From Surf Wiki (app.surf) — the open knowledge base
Selenol
Class of chemical compounds
Class of chemical compounds

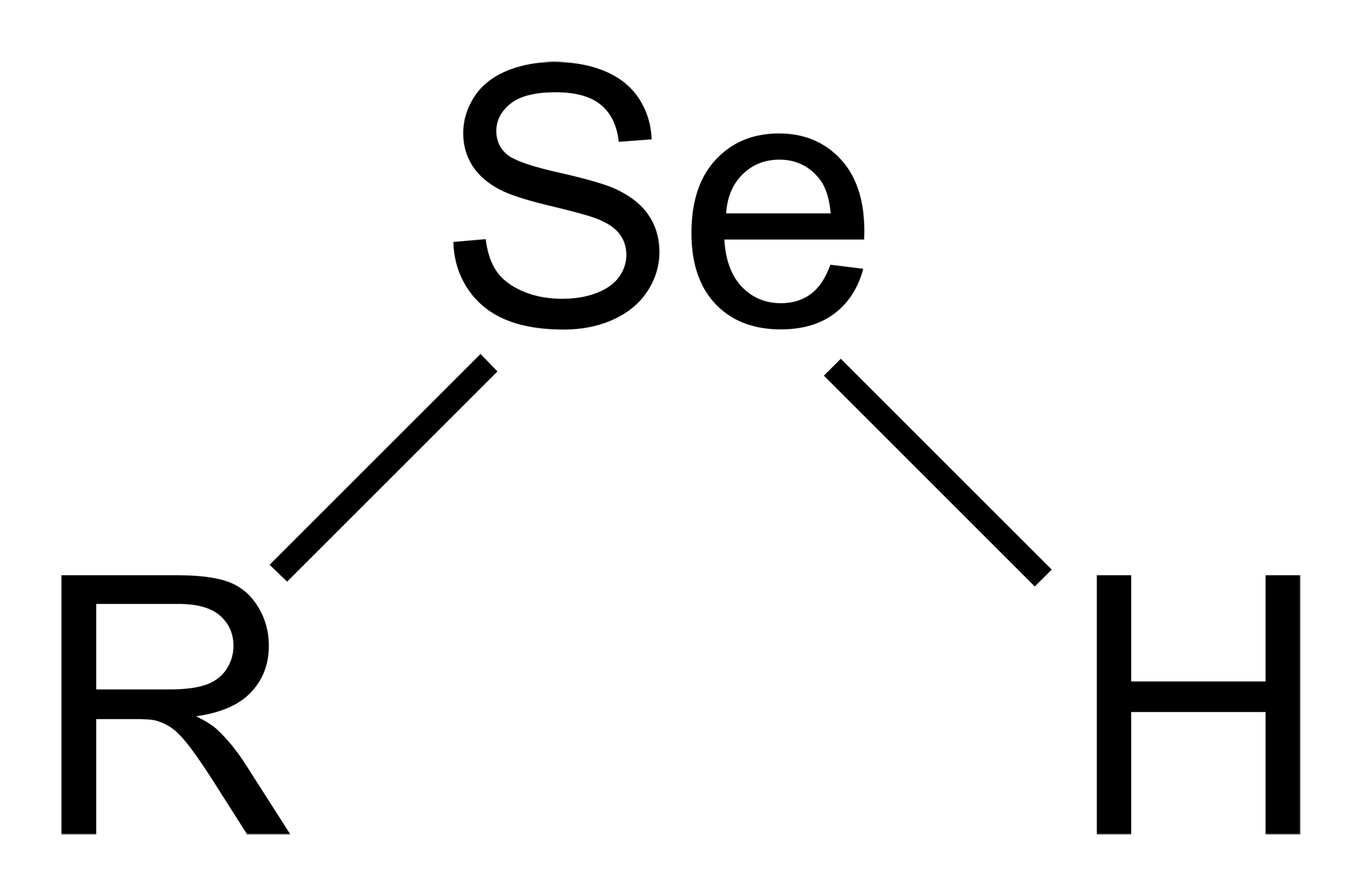
Selenols are organic compounds that contain the functional group with the connectivity . Selenols are sometimes also called selenomercaptans and selenothiols. Selenols are one of the principal classes of organoselenium compounds. A well-known selenol is the amino acid selenocysteine.
