From Surf Wiki (app.surf) — the open knowledge base
Wollastonite
Single chain calcium inosilicate (CaSiO<sub>3</sub>)
Single chain calcium inosilicate (CaSiO<sub>3</sub>)
| Field | Value |
|---|---|
| name | Wollastonite |
| category | Inosilicate mineral |
| image | WollastoniteUSGOV.jpg |
| formula | Calcium metasilicate, CaSiO3 |
| IMAsymbol | Wo |
| molweight | 116.159g/mol |
| strunz | 9.DG.05 |
| system | Triclinic |
| Monoclinic polytype exists | |
| class | Pinacoidal () |
| (same H-M symbol) | |
| symmetry | *P* (Triclinic) |
| unit cell | a = 7.925 Å, b = 7.32 Å, |
| c = 7.065 Å; α = 90.055°, | |
| β = 95.217°, γ = 103.42°; Z = 6 | |
| color | White, colorless or gray |
| habit | Rare as tabular crystals—commonly massive in lamellar, radiating, compact and fibrous aggregates. |
| twinning | Common |
| cleavage | Perfect in two directions at near 90° |
| fracture | Splintery to uneven |
| mohs | 4.5 to 5.0 |
| luster | Vitreous or dull to pearly on cleavage surfaces |
| refractive | nα = 1.616–1.640 |
| nβ = 1.628–1.650 | |
| nγ = 1.631–1.653 | |
| opticalprop | Biaxial (−) |
| birefringence | δ = 0.015 max |
| 2V | Measured: 36° to 60° |
| streak | White |
| gravity | 2.86–3.09 |
| other | Heat of formation (@298): −89.61kJ |
| melt | 1540 °C |
| solubility | Soluble in HCl, insoluble in water |
| diaphaneity | Transparent to translucent |
| references |
Monoclinic polytype exists (same H-M symbol)
P21/a (Monoclinic) c = 7.065 Å; α = 90.055°, β = 95.217°, γ = 103.42°; Z = 6 nβ = 1.628–1.650 nγ = 1.631–1.653
Gibbs free energy: 41.78kJ Wollastonite is a calcium inosilicate mineral (CaSiO3) that may contain small amounts of iron, magnesium, and manganese substituting for calcium. It is usually white. It forms when impure limestone or dolomite is subjected to high temperature and pressure, which sometimes occurs in the presence of silica-bearing fluids as in skarns or in contact with metamorphic rocks. Associated minerals include garnets, vesuvianite, diopside, tremolite, epidote, plagioclase feldspar, pyroxene and calcite. It is named after the English chemist and mineralogist William Hyde Wollaston (1766–1828).
Despite its chemical similarity to the compositional spectrum of the pyroxene group of minerals—where magnesium (Mg) and iron (Fe) substitution for calcium ends with diopside and hedenbergite respectively—it is structurally very different, with a third tetrahedron in the linked chain (as opposed to two in the pyroxenes).
Production trends
Estimated world production of crude wollastonite ore was 1,200,000 tonnes in 2021. World reserves of wollastonite are estimated to exceed 100 million tonnes, though some existing deposits have not been surveyed.
Major producers of wollastonite include China, India, the United States, Mexico, and Finland.
In the United States, wollastonite is mined in Willsboro, New York (the first laboratory for local wollastonite research was in Essex, New York by Koert Burnham in the 1940s. The original laboratory building still exists as a residential & commercial building) and Gouverneur, New York. Deposits have also been mined commercially in North Western Mexico.
The price of raw wollastonite in 2008 varied between US$80 and US$500 per tonne depending on the country and size and shape of the powder particles.
Uses
Wollastonite is among the fastest reacting silicates, but may have high costs associated with carbon storage. Addition of wollastonite to soil stimulates organic carbon mineralization.
Ceramics
Wollastonite has industrial importance in ceramics manufacturing as an additive.
In ceramics, wollastonite decreases shrinkage and gas evolution during firing, increases green and fired strength, maintains brightness during firing, permits fast firing, and reduces crazing, cracking, and glaze defects.
Construction
Wollastonite can serve as a substitute for asbestos in floor tiles, friction products, insulating board and panels, paint, plastics, and roofing products. Similar to asbestos, wollastonite is resistant to chemical attack, stable at high temperatures, and improves flexural and tensile strength in composites. In some industries, wollastonite is used in different percentages of impurities, such as its use as a fabricator of mineral wool insulation, or as an ornamental building material. Wollastonite is used in a cement announced in 2019 which "reduces the overall carbon footprint in precast concrete by 70%."
