From Surf Wiki (app.surf) — the open knowledge base
Tryptamine
Metabolite of the amino acid tryptophan

Metabolite of the amino acid tryptophan
| Field | Value | ||
|---|---|---|---|
| image | Tryptamine structure.svg | ||
| image_class | skin-invert-image | ||
| width | 225px | ||
| image2 | Tryptamine molecule from xtal ball.png | ||
| image_class2 | bg-transparent | ||
| width2 | 175px | ||
| routes_of_administration | Intravenous injection | ||
| class | Serotonin receptor agonist; Trace amine-associated receptor 1 (TAAR1) agonist; Serotonin–norepinephrine–dopamine releasing agent; Serotonergic psychedelic; Hallucinogen | ||
| ATC_prefix | None | ||
| legal_status | Legal or unregulated | ||
| bioavailability | Oral: Very low | ||
| metabolism | Very rapid (oxidative deamination by ) | ||
| metabolites | Indole-3-acetic acid (IAA) | ||
| onset | : Very rapid | ||
| elimination_half-life | Very short | ||
| duration_of_action | : Very short | ||
| excretion | Urine | ||
| CAS_number_Ref | |||
| CAS_number | 61-54-1 | ||
| PubChem | 1150 | ||
| IUPHAR_ligand | 125 | ||
| DrugBank | DB08653 | ||
| ChemSpiderID_Ref | |||
| ChemSpiderID | 1118 | ||
| UNII_Ref | |||
| UNII | 422ZU9N5TV | ||
| KEGG | C00398 | ||
| ChEBI | 16765 | ||
| ChEMBL_Ref | |||
| ChEMBL | 6640 | ||
| synonyms | T; Triptamine; 3-(2-Aminoethyl)indole; 2-(3-Indolyl)ethylamine; Indolylethylamine; Indolethylamine; PAL-235; PAL235 | ||
| IUPAC_name | 2-(1*H*-indol-3-yl)ethan-1-amine | ||
| C | 10 | H=12 | N=2 |
| SMILES | c1ccc2c(c1)c(c[nH]2)CCN | ||
| StdInChI_Ref | |||
| StdInChI | 1S/C10H12N2/c11-6-5-8-7-12-10-4-2-1-3-9(8)10/h1-4,7,12H,5-6,11H2 | ||
| StdInChIKey_Ref | |||
| StdInChIKey | APJYDQYYACXCRM-UHFFFAOYSA-N | ||
| melting_point | 118 | ||
| melting_notes | |||
| boiling_point | 137 | ||
| boiling_notes | (0.15 mmHg) | ||
| solubility | Negligible |
the specific substance
| Drugs.com =
| elimination_half-life = Very short
Tryptamine, also known as 2-(3-indolyl)ethylamine, is an indolamine metabolite of the essential amino acid tryptophan. The chemical structure is defined by an indole—a fused benzene and pyrrole ring, and a 2-aminoethyl group at the second carbon (third aromatic atom, with the first one being the heterocyclic nitrogen). The structure of tryptamine is a shared feature of certain aminergic neuromodulators including melatonin, serotonin, bufotenin and psychedelic derivatives such as dimethyltryptamine (DMT), psilocybin, psilocin and others.