Structure and properties
Selenols are structurally similar to thiols, but the bond is about 8% longer at 196 pm. The angle approaches 90°. The bonding involves almost pure p-orbitals on Se, hence the near 90 angles. The bond energy is weaker than the bond, consequently selenols are easily oxidized and serve as H-atom donors. The Se-H bond is weaker than the bond as reflected in their respective bond dissociation energy (BDE). For , the BDE is 326 kJ/mol, while for , the BDE is 368 kJ/mol.
Selenols are about 1000 times stronger acids than thiols: the pKa of is 5.2 vs 8.3 for . Deprotonation affords the selenolate anion, , most examples of which are highly nucleophilic and rapidly oxidized by air.
The boiling points of selenols tend to be slightly greater than for thiols. This difference can be attributed to the increased importance of stronger van der Waals bonding for larger atoms. Volatile selenols have highly offensive odors.
Applications and occurrence
Main article: Organoselenium chemistry
Selenols have few commercial applications, being limited by the toxicity of selenium as well as the sensitivity of the bond. Their conjugate bases, the selenolates, also have limited applications in organic synthesis.
Biochemical role
Selenols are important in certain biological processes. Three enzymes found in mammals contain selenols at their active sites: glutathione peroxidase, iodothyronine deiodinase, and thioredoxin reductase. The selenols in these proteins are part of the essential amino acid selenocysteine. Precursors of methaneselenol are under active investigation in cancer prevention and therapy. In these studies, methaneselenol is found to be more biologically active than ethaneselenol () or 2-propaneselenol ().
Preparation
Selenols are usually prepared by the reaction of organolithium reagents or Grignard reagents with elemental Se. For example, benzeneselenol is generated by the reaction of phenylmagnesium bromide with selenium followed by acidification: :[[File:Selenophenol.png|360px]] Another preparative route to selenols involves the alkylation of selenourea, followed by hydrolysis.
Selenols are often generated by reduction of diselenides followed by protonation of the resulting selenolate: : :
Dimethyl diselenide can be easily reduced to methaneselenol within cells.
Reactions
Selenols are easily oxidized to diselenides, compounds containing an bond. For example, treatment of benzeneselenol with bromine gives diphenyl diselenide. : In the presence of base, selenols are readily alkylated to give selenides. This relationship is illustrated by the methylation of methaneselenol to give dimethylselenide.
Safety
Organoselenium compounds (or any selenium compound) are cumulative poisons despite the fact that trace amounts of Se are required for health.
References
References
- (2021). "Synthesis and Applications of Organic Selenols". Advanced Synthesis & Catalysis.
- {{RubberBible87th
- (2007). "Selenium in Chemistry and Biochemistry in Comparison to Sulfur". Biological Chemistry.
- (2012). "Methylselenol, a Selenium Metabolite, Plays Common and Different Roles in Cancerous Colon HCT116 Cell and Noncancerous NCM460 Colon Cell Proliferation". Nutrition and Cancer.
- (2012). "Methylselenol Formed by Spontaneous Methylation of Selenide Is a Superior Selenium Substrate to the Thioredoxin and Glutaredoxin Systems". PLOS ONE.
- (2002). "New Concepts in Selenium Chemoprevention". Cancer and Metastasis Reviews.
- (2012). "Cytotoxic and Proapototic Activities of Imidoselenocarbamate Derivatives Are Dependent on the Release of Methylselenol". [[Chemical Research in Toxicology]].
- Reich, Hans J.. (1979). "Functional group manipulation using organoselenium reagents". American Chemical Society.
- (1944). "Selenophenol". [[Organic Syntheses]].
- (2010). "Formation of methylselenol, dimethylselenide and dimethyldiselenide in in vitro metabolism models determined by headspace GC-MS". [[Metallomics (journal).
- (2012). "Selenium and human health". The Lancet.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Selenol — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report