From Surf Wiki (app.surf) — the open knowledge base
MLH1
Protein-coding gene in humans
Protein-coding gene in humans
DNA mismatch repair protein Mlh1 or MutL protein homolog 1 is a protein that in humans is encoded by the MLH1 gene located on chromosome 3. The gene is commonly associated with hereditary nonpolyposis colorectal cancer. Orthologs of human MLH1 have also been studied in other organisms including mouse and the budding yeast Saccharomyces cerevisiae.
Function
Variants in this gene can cause hereditary nonpolyposis colon cancer (Lynch syndrome). It is a human homolog of the E. coli DNA mismatch repair gene, mutL, which mediates protein-protein interactions during mismatch recognition, strand discrimination, and strand removal. Defects in MLH1 are associated with the microsatellite instability observed in hereditary nonpolyposis colon cancer. Alternatively spliced transcript variants encoding different isoforms have been described, but their full-length natures have not been determined.
Role in DNA mismatch repair
MLH1 protein is one component of a system of seven DNA mismatch repair proteins that work coordinately in sequential steps to initiate repair of DNA mismatches in humans. Defects in mismatch repair, found in about 13% of colorectal cancers, are much more frequently due to deficiency of MLH1 than deficiencies of other DNA mismatch repair proteins. The seven DNA mismatch repair proteins in humans are MLH1, MLH3, MSH2, MSH3, MSH6, PMS1 and PMS2.
DNA mismatches occur where one base is improperly paired with another base, or where there is a short addition or deletion in one strand of DNA that is not matched in the other strand. Mismatches commonly occur as a result of DNA replication errors or during genetic recombination. Recognizing those mismatches and repairing them is important for cells because failure to do so results in microsatellite instability] and an elevated spontaneous mutation rate (mutator phenotype). Among 20 cancers evaluated, microsatellite instable colon cancer (mismatch repair deficient) had the second highest frequency of mutations (after melanoma).
A heterodimer between MSH2 and MSH6 first recognizes the mismatch, although a heterodimer between MSH2 and MSH3 also can start the process. The formation of the MSH2-MSH6 heterodimer accommodates a second heterodimer of MLH1 and PMS2, although a heterodimer between MLH1 and either PMS3 or MLH3 can substitute for PMS2. This protein complex formed between the 2 sets of heterodimers enables initiation of repair of the mismatch defect.
Other gene products involved in mismatch repair (subsequent to initiation by DNA mismatch repair genes) include DNA polymerase delta, PCNA, RPA, HMGB1, RFC and DNA ligase I, plus histone and chromatin modifying factors.
Deficient expression in cancer
| Cancer type | Frequency of deficiency in cancer | Frequency of deficiency in adjacent field defect | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Stomach | vauthors=Kupčinskaitė-Noreikienė R, Skiecevičienė J, Jonaitis L, Ugenskienė R, Kupčinskas J, Markelis R, Baltrėnas V, Sakavičius L, Semakina I, Grižas S, Juozaitytė E | title=CpG island methylation of the MLH1, MGMT, DAPK, and CASP8 genes in cancerous and adjacent noncancerous stomach tissues | journal=Medicina (Kaunas) | volume=49 | issue=8 | pages=361–6 | year=2013 | pmid=24509146 }} | 24–28% | ||
| Stomach (foveolar type tumors) | vauthors=Endoh Y, Tamura G, Ajioka Y, Watanabe H, Motoyama T | title=Frequent hypermethylation of the hMLH1 gene promoter in differentiated-type tumors of the stomach with the gastric foveolar phenotype | journal=Am. J. Pathol. | volume=157 | issue=3 | pages=717–22 | year=2000 | pmid=10980110 | pmc=1949419 | doi=10.1016/S0002-9440(10)64584-1 }} | 71% |
| Stomach in high-incidence Kashmir Valley | vauthors=Wani M, Afroze D, Makhdoomi M, Hamid I, Wani B, Bhat G, Wani R, Wani K | title=Promoter methylation status of DNA repair gene (hMLH1) in gastric carcinoma patients of the Kashmir valley | journal=Asian Pac. J. Cancer Prev. | volume=13 | issue=8 | pages=4177–81 | year=2012 | pmid=23098428 | doi= 10.7314/apjcp.2012.13.8.4177 | doi-access=free }} | 20% |
| Esophageal | vauthors=Chang Z, Zhang W, Chang Z, Song M, Qin Y, Chang F, Guo H, Wei Q | title=Expression characteristics of FHIT, p53, BRCA2 and MLH1 in families with a history of oesophageal cancer in a region with a high incidence of oesophageal cancer | journal=Oncol Lett | volume=9 | issue=1 | pages=430–436 | year=2015 | pmid=25436004 | pmc=4246613 | doi=10.3892/ol.2014.2682 }} | 27% |
| Head and neck squamous cell carcinoma (HNSCC) | vauthors=Tawfik HM, El-Maqsoud NM, Hak BH, El-Sherbiny YM | title=Head and neck squamous cell carcinoma: mismatch repair immunohistochemistry and promoter hypermethylation of hMLH1 gene | journal=Am J Otolaryngol | volume=32 | issue=6 | pages=528–36 | year=2011 | pmid=21353335 | doi=10.1016/j.amjoto.2010.11.005 }} | 20–25% | |
| Non-small cell lung cancer (NSCLC) | vauthors=Safar AM, Spencer H, Su X, Coffey M, Cooney CA, Ratnasinghe LD, Hutchins LF, Fan CY | title=Methylation profiling of archived non-small cell lung cancer: a promising prognostic system | journal=Clin. Cancer Res. | volume=11 | issue=12 | pages=4400–5 | year=2005 | pmid=15958624 | doi=10.1158/1078-0432.CCR-04-2378 | doi-access=free }} | 72% |
| Colorectal | 10% |
Epigenetic repression
Only a minority of sporadic cancers with a DNA repair deficiency have a mutation in a DNA repair gene. However, a majority of sporadic cancers with a DNA repair deficiency do have one or more epigenetic alterations that reduce or silence DNA repair gene expression. In the table above, the majority of deficiencies of MLH1 were due to methylation of the promoter region of the MLH1 gene. Another epigenetic mechanism reducing MLH1 expression is over-expression of miR-155. MiR-155 targets MLH1 and MSH2 and an inverse correlation between the expression of miR-155 and the expression of MLH1 or MSH2 proteins was found in human colorectal cancer.
