From Surf Wiki (app.surf) — the open knowledge base
Nitrogen trifluoride
Chemical compound
Chemical compound
Trifluoramine Trifluorammonia 1.885 g/cm3 (liquid at b.p.) −109 kJ/mol | NFPA-H = 1 | NFPA-F = 0 | NFPA-R = 0 | NFPA-S = OX 9600 ppm (dog, 1 h) 7500 ppm (monkey, 1 h) 6700 ppm (rat, 1 h) 7500 ppm (mouse, 1 h) nitrogen tribromide nitrogen triiodide ammonia arsenic trifluoride antimony trifluoride bismuth trifluoride
Nitrogen trifluoride is the inorganic compound with the formula (). It is a colorless, non-flammable, toxic gas with a slightly musty odor. In contrast with ammonia, it is nonbasic. It finds increasing use within the manufacturing of flat-panel displays, photovoltaics, LEDs and other microelectronics. |access-date = 2008-09-07 |url-status = live |archive-url = https://web.archive.org/web/20180123073939/http://www.nytimes.com/2008/09/02/science/02nitr.html?ref=science |archive-date = 2018-01-23
Synthesis and reactivity
Nitrogen trifluoride can be prepared from the elements in the presence of an electric discharge. In 1903, Otto Ruff prepared nitrogen trifluoride by the electrolysis of a molten mixture of ammonium fluoride and hydrogen fluoride.{{cite journal
is slightly soluble in water without undergoing chemical reaction. It is nonbasic with a low dipole moment of 0.2340 D. By contrast, ammonia is basic and highly polar (1.47 D). This contrast reflects the differing electronegativities of H vs F.
Similar to dioxygen, NF3 is a potent yet sluggish oxidizer. It oxidizes hydrogen chloride to chlorine: :2 NF3 + 6 HCl → 6 HF + N2 + 3 Cl2 However, it only attacks (explosively) organic compounds at high temperatures. Consequently it is compatible under standard conditions with several plastics, as well as steel and Monel.
Above 200-300 °C, NF3 reacts with metals, carbon, and other reagents to give tetrafluorohydrazine: :
NF3 reacts with fluorine and antimony pentafluoride to give the tetrafluoroammonium salt: : NF3 + F2 + SbF5 → NFSbF
NF3 and B2H6 react vigorously even at cryogenic temperatures to give nitrogen gas, boron trifluoride, and hydrofluoric acid.
Applications
High-volume applications such as DRAM computer memory production, the manufacturing of flat panel displays and the large-scale production of thin-film solar cells use .
Etching
Main article: Etching (microfabrication)
Nitrogen trifluoride is primarily used to remove silicon and silicon-compounds during the manufacturing of semiconductor devices such as LCD displays, some thin-film solar cells, and other microelectronics. In these applications is initially broken down within a plasma. The resulting fluorine radicals are the active agents that attack polysilicon, silicon nitride and silicon oxide. They can be used as well to remove tungsten silicide, tungsten, and certain other metals. In addition to serving as an etchant in device fabrication, is also widely used to clean PECVD chambers.
dissociates more readily within a low-pressure discharge in comparison to perfluorinated compounds (PFCs) and sulfur hexafluoride (). The greater abundance of negatively-charged free radicals thus generated can yield higher silicon removal rates, and provide other process benefits such as less residual contamination and a lower net charge stress on the device being fabricated. As a somewhat more thoroughly consumed etching and cleaning agent, NF3 has also been promoted as an environmentally preferable substitute for or PFCs such as hexafluoroethane.{{cite journal
The utilization efficiency of the chemicals applied in plasma processes varies widely between equipment and applications. A sizeable fraction of the reactants are wasted into the exhaust stream and can ultimately be emitted into Earth's atmosphere. Modern abatement systems can substantially decrease atmospheric emissions. has not been subject to significant use restrictions. The annual reporting of production, consumption, and waste emissions by large manufacturers has been required in many industrialized countries as a response to the observed atmospheric growth and the international Kyoto Protocol.
