From Surf Wiki (app.surf) — the open knowledge base
Uranyl chloride
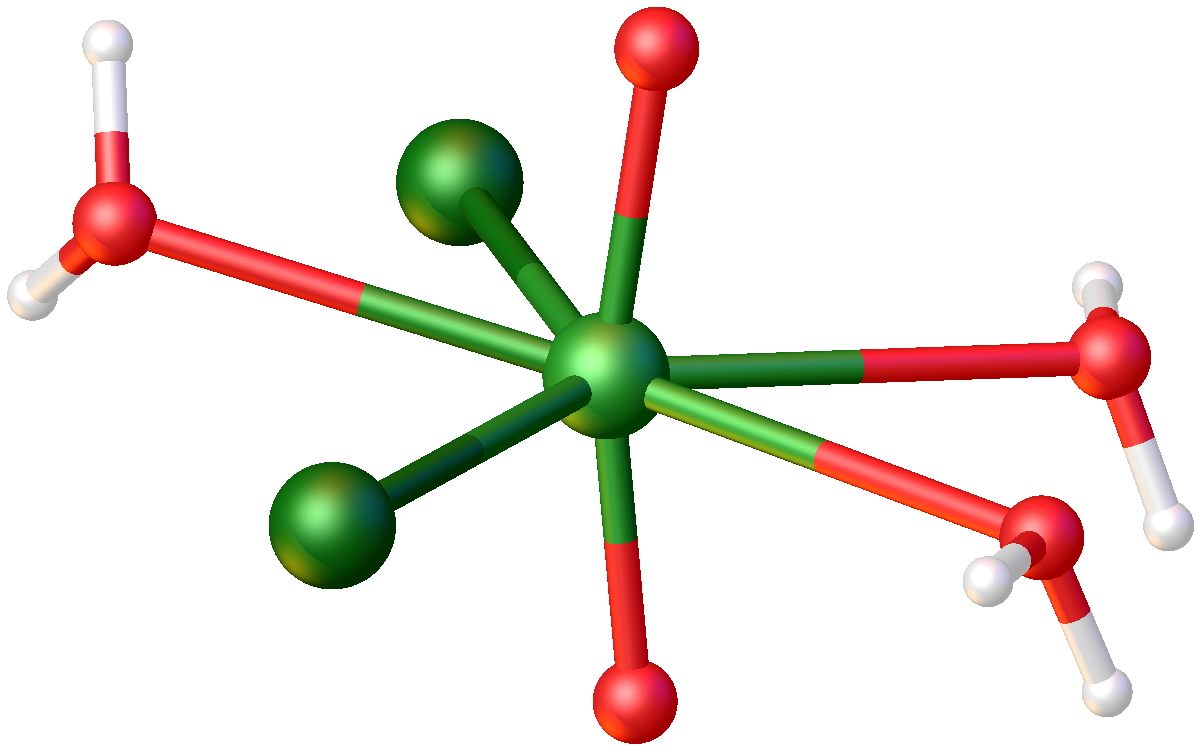
Uranium(VI) dichloride dioxide Uranyl chloride is a chemical compound with the chemical formula . It consists of uranyl cations and chloride anions . It is fluorescent. Uranyl chloride also refers to inorganic compounds with the formula where n = 0, 1, 2, or 3. These are yellow salts.
Synthesis and structures

The hydrates are obtained by dissolving uranyl sulfate or uranyl acetate in hydrochloric acid followed by crystallization from concentrated solutions. Depending on the method of drying, one obtains the mono- or the trihydrate. The monohydrate is described as a yellow, sulfur-like powder. It is very hygroscopic. The trihydrate is greenish-yellow. Both hydrates are fluorescent solids that are highly soluble in water.
The anhydrous material can be obtained by the reaction of oxygen with uranium tetrachloride: :
In terms of structures, all three of these compounds feature the uranyl center (trans-) bound to five additional ligands, which can include (bridging) chloride, water, or another uranyl oxygen.
Reactions
The aquo ligands can be replaced by a variety of donors, e.g. THF. Uranyl chloride, and its two hydrates, ( and ) decompose in the presence of light. This photosensitivity over the years, from time to time, has attracted scientific interest and there have been various unsuccessful attempts to develop applications in photography using these compounds.
Industrial importance
The company Indian Rare Earths Limited (IREL) has developed a process to extract uranium from the Western and Eastern coastal dune sands of India. After pre-processing with high-intensity magnetic separators and fine grinding, the mineral sands (known as monazite), are digested with caustic soda at about 120 C and water. The hydroxide concentrate is further digested with concentrated hydrochloric acid to solubilise all hydroxides to form a feed solution composed of chlorides of uranium, rare earth elements and thorium. The solution is subjected to liquid–liquid extraction with dual solvent systems to produce uranyl chloride and thorium oxalate. The crude uranyl chloride solution is subsequently refined to nuclear grade ammonium diuranate by a purification process involving precipitation and solvent extraction in a nitrate media.
Safety
Uranyl chloride is highly toxic by ingestion and inhalation. Cumulative toxic effects are also probable, with the target organs being the liver and the kidneys. It is toxic to aquatic organisms, and may cause long-term catastrophic effects in the aquatic environment. As all uranium compounds, this salt is radioactive.
References
References
- https://www.2spi.com/item/z02411/#:~:text=Uranyl%20chloride%20(UO2Cl,yellow%20colored%20compound%20of%20uranium.
- (1968). "The structures of uranyl chloride and its hydrates". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry.
- (1963). "Inorganic Syntheses".
- F. Hein, S. Herzog. (1963). "Handbook of Preparative Inorganic Chemistry, 2nd Ed.". Academic Press.
- (1973). "The Structure of Anhydrous Uranyl Chloride by Powder Neutron Diffraction". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry.
- (1957). "Inorganic Syntheses".
- (1999). "Synthesis and Crystal Structure of UO2Cl2(THF)3: A Simple Preparation of an Anhydrous Uranyl Reagent". Inorganic Chemistry.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Uranyl chloride — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report