Deficiency in field defects
A field defect is an area or "field" of epithelium that has been preconditioned by epigenetic changes and/or mutations so as to predispose it towards development of cancer. As pointed out by Rubin, "The vast majority of studies in cancer research has been done on well-defined tumors in vivo, or on discrete neoplastic foci in vitro. Yet there is evidence that more than 80% of the somatic mutations found in mutator phenotype human colorectal tumors occur before the onset of terminal clonal expansion." Similarly, Vogelstein et al. point out that more than half of somatic mutations identified in tumors occurred in a pre-neoplastic phase (in a field defect), during growth of apparently normal cells.
In the Table above, MLH1 deficiencies were noted in the field defects (histologically normal tissues) surrounding most of the cancers. If MLH1 is epigenetically reduced or silenced, it would not likely confer a selective advantage upon a stem cell. However, reduced or absent expression of MLH1 would cause increased rates of mutation, and one or more of the mutated genes may provide the cell with a selective advantage. The expression-deficient MLH1 gene could then be carried along as a selectively neutral or only slightly deleterious passenger (hitch-hiker) gene when the mutated stem cell generates an expanded clone. The continued presence of a clone with an epigenetically repressed MLH1 would continue to generate further mutations, some of which could produce a tumor.
Repression in coordination with other DNA repair genes
In a cancer, multiple DNA repair genes are often found to be simultaneously repressed. conducted a study where they evaluated the mRNA expression of 27 DNA repair genes in 40 astrocytomas compared to normal brain tissues from non-astrocytoma individuals. Among the 27 DNA repair genes evaluated, 13 DNA repair genes, MLH1, MLH3, MGMT, NTHL1, OGG1, SMUG1, ERCC1, ERCC2, ERCC3, ERCC4, RAD50, XRCC4 and XRCC5 were all significantly down-regulated in all three grades (II, III and IV) of astrocytomas. The repression of these 13 genes in lower grade as well as in higher grade astrocytomas suggested that they may be important in early as well as in later stages of astrocytoma. In another example, Kitajima et al. found that immunoreactivity for MLH1 and MGMT expression was closely correlated in 135 specimens of gastric cancer and loss of MLH1 and MGMTappeared to be synchronously accelerated during tumor progression.
Deficient expression of multiple DNA repair genes are often found in cancers, and may contribute to the thousands of mutations usually found in cancers (see Mutation frequencies in cancers).
Meiosis
In addition to its role in DNA mismatch repair, MLH1 protein is also involved in meiotic crossing over. MLH1 forms a heterodimer with MLH3 that appears to be necessary for oocytes to progress through metaphase II of meiosis. Female and male MLH1(-/-) mutant mice are infertile, and sterility is associated with a reduced level of chiasmata. During spermatogenesis in MLH1(-/-) mutant mice chromosomes often separate prematurely and there is frequent arrest in the first division of meiosis.