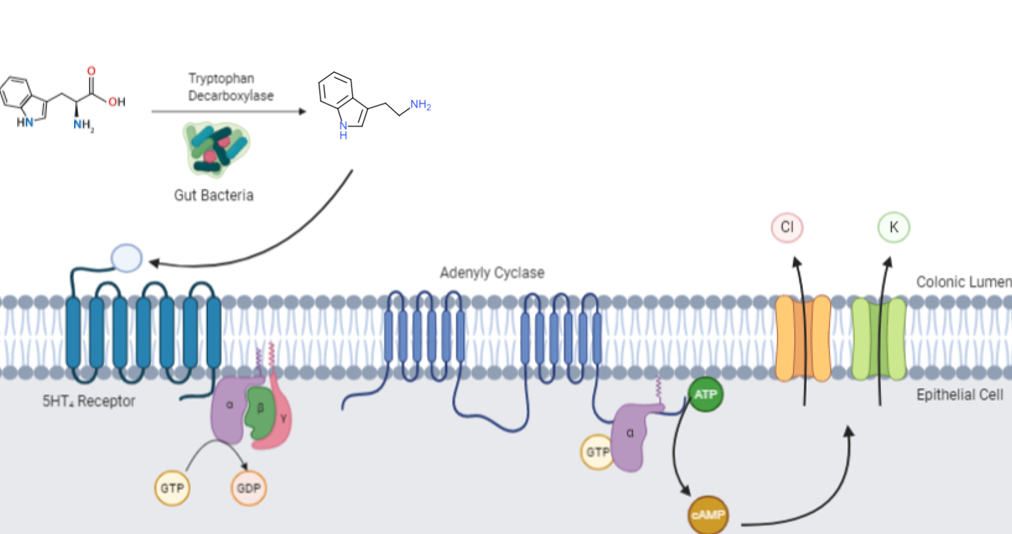
Tryptamine has been shown to activate serotonin receptors and trace amine-associated receptors expressed in the mammalian brain, and regulates the activity of dopaminergic, serotonergic and glutamatergic systems. In the human gut, bacteria convert dietary tryptophan to tryptamine, which activates 5-HT4 receptors and regulates gastrointestinal motility.
Multiple tryptamine-derived drugs have been developed to treat migraines, while trace amine-associated receptors are being explored as a potential treatment target for neuropsychiatric disorders.
Natural occurrences
For a list of plants, fungi and animals containing tryptamines, see List of psychoactive plants and List of naturally occurring tryptamines.
Mammalian brain
Endogenous levels of tryptamine in the mammalian brain are less than 100 ng per gram of tissue. However, elevated levels of trace amines have been observed in patients with certain neuropsychiatric disorders taking medications, such as bipolar depression and schizophrenia.
Mammalian gut microbiome
Tryptamine is relatively abundant in the gut and feces of humans and rodents. Commensal bacteria, including Ruminococcus gnavus and Clostridium sporogenes in the gastrointestinal tract, possess the enzyme tryptophan decarboxylase, which aids in the conversion of dietary tryptophan to tryptamine. Tryptamine is a ligand for gut epithelial serotonin type 4 (5-HT4) receptors and regulates gastrointestinal electrolyte balance through colonic secretions.
Metabolism
Biosynthesis
To yield tryptamine in vivo, tryptophan decarboxylase removes the carboxylic acid group on the α-carbon of tryptophan. Synthetic modifications to tryptamine can produce serotonin and melatonin; however, these pathways do not occur naturally as the main pathway for endogenous neurotransmitter synthesis.
Catabolism
Monoamine oxidases A and B are the primary enzymes involved in tryptamine metabolism to produce indole-3-acetaldehyde, however it is unclear which isoform is specific to tryptamine degradation.