Wollastonite has been studied for carbon mineralization for storage of carbon dioxide (CO2) according to the following reaction: :
Metallurgy
In metallurgical applications, wollastonite serves as a flux for welding, a source for calcium oxide, a slag conditioner, and to protect the surface of molten metal during the continuous casting of steel.
Paint
As an additive in paint, wollastonite improves the durability of the paint film, acts as a pH buffer, improves its resistance to weathering, reduces gloss, reduces pigment consumption, and acts as a flatting and suspending agent.
Plastic
In plastics, wollastonite improves tensile and flexural strength, reduces resin consumption, and improves thermal and dimensional stability at elevated temperatures. Surface treatments are used to improve the adhesion between the wollastonite and the polymers to which it is added.
Plastics and rubber applications were estimated to account for 25% to 35% of U.S. sales in 2009, followed by ceramics with 20% to 25%; paint, 10% to 15%; metallurgical applications, 10% to 15%; friction products, 10% to 15%; and miscellaneous, 10% to 15%. Ceramic applications probably account for 30% to 40% of wollastonite sales worldwide, followed by polymers (plastics and rubber) with 30% to 35% of sales, and paint with 10% to 15% of sales. The remaining sales were for construction, friction products, and metallurgical applications.
Substitutes

The acicular nature of many wollastonite products allows it to compete with other acicular materials, such as ceramic fiber, glass fiber, steel fiber, and several organic fibers, such as aramid, polyethylene, polypropylene, and polytetrafluoroethylene in products where improvements in dimensional stability, flexural modulus, and heat deflection are sought.
Wollastonite also competes with several nonfibrous minerals or rocks, such as kaolin, mica, and talc, which are added to plastics to increase flexural strength, and such minerals as barite, calcium carbonate, gypsum, and talc, which impart dimensional stability to plastics.
In ceramics, wollastonite competes with carbonates, feldspar, lime, and silica as a source of calcium and silicon. Its use in ceramics depends on the formulation of the ceramic body and the firing method.
Composition
In a pure CaSiO3, each component forms nearly half of the mineral by weight: 48.3% of CaO and 51.7% of SiO2. In some cases, small amounts of iron (Fe), and manganese (Mn), and lesser amounts of magnesium (Mg) substitute for calcium (Ca) in the mineral formula (e.g., rhodonite). Wollastonite can form a series of solid solutions in the system CaSiO3-FeSiO3, or hydrothermal synthesis of phases in the system MnSiO3-CaSiO3.
Geologic occurrence
Wollastonite usually occurs as a common constituent of a thermally metamorphosed impure limestone, it also could occur when the silicon is due to metamorphism in contact altered calcareous sediments, or to contamination in the invading igneous rock. In most of these occurrences it is the result of the following reaction between calcite and silica with the loss of carbon dioxide:
:CaCO3 + SiO2 → CaSiO3 + CO2
Wollastonite may also be produced in a diffusion reaction in skarn, it develops when limestone within a sandstone is metamorphosed by a dike, which results in the formation of wollastonite in the sandstone as a result of outward migration of Ca.
Structure

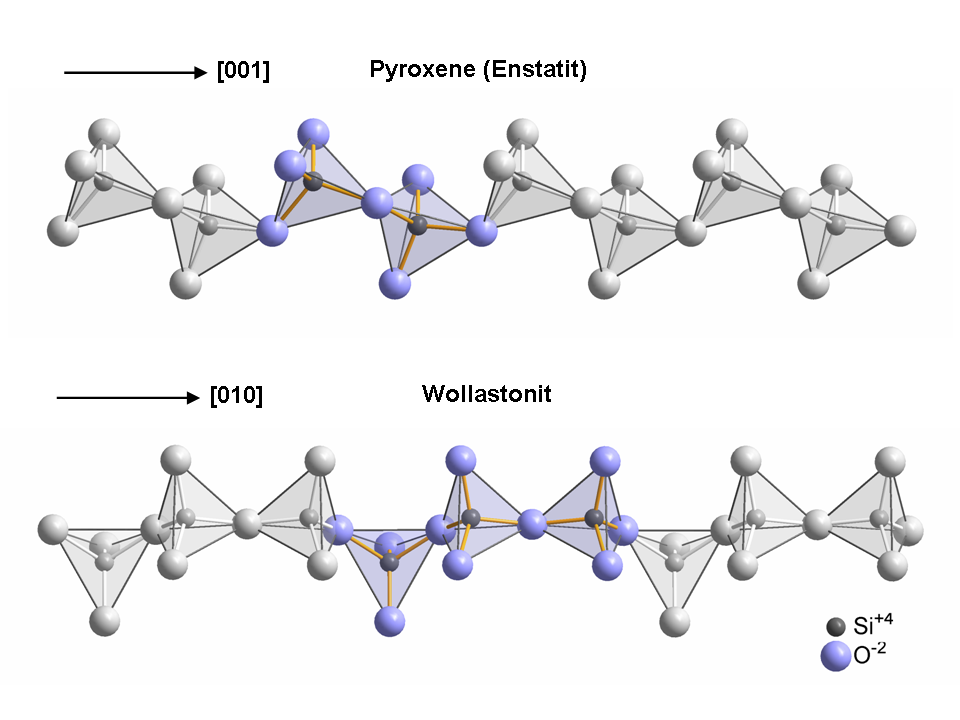
