapest variety of sealed X-ray tube has a stationary anode (the Crookes tube) and runs with ~2 kW of electron beam power. The more expensive variety has a rotating-anode type source that runs with ~14 kW of e-beam power.
X-rays are generally filtered (by use of X-ray filters) to a single wavelength (made monochromatic) and collimated to a single direction before they are allowed to strike the crystal. The filtering not only simplifies the data analysis, but also removes radiation that degrades the crystal without contributing useful information. Collimation is done either with a collimator (basically, a long tube) or with an arrangement of gently curved mirrors. Mirror systems are preferred for small crystals (under 0.3 mm) or with large unit cells (over 150 Å).
Microfocus tube
A more recent development is the microfocus tube, which can deliver at least as high a beam flux (after collimation) as rotating-anode sources but only require a beam power of a few tens or hundreds of watts rather than requiring several kilowatts.
Synchrotron radiation
Synchrotron radiation sources are some of the brightest light sources on earth and are some of the most powerful tools available for X-ray diffraction and crystallography. X-ray beams are generated in synchrotrons which accelerate electrically charged particles, often electrons, to nearly the speed of light and confine them in a (roughly) circular loop using magnetic fields.
Synchrotrons are generally national facilities, each with several dedicated beamlines where data is collected without interruption. Synchrotrons were originally designed for use by high-energy physicists studying subatomic particles and cosmic phenomena. The largest component of each synchrotron is its electron storage ring. This ring is not a perfect circle, but a many-sided polygon. At each corner of the polygon, or sector, precisely aligned magnets bend the electron stream. As the electrons' path is bent, they emit bursts of energy in the form of X-rays.
The intense ionizing radiation can cause radiation damage to samples, particularly macromolecular crystals. Cryo crystallography can protect the sample from radiation damage, by freezing the crystal at liquid nitrogen temperatures (~100 K). Cryocrystallography methods are applied to home source rotating anode sources as well. However, synchrotron radiation frequently has the advantage of user-selectable wavelengths, allowing for anomalous scattering experiments which maximizes anomalous signal. This is critical in experiments such as single wavelength anomalous dispersion (SAD) and multi-wavelength anomalous dispersion (MAD).
Free-electron laser
Free-electron lasers have been developed for use in X-ray diffraction and crystallography. These are the brightest X-ray sources currently available; with the X-rays coming in femtosecond bursts. The intensity of the source is such that atomic resolution diffraction patterns can be resolved for crystals otherwise too small for collection. However, the intense light source also destroys the sample, requiring multiple crystals to be shot. As each crystal is randomly oriented in the beam, hundreds of thousands of individual diffraction images must be collected in order to get a complete data set. This method, serial femtosecond crystallography, has been used in solving the structure of a number of protein crystal structures, sometimes noting differences with equivalent structures collected from synchrotron sources.
References
References
- (1 March 2022). "Structural biology: How proteins got their close-up". Knowable Magazine.
- Barkla, Charles G. (1911). "XXXIX.The spectra of the fluorescent Röntgen radiations". Philosophical Magazine. Series 6. 22 (129): 396–412. doi:10.1080/14786440908637137.
- Michael Eckert, Disputed discovery: the beginnings of X-ray diffraction in crystals in 1912 and its repercussions, January 2011, Acta crystallographica. Section A, Foundations of crystallography 68(1):30–39 This Laue centennial article has also been published in Zeitschrift für Kristallographie [Eckert (2012). Z. Kristallogr. 227, 27–35].
- Nisio, Sigeko. "The Formation of the Sommerfeld Quantum Theory of 1916." (1974) JSHS, No.12. pp39-78.
- (1905). "Über einen die Erzeugung und Verwandlung des Lichtes betreffenden heuristischen Gesichtspunkt". [[Annalen der Physik]].
- (1909). "Über die Entwicklung unserer Anschauungen über das Wesen und die Konstitution der Strahlung". Physikalische Zeitschrift.
- (1982). "Subtle is the Lord: The Science and the Life of Albert Einstein". [[Oxford University Press]].
- (1923). "A Quantum Theory of the Scattering of X-rays by Light Elements". Phys. Rev..
- (1907). "The nature of Röntgen rays". Transactions of the Royal Society of Science of Australia.
- (1908). ["The nature of γ- and X-rays"](https://zenodo.org/record/1429495}} See also {{cite journal). Nature.
- (1910). "The consequences of the corpuscular hypothesis of the γ- and X-rays, and the range of β-rays". Phil. Mag..
- (1912). "On the direct or indirect nature of the ionization by X-rays". Phil. Mag..
- (1912). "Interferenz-Erscheinungen bei Röntgenstrahlen". Sitzungsberichte der Mathematisch-Physikalischen Classe der Königlich-Bayerischen Akademie der Wissenschaften zu München.
- (1962). "Fifty Years of X-Ray Diffraction: Dedicated to the International Union of Crystallography on the Occasion of the Commemoration Meeting in Munich July 1962". Springer US.
- (1920). "Concerning the detection of x-ray interferences". Nobel Lectures, Physics.
- (1932). "A Textbook of Mineralogy". John Wiley & Sons.
- (1912). "The Specular Reflexion of X-rays". Nature.
- (1913). "The Diffraction of Short Electromagnetic Waves by a Crystal". Proceedings of the Cambridge Philosophical Society.
- (1914). "Die Reflexion der Röntgenstrahlen". Jahrbuch der Radioaktivität und Elektronik.
- (1952). "X-ray Crystallographic Technology". Hilger and Watts LTD..
- Cullity, B. D.. (2001). "Elements of x-ray diffraction". Prentice Hall.
- Authier, André. (2008). "Dynamical theory of X-ray diffraction". Oxford Univ. Press.
- (1997). "Macromolecular Cryocrystallography". Journal of Applied Crystallography.
- (June 2015). "Practical macromolecular cryocrystallography". Acta Crystallographica. Section F, Structural Biology Communications.
- (October 2012). "Emerging opportunities in structural biology with X-ray free-electron lasers". Current Opinion in Structural Biology.
- (August 2000). "Potential for biomolecular imaging with femtosecond X-ray pulses". Nature.
- (December 2013). "Serial femtosecond crystallography of G protein-coupled receptors". Science.
- Cowley, John M.. (1995). "Diffraction physics". Elsevier.
- Bethe, H.. (1928). "Theorie der Beugung von Elektronen an Kristallen". Annalen der Physik.
- (1987). "Low-energy electron diffraction". Springer-Verlag Berlin.
- Braun, Wolfgang. (1999). "Applied RHEED : reflection high-energy electron diffraction during crystal growth". Springer.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about X-ray diffraction — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report