From Surf Wiki (app.surf) — the open knowledge base
Succinaldehyde
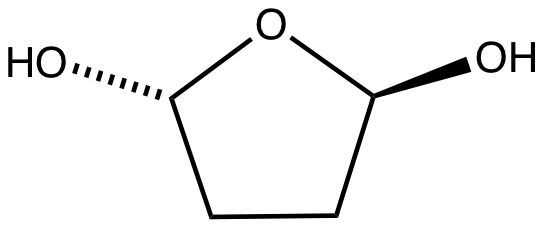
Succinaldehyde or Butanedial is an organic compound with the formula . It is a colorless viscous liquid. Succinaldehyde can be used as a crosslinking agent for proteins, but it is less widely used than the related dialdehyde glutaraldehyde.
Preparation and reactions

Succinaldehyde is generated by the oxidation of tetrahydrofuran with chlorine followed by hydrolysis of the chlorinated product. It can also be prepared by the hydroformylation of acrolein or the acetals thereof. Oxidation of 2,5-dimethoxytetrahydrofuran with hydrogen peroxide is yet another route to succinaldehyde.
In the presence of water, succinaldehyde converts to the cyclic hydrate. In methanol it converts to the cyclic acetal, 2,5-dimethoxyltetrahydrofuran.
References
References
- [[International Union of Pure and Applied Chemistry]]. (2014). "Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013". [[Royal Society of Chemistry.
- Typical of some other saturated [[dialdehyde]]s, succinaldehyde is handled as the hydrates or methanol-derived [[acetal]]. It is a precursor to [[tropinone]].{{US patent. 2710883
- (2022). "Organocatalytic Dimerization of Succinaldehyde". Organic Syntheses.
- (1972). "The Hydration and Polymerisation of Succinaldehyde, Glutaraldehyde, and Adipaldehyde". Journal of the Chemical Society, Perkin Transactions 2.
- Christian Kohlpaintner. (2008). "Aldehydes, Aliphatic".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Succinaldehyde — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report