From Surf Wiki (app.surf) — the open knowledge base
Potassium peroxide
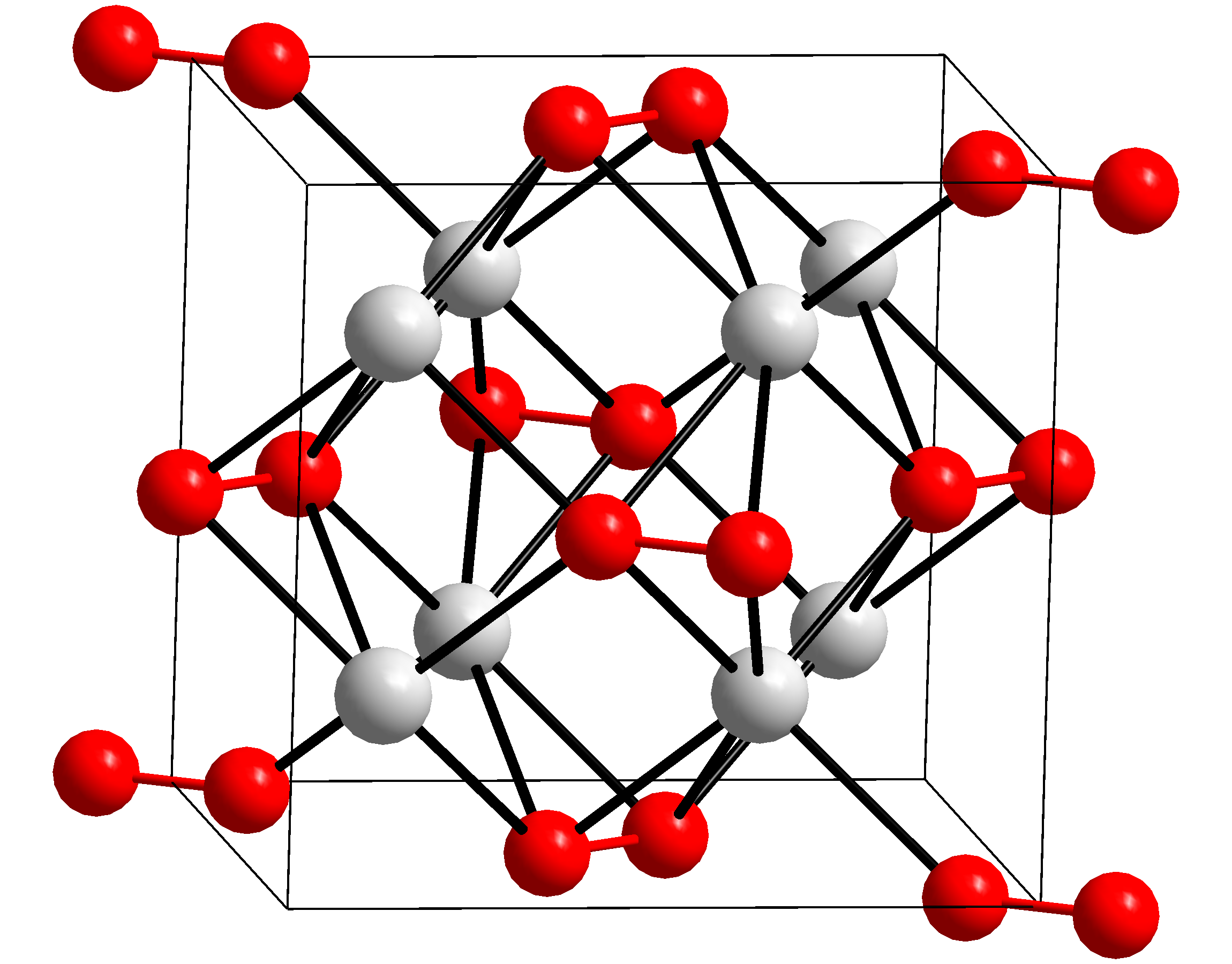
| NFPA-H = 3 | NFPA-F = 0 | NFPA-R = 2 | NFPA-S = W+OX Sodium peroxide Rubidium peroxide Caesium peroxide Potassium superoxide Potassium ozonide
Potassium peroxide is an inorganic compound with the molecular formula K2O2. It is formed as potassium reacts with oxygen in the air, along with potassium oxide (K2O) and potassium superoxide (KO2).

Potassium peroxide reacts with water to form potassium hydroxide and oxygen:
:
Properties
Potassium peroxide is a highly reactive, oxidizing white to yellowish solid which, while not flammable itself, reacts violently with flammable materials. It decomposes violently on contact with water.
The standard enthalpy of formation of potassium peroxide is ΔH f 0 = −496 kJ/mol.
Usage
Potassium peroxide is used as an oxidizing agent and bleach (due to the peroxide), and to purify air.
References
References
- Zumdahl, Steven S.. (2009). "Chemical Principles 6th Ed.". Houghton Mifflin Company.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Potassium peroxide — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report