Figure

Biological activity
| Target | Affinity (Ki, nM) | Species |
|---|---|---|
| [5-HT1A](5-ht1a-receptor) | 32–105 (Ki) | |
| 899–10,000 () | ||
| ND () | Human | |
| Human | ||
| Human | ||
| [5-HT1B](5-ht1b-receptor) | 36–525 | Human |
| [5-HT1D](5-ht1d-receptor) | 23–521 | Human |
| [5-HT1E](5-ht1e-receptor) | 2,559 | Human |
| [5-HT1F](5-ht1f-receptor) | 2,409 | Human |
| [5-HT2A](5-ht2a-receptor) | 37–4,070 (Ki) | |
| 7.4–257 (EC50) | ||
| 71–104% (Emax) | Human | |
| Human | ||
| Human | ||
| [5-HT2B](5-ht2b-receptor) | 25–113 (Ki) | |
| 29.5 (EC50) | ||
| 92% (Emax) | Human | |
| Human | ||
| Human | ||
| [5-HT2C](5-ht2c-receptor) | 17–3,000 (Ki) | |
| 1.17–45.7 (EC50) | ||
| 85–108% (Emax) | Human | |
| Human | ||
| Human | ||
| [5-HT3](5-ht3-receptor) | ND | ND |
| [5-HT4](5-ht4-receptor) | 10,000 | |
| 13,500 (EC50) | ||
| 96% (Emax) | Mouse | |
| Pig | ||
| Pig | ||
| [5-HT5A](5-ht5a-receptor) | ND | ND |
| [5-HT6](5-ht6-receptor) | 70–438 | Human |
| [5-HT7](5-ht7-receptor) | 148–158 | Human |
| α2A | 19,000 | Rat |
| 1,400 (Ki) | ||
| 2,700 (EC50) | ||
| 117% (Emax) | ||
| 130 (Ki) | ||
| 410 (EC50) | ||
| 91% (Emax) | ||
| 1,084 (Ki) | ||
| 2,210–21,000 (EC50) | ||
| 73% (Emax) | Mouse | |
| Mouse | ||
| Mouse | ||
| Rat | ||
| Rat | ||
| Rat | ||
| Human | ||
| Human | ||
| Human | ||
| 32.6 (EC50) a | Rat | |
| 716 (EC50) a | Rat | |
| 164 (EC50) a | Rat | |
| **Note:** The smaller the value, the more avidly the compound binds to or activates the site. **Footnotes:** a = Neurotransmitter release. **Refs:** *Main:* *Additional:* |
Serotonin receptor agonist
Tryptamine is known to act as a serotonin receptor agonist, although its potency is limited by rapid inactivation by monoamine oxidases. It has specifically been found to act as a full agonist of the serotonin 5-HT2A receptor ( = 7.36 ± 0.56nM; Emax = 104 ± 4%). Tryptamine was of much lower potency in stimulating the 5-HT2A receptor β-arrestin pathway (EC50 = 3,485 ± 234nM; Emax = 108 ± 16%). In contrast to the 5-HT2A receptor, tryptamine was found to be inactive at the serotonin 5-HT1A receptor.
Gastrointestinal motility
Tryptamine produced by mutualistic bacteria in the human gut activates serotonin GPCRs ubiquitously expressed along the colonic epithelium. Upon tryptamine binding, the activated 5-HT4 receptor undergoes a conformational change which allows its Gs alpha subunit to exchange GDP for GTP, and its liberation from the 5-HT4 receptor and βγ subunit. GTP-bound Gs activates adenylyl cyclase, which catalyzes the conversion of ATP into cyclic adenosine monophosphate (cAMP). cAMP opens chloride and potassium ion channels to drive colonic electrolyte secretion and promote intestinal motility.
Monoamine releasing agent
Tryptamine has been found to act as a monoamine releasing agent (MRA). It is a releaser of serotonin, dopamine, and norepinephrine, in that order of potency (EC50 = 32.6nM, 164nM, and 716nM, respectively). That is, it acts as a serotonin–norepinephrine–dopamine releasing agent (SNDRA).