Highly toxic fluorine gas (F2, diatomic fluorine) is a climate neutral replacement for nitrogen trifluoride in some manufacturing applications. It requires more stringent handling and safety precautions, especially to protect manufacturing personnel.{{cite journal
Nitrogen trifluoride is also used in hydrogen fluoride and deuterium fluoride lasers, which are types of chemical lasers. There it is also preferred to fluorine gas due to its more convenient handling properties
Greenhouse gas

The GWP of is second only to in the group of Kyoto-recognised greenhouse gases, and was included in that grouping with effect from 2013 and the commencement of the second commitment period of the Kyoto Protocol. It has an estimated atmospheric lifetime of 740 years, although other work suggests a slightly shorter lifetime of 550 years (and a corresponding GWP of 16,800).

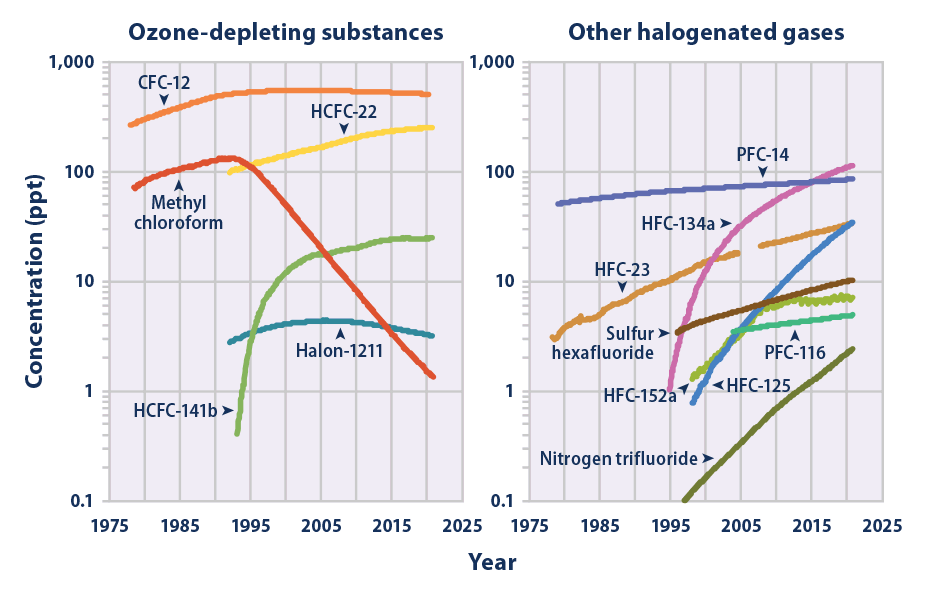
Since 1992, when less than 100 tons were produced, production grew to an estimated 4000 tons in 2007 and is projected to increase significantly. World production of NF3 is expected to reach 8000 tons a year by 2010. By far the world's largest producer of is the US industrial gas and chemical company Air Products & Chemicals. An estimated 2% of produced is released into the atmosphere. Robson projected that the maximum atmospheric concentration is less than 0.16 parts per trillion (ppt) by volume, which will provide less than 0.001 Wm−2 of IR forcing. The mean global tropospheric concentration of NF3 has risen from about 0.02 ppt (parts per trillion, dry air mole fraction) in 1980, to 0.86 ppt in 2011, with a rate of increase of 0.095 ppt yr−1, or about 11% per year, and an interhemispheric gradient that is consistent with emissions occurring overwhelmingly in the Northern Hemisphere, as expected. This rise rate in 2011 corresponds to about 1200 metric tons/y NF3 emissions globally, or about 10% of the NF3 global production estimates. This is a significantly higher percentage than has been estimated by industry, and thus strengthens the case for inventorying NF3 production and for regulating its emissions. One study co-authored by industry representatives suggests that the contribution of the NF3 emissions to the overall greenhouse gas budget of thin-film Si-solar cell manufacturing is clear.
The UNFCCC, within the context of the Kyoto Protocol, decided to include nitrogen trifluoride in the second Kyoto Protocol compliance period, which begins in 2012 and ends in either 2017 or 2020. Following suit, the WBCSD/WRI GHG Protocol is amending all of its standards (corporate, product and Scope 3) to also cover NF3.