Wollastonite crystallizes triclinically in space group P with the lattice constants a = 7.94 Å, b = 7.32 Å, c = 7.07 Å; α = 90,03°, β = 95,37°, γ = 103,43° and six formula units per unit cell. Wollastonite was once classed structurally among the pyroxene group, because both of these groups have a ratio of Si:O = 1:3. In 1931, Warren and Biscoe showed that the crystal structure of wollastonite differs from minerals of the pyroxene group, and they classified this mineral within a group known as the pyroxenoids. It has been shown that the pyroxenoid chains are more kinked than those of pyroxene group, and exhibit longer repeat distance. The structure of wollastonite contains infinite chains of [SiO4] tetrahedra sharing common vertices, running parallel to the b-axis. The chain motif in wollastonite repeats after three tetrahedra, whereas in pyroxenes only two are needed. The repeat distance in the wollastonite chains is 7.32 Å and equals the length of the crystallographic b-axis.
Molten CaSiO3 maintains a tetrahedral SiO4 local structure at temperatures up to 2000 °C. The nearest neighbor Ca-O coordination decreases from 6.0(2) in the room temperature glass to 5.0(2) in the 1700 °C liquid, coincident with an increasing number of longer Ca-O neighbors.
References
References
- Warr, L.N.. (2021). "IMA–CNMNC approved mineral symbols". Mineralogical Magazine.
- [http://www.mindat.org/min-4323.html Wollastonite], Mindat
- [http://webmineral.com/data/Wollastonite-1A.shtml Wollastonite], Webmineral
- [http://rruff.geo.arizona.edu/doclib/hom/wollastonite.pdf Wollastonite], ''Handbook of Mineralogy''
- [http://www.minsocam.org/ammin/am79/am79_134.pdf American Mineralogist, V. 79, pp. 134-144, 1994]
- (January 27, 2015). "Mineral Resource of the Month: Wollastonite". American Geosciences Institute.
- (2020-10-18). "Magmatic and Metasomatic Effects of Magma–Carbonate Interaction Recorded in Calc-silicate Xenoliths from Merapi Volcano (Indonesia)". Journal of Petrology.
- (1992). "An introduction to the rock-forming minerals". Longman Scientific & Technical.
- [https://pubs.usgs.gov/periodicals/mcs2022/mcs2022-wollastonite.pdf Wollastonite], ''Mineral Commodity Summaries'' 2021
- Robert L. Virta [https://minerals.usgs.gov/minerals/pubs/commodity/wollastonite/myb1-2009-wolla.pdf Wollastonite], ''USGS 2009 Minerals Yearbook'' (October 2010)
- National Academies of Sciences, Engineering, and Medicine. (2019). "Negative Emissions Technologies and Reliable Sequestration: A Research Agenda". The National Academies Press.
- (2023). "Wollastonite addition stimulates soil organic carbon mineralization: Evidences from 12 land-use types in subtropical China". Catena.
- Deer, Howie and Zussman. ''Rock Forming Minerals; Single Chain Silicates'', Vol. 2A, Second Edition, London, The Geological Society, 1997.
- Andrews, R. W. (1970). ''Wollastonite''. London, Her Majesty's Stationery Office.
- Alter, Lloyd. (August 15, 2019). "LafargeHolcim is selling CO2-sucking cement for precast, reduces emissions by 70 percent".
- (January 2019). "Co-Benefits of Wollastonite Weathering in Agriculture: CO2 Sequestration and Promoted Plant Growth". ACS Omega.
- (1961). "The crystal structures of wollastonite and pectolite". Proceedings of the National Academy of Sciences.
- Benmore, C.J.. (2010). "Temperature-dependent structural heterogeneity in calcium silicate liquids". Phys. Rev. B.
- Skinner, L.B.. (2012). "Structure of molten CaSiO3: Neutron diffraction isotope substitution with aerodynamic levitation and molecular dynamics Study". J. Phys. Chem. B.
- Eckersley, M.C.. (1988). "Structural ordering in a calcium silicate glass". Nature.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Wollastonite — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report