| Compound | Ref |
|---|---|
| Tryptamine | 32.6 |
| Serotonin | 44.4 |
| Phenethylamine | 10,000 |
| Tyramine | 2,775 |
| [5-Methoxytryptamine](5-methoxytryptamine) | 2,169 |
| *N*-Methyltryptamine | 22.4 |
| Dimethyltryptamine | 114 |
| Psilocin | 561 |
| Bufotenin | 30.5 |
| [5-MeO-DMT](5-meo-dmt) | 10,000 |
| α-Methyltryptamine | 21.7–68 |
| α-Ethyltryptamine | 23.2 |
| D-Amphetamine | 698–1,765 |
| **Notes:** The smaller the value, the more strongly the drug releases the neurotransmitter. The assays were done in rat brain synaptosomes and human potencies may be different. See also Monoamine releasing agent § Activity profiles for a larger table with more compounds. **Refs:** |
Monoaminergic activity enhancer
Tryptamine is a monoaminergic activity enhancer (MAE) of serotonin, norepinephrine, and dopamine in addition to its serotonin receptor agonism. That is, it enhances the action potential-mediated release of these monoamine neurotransmitters. The MAE actions of tryptamine and other MAEs may be mediated by TAAR1 agonism. Synthetic and more potent MAEs like benzofuranylpropylaminopentane (BPAP) and indolylpropylaminopentane (IPAP) have been derived from tryptamine.
TAAR1 agonist
Tryptamine is an agonist of the trace amine-associated receptor 1 (TAAR1). It is a potent TAAR1 full agonist in rats, a weak TAAR1 full agonist in mice, and a very weak TAAR1 partial agonist in humans. Tryptamine may act as a trace neuromodulator in some species via activation of TAAR1 signaling.
The TAAR1 is a stimulatory G protein-coupled receptor (GPCR) that is weakly expressed in the intracellular compartment of both pre- and postsynaptic neurons. TAAR1 agonists have been implicated in regulating monoaminergic neurotransmission, for instance by activating G protein-coupled inwardly-rectifying potassium channels (GIRKs) and reducing neuronal firing via facilitation of membrane hyperpolarization through the efflux of potassium ions.
TAAR1 agonists are under investigation as a novel treatment for neuropsychiatric conditions like schizophrenia, drug addiction, and depression. The TAAR1 is expressed in brain structures associated with dopamine systems, such as the ventral tegmental area (VTA) and serotonin systems in the dorsal raphe nuclei (DRN). Additionally, the human TAAR1 gene is localized at 6q23.2 on the human chromosome, which is a susceptibility locus for mood disorders and schizophrenia. Activation of TAAR1 suggests a potential novel treatment for neuropsychiatric disorders, as TAAR1 agonists produce antipsychotic-like, anti-addictive, and antidepressant-like effects in animals.
| Compound | Human TAAR1 | Mouse TAAR1 | Rat TAAR1 | EC50 (nM) | Ki (nM) | EC50 (nM) | Ki (nM) | EC50 (nM) | Ki (nM) |
|---|---|---|---|---|---|---|---|---|---|
| Tryptamine | 21,000 | N/A | 2,700 | 1,400 | 410 | 130 | |||
| Serotonin | 50,000 | N/A | 50,000 | N/A | 5,200 | N/A | |||
| Psilocin | 30,000 | N/A | 2,700 | 17,000 | 920 | 1,400 | |||
| Dimethyltryptamine | 10,000 | N/A | 1,200 | 3,300 | 1,500 | 22,000 | |||
| **Notes:** (1) EC50 and Ki values are in nanomolar (nM). (2) EC50 reflects the concentration required to elicit 50% of the maximum TAAR1 response. (3) The smaller the Ki value, the stronger the compound binds to the receptor. |
Effects in animals and humans
In a published clinical study, tryptamine, at a total dose of 23 to 277mg by intravenous infusion, produced hallucinogenic effects or perceptual disturbances similar to those of small doses of lysergic acid diethylamide (LSD). It also produced other LSD-like effects, including pupil dilation, increased blood pressure, and increased force of the patellar reflex. Tryptamine produced side effects including nausea, vomiting, dizziness, tingling sensations, sweating, and bodily heaviness among others as well. Conversely, there were no changes in heart rate or respiratory rate. The onset of the effects was rapid and the duration was very short. This can be attributed to the very rapid metabolism of tryptamine by monoamine oxidase (MAO) and its very short elimination half-life.