Safety
Skin contact with is not hazardous, and it is a relatively minor irritant to mucous membranes and eyes. It is a pulmonary irritant with a toxicity considerably lower than nitrogen oxides, and overexposure via inhalation causes the conversion of hemoglobin in blood to methemoglobin, which can lead to the condition methemoglobinemia. The National Institute for Occupational Safety and Health (NIOSH) specifies that the concentration that is immediately dangerous to life or health (IDLH value) is 1,000 ppm.
Notes
References
References
- Air Products; [http://www.airproducts.com/products/Gases/gas-facts/physical-properties/physical-properties-nitrogen-trifluoride.aspx Physical Properties for Nitrogen Trifluoride]
- (1967). "The enthalpy of dissociation of nitrogen trifluoride". J. Phys. Chem..
- "Inorganic Chemistry".
- {{PGCH. 0455
- {{IDLH. 7783542. Nitrogen trifluoride
- "Climate Change 2007: The Physical Sciences Basis". [[Intergovernmental Panel on Climate Change.
- Robson, J. I.. (2006). "Revised IR spectrum, radiative efficiency and global warming potential of nitrogen trifluoride". [[Geophysical Research Letters.
- (1995)
- Philip B. Henderson, Andrew J. Woytek "Fluorine Compounds, Inorganic, Nitrogen" in Kirk‑Othmer ''Encyclopedia of Chemical Technology'', 1994, John Wiley & Sons, NY. {{doi. 10.1002/0471238961.1409201808051404.a01 Article Online Posting Date: December 4, 2000
- (2006). "Nitrogen–fluorine compounds". Journal of Fluorine Chemistry.
- (1967). "Derivatives of Nitrogen Fluorides". Chemical Reviews.
- Parry, Robert W., and Thomas C. Bissot. "The Preparation and Properties of Phosphorus Trifluoride-Borane and Phosphorus Trifluoride-Borane-d31." Journal of the American Chemical Society 78, no. 8 (1956): 1524-1527.
- Tsai, W.-T.. (2008). "Environmental and health risk analysis of nitrogen trifluoride ({{chem". J. Hazard. Mater..
- (2016-09-30). "F-GHG Emissions Reduction Efforts: Flat Panel Display Supplier Profiles". U.S. EPA.
- (27 September 2015). "Fluorinated Greenhouse Gas Emissions and Supplies Reported to the Greenhouse Gas Reporting Program (GHGRP)". U.S. Environmental Protection Agency.
- (27 June 2016). "Climate Change Indicators: Atmospheric Concentrations of Greenhouse Gases - Figure 4". U.S. Environmental Protection Agency.
- (2020-06-30). "Atmospheric Flask NF3". National Oceanic and Atmospheric Administration.
- Prather, M.J.. (2008). "{{chem". [[Geophysical Research Letters.
- M. Roosevelt. (2008-07-08). "A climate threat from flat TVs, microchips". Los Angeles Times.
- Hoag, Hannah. (2008-07-10). "The Missing Greenhouse Gas". [[Nature (journal).
- Robson, Jon. "Nitrogen trifluoride (NF3)". [[Royal Meteorological Society]].
- (2013-02-05). "Nitrogen trifluoride global emissions estimated from updated atmospheric measurements". [[Proc. Natl. Acad. Sci. USA]].
- (2010-10-25). "Life-Cycle Nitrogen Trifluoride Emissions from Photovoltaics". [[American Chemical Society]].
- Rivers, Ali. (2012-08-15). "Nitrogen trifluoride: the new mandatory Kyoto Protocol greenhouse gas". www.ecometrica.com.
- Malik, Yogender. (2008-07-03). "Nitrogen trifluoride – Cleaning up in electronic applications". [[Gasworld]].
- (2 November 2018). "Immediately Dangerous to Life or Health Concentrations (IDLH): Nitrogen Trifluoride". [[National Institute for Occupational Safety and Health]].
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Nitrogen trifluoride — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report