From Surf Wiki (app.surf) — the open knowledge base
Triazole
Chemical compound
Chemical compound
A triazole is a heterocyclic compound featuring a five-membered ring of two carbon atoms and three nitrogen atoms with molecular formula C2H3N3. Triazoles exhibit substantial isomerism, depending on the positioning of the nitrogen atoms within the ring.
Many triazoles are versatile, biologically active compounds commonly used as fungicides and plant retardants. However, triazoles are also useful in bioorthogonal chemistry, because the large number of nitrogen atoms causes triazoles to react similar to azides. Lastly, the many free lone pairs in triazoles make them useful as coordination compounds, although not typically as haptic ligands.
Isomerism
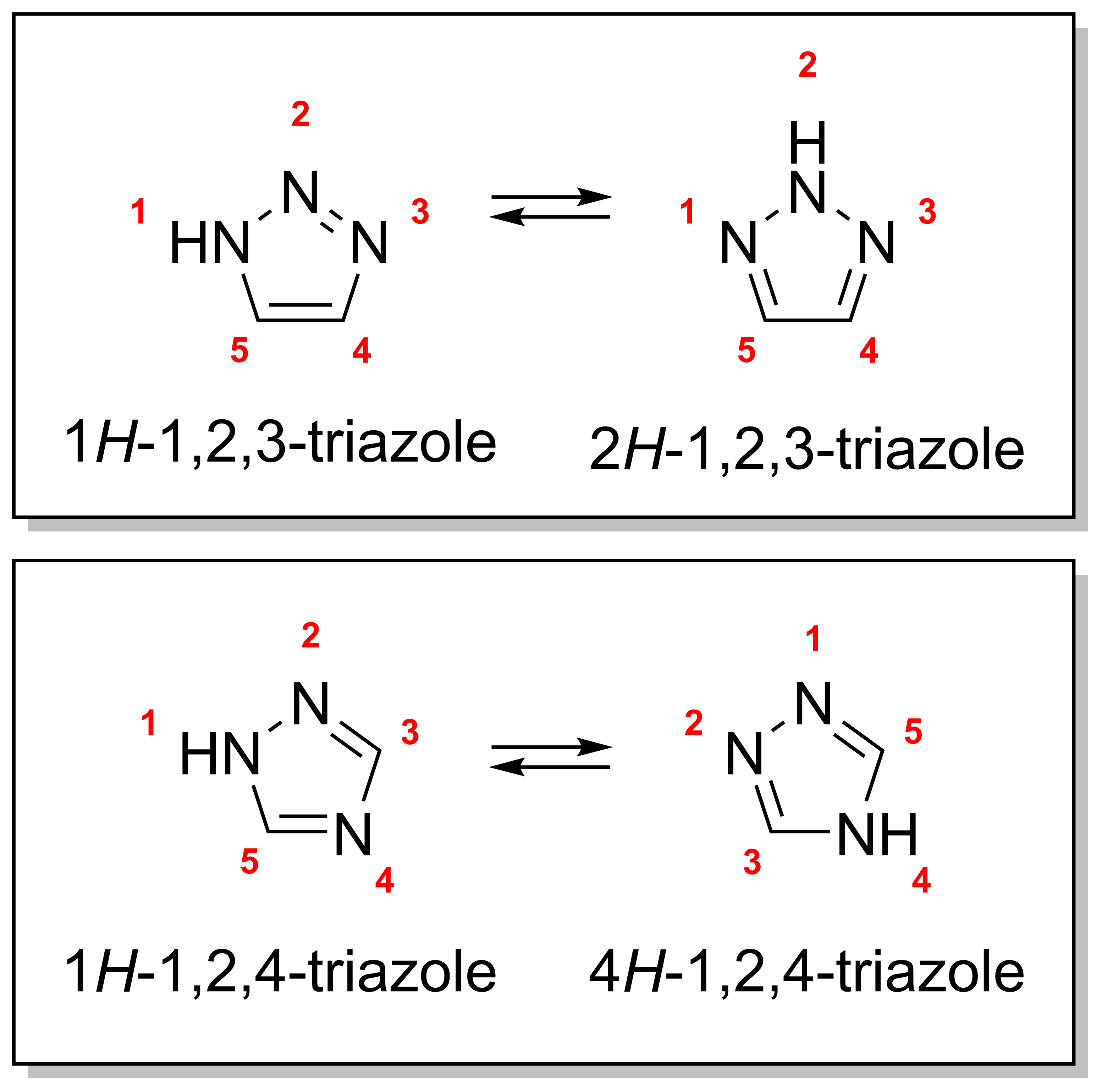
There are four triazole isomers, which are conventionally divided into two pairs of tautomers. In the 1,2,3-triazoles, the three nitrogen atoms are adjacent; in the 1,2,4-triazoles, an interstitial carbon separates out one nitrogen atom. Each category has two tautomers that differ by which nitrogen has a hydrogen bonded to it.

Preparation
There are several methods to prepare triazoles.