In animals, tryptamine, alone and/or in combination with a monoamine oxidase inhibitor (MAOI), produces behavioral changes such as hyperlocomotion and reversal of reserpine-induced behavioral depression. In addition, it produces effects like hyperthermia, tachycardia, myoclonus, and seizures or convulsions, among others. Findings on tryptamine and the head-twitch response in rodents have been mixed, with some studies reporting no effect, some studies reporting induction of head twitches by tryptamine, and others reporting that tryptamine actually antagonized 5-hydroxytryptophan (5-HTP)-induced head twitches. Another study found that combination of tryptamine with an MAOI dose-dependently produced head twitches. Head twitches in rodents are a behavioral proxy of psychedelic-like effects. Many of the effects of tryptamine can be reversed by serotonin receptor antagonists like metergoline, metitepine (methiothepin), and cyproheptadine. Conversely, the effects of tryptamine in animals are profoundly augmented by MAOIs due to inhibition of its metabolism.
Tryptamine seems to also elevate prolactin and cortisol levels in animals and/or humans.
The values of tryptamine in animals include 100mg/kg i.p. in mice, 500mg/kg s.c. in mice, and 223mg/kg i.p. in rats.
Pharmacokinetics
Tryptamine produced endogenously or administered peripherally is readily able to cross the blood–brain barrier and enter the central nervous system. This is in contrast to serotonin, which is peripherally selective.
Tryptamine is metabolized by monoamine oxidase (MAO) to form indole-3-acetic acid (IAA). Its metabolism is described as extremely rapid and its elimination half-life and duration as very short. In addition, its duration is described as shorter than that of dimethyltryptamine (DMT). Brain tryptamine levels are increased up to 300-fold by MAOIs in animals. In addition, the effects of exogenous tryptamine are strongly augmented by monoamine oxidase inhibitors (MAOIs).
Tryptamine is excreted in urine and its rate of urinary excretion has been reported to be pH-dependent.
Chemistry
Tryptamine is a substituted tryptamine derivative and trace amine and is structurally related to the amino acid tryptophan.
Properties
The experimental log P of tryptamine is 1.55.
Synthesis
The chemical synthesis of tryptamine has been described.
Derivatives
Main article: Substituted tryptamine
The endogenous monoamine neurotransmitters serotonin (5-hydroxytryptamine or 5-HT) and melatonin (5-methoxy-N-acetyltryptamine), as well as trace amines like N-methyltryptamine (NMT), N,N-dimethyltryptamine (DMT), and bufotenin (N,N-dimethylserotonin), are derivatives of tryptamine.
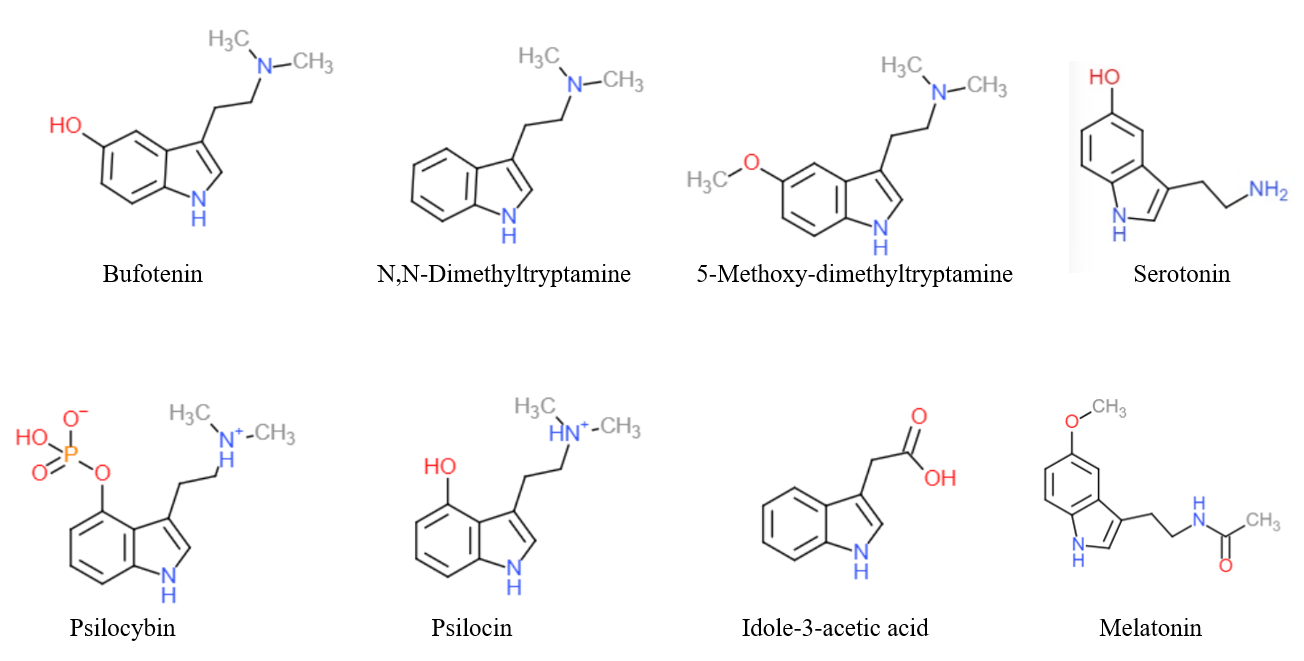
A variety of drugs, including both naturally occurring and pharmaceutical substances, are derivatives of tryptamine. These include the tryptamine psychedelics like psilocybin, psilocin, DMT, and 5-MeO-DMT; tryptamine stimulants, entactogens, psychedelics, and/or antidepressants like α-methyltryptamine (αMT) and α-ethyltryptamine (αET); triptan antimigraine agents like sumatriptan; certain antipsychotics like oxypertine; and the sleep aid melatonin.
Various other drugs, including ergolines and lysergamides like the psychedelic lysergic acid diethylamide (LSD), the antimigraine agents ergotamine, dihydroergotamine, and methysergide, and the antiparkinsonian agents bromocriptine, cabergoline, lisuride, and pergolide; β-carbolines like harmine (some of which are monoamine oxidase inhibitors (MAOIs)); Iboga alkaloids like the hallucinogen ibogaine; yohimbans like the α2 blocker yohimbine; antipsychotics like ciclindole and flucindole; and the MAOI antidepressant metralindole, can all be thought of as cyclized tryptamine derivatives.
Drugs very closely related to tryptamines, but technically not tryptamines themselves, include certain triptans like avitriptan and naratriptan; the antipsychotics sertindole and tepirindole; and the MAOI antidepressants pirlindole and tetrindole.
References
References
- (2005). "CRC Handbook of Chemistry and Physics". [[CRC Press]].
- "Tryptamine".
- (January 2016). "Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis". Nutrients.
- (March 2014). "Psilocybin--summary of knowledge and new perspectives". European Neuropsychopharmacology.
- (January 2015). "Recreational use, analysis and toxicity of tryptamines". Current Neuropharmacology.
- (21 November 2014). "The Ayahuasca Phenomenon".
- (October 2016). "The emerging roles of human trace amines and human trace amine-associated receptors (hTAARs) in central nervous system". Biomedicine & Pharmacotherapy.
- (December 2017). "Pharmacology of human trace amine-associated receptors: Therapeutic opportunities and challenges". Pharmacology & Therapeutics.
- (June 2018). "Gut Microbiota-Produced Tryptamine Activates an Epithelial G-Protein-Coupled Receptor to Increase Colonic Secretion". Cell Host & Microbe.
- (April 2003). "Intestinal ion transport and the pathophysiology of diarrhea". The Journal of Clinical Investigation.
- (2012). "LiverTox: Clinical and Research Information on Drug-Induced Liver Injury". National Institute of Diabetes and Digestive and Kidney Diseases.