1,2,3-Triazoles
Main article: 1,2,3-Triazole
1,2,3-Triazoles, also known as vicinal triazoles, are usually prepared following (3+2) cycloaddition protocols. A common technique for unsubstituted triazoles is the Huisgen azide-alkyne 1,3-dipolar cycloaddition: a azide and an alkyne react at high temperature to form a ring. However, the Huisgen strategy produces a mixture of isomers (typically 1,4- and 1,5-disubstituted) when used to produce substituted triazoles. [[File:Huisgen-azide-alkyne-cycloaddition.png|frameless|center|upright=2|Thermal addition gives a mixture of 1,4 and 1,5 isomers]]
In order to selectively prepare a desired isomer, metal catalysts are employed. In the copper-catalysed azide-alkyne cycloaddition (CuAAC), copper(I) salts select for the formation of 1,4-disubstituted 1,2,3-triazoles. One such catalyst is CuBr(PPh3)3, which is relatively stable towards oxidation even at elevated temperatures and can produce triazoles with a broad range of substituents either in solvent or under neat reaction conditions. [[File:CuAAC-triazole-synthesis.png|frameless|center|upright=2|1,4 isomer from a CuI catalyst]]
Conversely, ruthenium catalysts (RuAAC) select for 1,5-disubstituted 1,2,3-triazoles. [[File:RuAAC-triazole-synthesis.png|frameless|center|upright=2|1,5 isomer from a Ru catalyst]]
1,2,4-Triazoles
Main article: 1,2,4-Triazole
Most techniques for producing 1,2,4-triazoles use the free energy of water, either by dehydrating a mixture of amides and hydrazides (the Pellizzari reaction) or imides and alkyl hydrazines (the Einhorn-Brunner reaction). Of those two, only the Einhorn-Brunner reaction is regioselective. Recent research has focused on grinding and microwave irradiation as greener substitutes.
Applications
Triazoles are compounds with a vast spectrum of applications, varying from materials (polymers), agricultural chemicals, pharmaceuticals, photoactive chemicals and dyes.
Benzotriazole is used in chemical photography as a restrainer and fog suppressant.
Cyclohexylethyltriazol was briefly used as an alternative to Cardiazol (Metrazol) in convulsive shock therapy treatment of mental illnesses during the 1940s.
Importance in agriculture
Many triazoles have antifungal effects: the triazole antifungal drugs include fluconazole, isavuconazole, itraconazole, voriconazole, pramiconazole, ravuconazole, and posaconazole and triazole plant-protection fungicides include epoxiconazole, , myclobutanil, propiconazole, prothioconazole, metconazole, cyproconazole, tebuconazole, flusilazole and paclobutrazol.
Due to spreading resistance of plant pathogens towards fungicides of the strobilurin class, control of fungi such as Septoria tritici or Gibberella zeae relies heavily on triazoles. Food, like store bought potatoes, contain retardants such as triazole or tetcyclacis.
In addition, paclobutrazol, uniconazole, , and triadimefon are used as plant growth retardants. Brassinazole inhibits brassinosteroid biosynthesis.
Importance in chemical synthesis
The azide alkyne Huisgen cycloaddition
References
References
- (2019). "Arylation of Click Triazoles with Diaryliodonium Salts". [[J. Org. Chem.]].
- Virant, Miha. (2019). "Development of homogeneous palladium catalytic systems for selected transformations of terminal acetylenes". [[University of Ljubljana]].
- (2014). "Synthesis and NMR Analysis of 1,4-Disubstituted 1,2,3-Triazoles Tethered to Pyridine, Pyrimidine, and Pyrazine Rings". [[Eur. J. Org. Chem.]].
- Košmrlj, Janez. (2012). "Click Triazoles". Springer.
- (1963). "1,3-Dipolar Cycloadditions, Past and Future". [[Angew. Chem. Int. Ed.]].
- Temple, Carroll. (2009). "1,2,4-Triazoles". Wiley-Blackwell.
- Farooq, Tahir. (2021). "Advances in Triazole Chemistry". [[Satan.
- (1961). "The Chemistry of 1,2,4-Triazoles". [[Chem. Rev.]].
- (2011). "Click Chemistry: 1,2,3-Triazoles as Pharmacophores". [[Chem. Asian J.]].
- (2002). "Mechanisms influencing the evolution of resistance to Qo inhibitor fungicides". [[Pest Manag. Sci.]].
- (2007). "Comparison of the declining triazole sensitivity of ''Gibberella zeae'' and increased sensitivity achieved by advances in triazole fungicide development". [[J. Crop Prot.]].
- (2009). "Control of potato early blight with triazole fungicide using preventive and curative spraying, or a forecasting system". [[Cienc. Inv. Agr.]].
- (1987). "Tetcyclacis and triazole-type plant growth retardants: Their influence on the biosynthesis of gibberellins and other metabolic processes". [[Pestic. Sci.]].
- Latimer, Joyce G.. (2022). "Growth Regulators for Containerized Herbaceous Perennial Plants". Ball Publishing.
- (2008). "Triazole-Linked Analogue of Deoxyribonucleic Acid (TLDNA): Design, Synthesis, and Double-Strand Formation with Natural DNA". [[Org. Lett.]].
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Triazole — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report