- (2020-12-09). "New Compound Related to Psychedelic Ibogaine Could Treat Addiction, Depression".
- "Chemists re-engineer a psychedelic to treat depression and addiction in rodents". AAAS.
- (January 2011). "The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity". Journal of Neurochemistry.
- (2020). "Serotonin Synthesis and Metabolism".
- "MetaCyc L-tryptophan degradation VI (via tryptamine)".
- "PDSP Database".
- "BindingDB BDBM50024210 1H-indole-3-ethanamine::2-(1H-indol-3-yl)ethanamine::2-(3-indolyl)ethylamine::CHEMBL6640::tryptamine".
- (2019). "5-HT2 receptor binding, functional activity and selectivity in N-benzyltryptamines". PLOS ONE.
- (April 2016). "In Vitro Characterization of Psychoactive Substances at Rat, Mouse, and Human Trace Amine-Associated Receptor 1". The Journal of Pharmacology and Experimental Therapeutics.
- (September 1999). "Functional characterization of agonists at recombinant human 5-HT2A, 5-HT2B and 5-HT2C receptors in CHO-K1 cells". British Journal of Pharmacology.
- (1997). "Pharmacochemistry Library". Elsevier.
- (October 2023). "A cane toad (Rhinella marina) N-methyltransferase converts primary indolethylamines to tertiary psychedelic amines". The Journal of Biological Chemistry.
- (10 March 2023). "Bioproduction platform using a novel cane toad (Rhinella marina) N-methyltransferase for psychedelic-inspired drug discovery". Research Square.
- (February 2000). "Agonist high and low affinity state ratios predict drug intrinsic activity and a revised ternary complex mechanism at serotonin 5-HT(2A) and 5-HT(2C) receptors". Synapse.
- (November 1993). "Characterization of the 5-HT4 receptor mediating tachycardia in piglet isolated right atrium". British Journal of Pharmacology.
- (March 1993). "Tryptamine: a metabolite of tryptophan implicated in various neuropsychiatric disorders". Metabolic Brain Disease.
- (April 2012). "Vasoconstrictor and vasodilator responses to tryptamine of rat-isolated perfused mesentery: comparison with tyramine and β-phenylethylamine". British Journal of Pharmacology.
- (April 1985). "Tryptamine-induced vasoconstrictor responses in rat caudal arteries are mediated predominantly via 5-hydroxytryptamine receptors". British Journal of Pharmacology.
- (January 1987). "Central serotonin receptors as targets for drug research". Journal of Medicinal Chemistry.
- (2018-06-15). "Microbiome-Lax May Relieve Constipation".
- (1982). "Tryptamine: a neuromodulator or neurotransmitter in mammalian brain?". Progress in Neurobiology.
- (October 2014). "Alpha-ethyltryptamines as dual dopamine-serotonin releasers". Bioorganic & Medicinal Chemistry Letters.
- (February 2015). "Behavioral, biological, and chemical perspectives on atypical agents targeting the dopamine transporter". Drug and Alcohol Dependence.
- (22 May 2012). "Synthesis and Biological Evaluation of Rigid Analogues of Methamphetamines".
- (July 2008). "Dopamine Transporters: Chemistry, Biology and Pharmacology". Wiley.
- (April 2012). "Studies of the biogenic amine transporters. 14. Identification of low-efficacy "partial" substrates for the biogenic amine transporters". The Journal of Pharmacology and Experimental Therapeutics.
- (March 2007). "The effects of non-medically used psychoactive drugs on monoamine neurotransmission in rat brain". European Journal of Pharmacology.
- (January 2001). "Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin". Synapse.
- (March 2013). "Powerful cocaine-like actions of 3,4-methylenedioxypyrovalerone (MDPV), a principal constituent of psychoactive 'bath salts' products". Neuropsychopharmacology.
- (October 2003). "Monoamine transporters and psychostimulant drugs". European Journal of Pharmacology.
- (2006). "Therapeutic potential of monoamine transporter substrates". Current Topics in Medicinal Chemistry.
- (May 2004). "Pharmacological studies with endogenous enhancer substances: beta-phenylethylamine, tryptamine, and their synthetic derivatives". Progress in Neuro-Psychopharmacology & Biological Psychiatry.
- (August 2003). "Enhancer regulation/endogenous and synthetic enhancer compounds: a neurochemical concept of the innate and acquired drives". Neurochemical Research.
- (August 2022). "Enhancer Regulation of Dopaminergic Neurochemical Transmission in the Striatum". International Journal of Molecular Sciences.
- (August 2023). "Striking Neurochemical and Behavioral Differences in the Mode of Action of Selegiline and Rasagiline". International Journal of Molecular Sciences.
- (2001). "Antiaging compounds: (-)deprenyl (selegeline) and (-)1-(benzofuran-2-yl)-2-propylaminopentane, [(-)BPAP], a selective highly potent enhancer of the impulse propagation mediated release of catecholamine and serotonin in the brain". CNS Drug Reviews.
- (May 2001). "Structure-activity studies leading to (-)1-(benzofuran-2-yl)-2-propylaminopentane, ((-)BPAP), a highly potent, selective enhancer of the impulse propagation mediated release of catecholamines and serotonin in the brain". Bioorganic & Medicinal Chemistry.
- (December 1999). "(-)1-(Benzofuran-2-yl)-2-propylaminopentane, [(-)BPAP], a selective enhancer of the impulse propagation mediated release of catecholamines and serotonin in the brain". British Journal of Pharmacology.
- (July 2018). "Trace Amines and Their Receptors". Pharmacological Reviews.
- (December 2006). "Trace amine-associated receptors and their ligands". British Journal of Pharmacology.
- (February 2016). ""TAARgeting Addiction"--The Alamo Bears Witness to Another Revolution: An Overview of the Plenary Symposium of the 2015 Behavior, Biology and Chemistry Conference". Drug and Alcohol Dependence.
- (1977). "Drug Addiction II". Springer Berlin Heidelberg.
- (1997). "Tihkal: The Continuation". Transform Press.
- (October 2014). "Interaction of psychoactive tryptamines with biogenic amine transporters and serotonin receptor subtypes". Psychopharmacology.
- (1970). "Effects of infused tryptamine in man". Psychopharmacologia.
- (January 1987). "The behavioural effects of intravenously administered tryptamine in mice". Neuropharmacology.
- (November 2013). "Animal models of the serotonin syndrome: a systematic review". Behavioural Brain Research.
- (January 1986). "Effect of tryptamine on the behavior of mice". Journal of Pharmacobio-Dynamics.
- (1988). "Effects of 5,7-dihydroxytryptamine and 6-hydroxydopamine on head-twitch response induced by serotonin, p-chloroamphetamine, and tryptamine in mice". Psychopharmacology.
- (June 1981). "In vivo pharmacological studies on the interactions between tryptamine and 5-hydroxytryptamine". British Journal of Pharmacology.
- (1984). "Neurobiology of the Trace Amines". Humana Press.
- (2012). "Head-twitch response in rodents induced by the hallucinogen 2,5-dimethoxy-4-iodoamphetamine: a comprehensive history, a re-evaluation of mechanisms, and its utility as a model". Drug Testing and Analysis.
- (2013). "The Neuroscience of Hallucinations". Springer New York.
- (1985). "Neuropsychopharmacology of the Trace Amines". Humana Press.
- (1986). "Receptor Binding". Humana Press.
- (June 1965). "Tryptamine, N,N-dimethyltryptamine, N,N-dimethyl-5-hydroxytryptamine and 5-methoxytryptamine in human blood and urine". Nature.
- (December 1975). "The dependence of tryptamine excretion on urinary pH". Clinica Chimica Acta; International Journal of Clinical Chemistry.
- "Tryptamine".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Tryptamine — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report