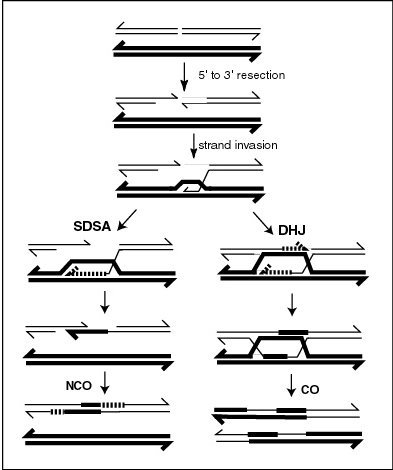
MLH1 protein appears to localize to sites of crossing over in meiotic chromosomes. Recombination during meiosis is often initiated by a DNA double-strand break (DSB) as illustrated in the accompanying diagram. During recombination, sections of DNA at the 5' ends of the break are cut away in a process called resection. In the strand invasion step that follows, an overhanging 3' end of the broken DNA molecule then "invades" the DNA of an homologous chromosome that is not broken forming a displacement loop (D-loop). After strand invasion, the further sequence of events may follow either of two main pathways leading to a crossover (CO) or a non-crossover (NCO) recombinant (see Genetic recombination). The pathway leading to a CO involves a double Holliday junction (DHJ) intermediate. Holliday junctions need to be resolved for CO recombination to be completed.
In the budding yeast Saccharomyces cerevisiae, as in the mouse, MLH1 forms a heterodimer with MLH3. Meiotic CO requires resolution of Holliday junctions through actions of the MLH1-MLH3 heterodimer. The MLH1-MLH3 heterodimer is an endonuclease that makes single-strand breaks in supercoiled double-stranded DNA. MLH1-MLH3 binds specifically to Holliday junctions and may act as part of a larger complex to process Holliday junctions during meiosis.
Clinical significance
It can also be associated with Turcot syndrome.
Interactions
MLH1 has been shown to interact with:
- Bloom syndrome protein
- Exonuclease 1,
- MBD4,
- MSH4,
- Myc, and
- PMS2.
References
References
- "Entrez Gene: MLH1 mutL homolog 1, colon cancer, nonpolyposis type 2 (E. coli)".
- (2008). "A review of the clinical relevance of mismatch-repair deficiency in ovarian cancer". Cancer.
- (2005). "Immunohistochemical analysis reveals high frequency of PMS2 defects in colorectal cancer". Gastroenterology.
- (2015). "Exonuclease 1-dependent and independent mismatch repair". DNA Repair (Amst.).
- (2008). "Mechanisms and functions of DNA mismatch repair". Cell Res..
- (2014). "New insights and challenges in mismatch repair: getting over the chromatin hurdle". DNA Repair (Amst.).
- (2013). "CpG island methylation of the MLH1, MGMT, DAPK, and CASP8 genes in cancerous and adjacent noncancerous stomach tissues". Medicina (Kaunas).
- (2002). "Promoter methylation status of E-cadherin, hMLH1, and p16 genes in nonneoplastic gastric epithelia". Am. J. Pathol..
- (2000). "Frequent hypermethylation of the hMLH1 gene promoter in differentiated-type tumors of the stomach with the gastric foveolar phenotype". Am. J. Pathol..
- (2012). "Promoter methylation status of DNA repair gene (hMLH1) in gastric carcinoma patients of the Kashmir valley". Asian Pac. J. Cancer Prev..
- (2015). "Expression characteristics of FHIT, p53, BRCA2 and MLH1 in families with a history of oesophageal cancer in a region with a high incidence of oesophageal cancer". Oncol Lett.
- (2011). "Head and neck squamous cell carcinoma: mismatch repair immunohistochemistry and promoter hypermethylation of hMLH1 gene". Am J Otolaryngol.
- (2009). "Increased microsatellite instability and epigenetic inactivation of the hMLH1 gene in head and neck squamous cell carcinoma". Otolaryngol Head Neck Surg.
- (2005). "Methylation profiling of archived non-small cell lung cancer: a promising prognostic system". Clin. Cancer Res..
- (2015). "Epigenetic reduction of DNA repair in progression to gastrointestinal cancer". World J Gastrointest Oncol.
- (2010). "Modulation of mismatch repair and genomic stability by miR-155". Proc. Natl. Acad. Sci. U.S.A..
- Rubin H. (March 2011). "Fields and field cancerization: the preneoplastic origins of cancer: asymptomatic hyperplastic fields are precursors of neoplasia, and their progression to tumors can be tracked by saturation density in culture". BioEssays.
- (February 2000). "Genetic reconstruction of individual colorectal tumor histories". Proc. Natl. Acad. Sci. U.S.A..
- (March 2013). "Cancer genome landscapes". Science.
- (2006). "Expression analyses of 27 DNA repair genes in astrocytoma by TaqMan low-density array". Neurosci. Lett..
- (2003). "Loss of expression of DNA repair enzymes MGMT, hMLH1, and hMSH2 during tumor progression in gastric cancer". Gastric Cancer.
- (1996). "Involvement of mouse Mlh1 in DNA mismatch repair and meiotic crossing over". Nat. Genet..
- (2008). "Comparative analysis of meiotic progression in female mice bearing mutations in genes of the DNA mismatch repair pathway". Biol. Reprod..
- (2002). "Mouse models for human DNA mismatch-repair gene defects". Trends Mol Med.
- (2014). "The Saccharomyces cerevisiae Mlh1-Mlh3 heterodimer is an endonuclease that preferentially binds to Holliday junctions". J. Biol. Chem..
- (2014). "Mlh1-Mlh3, a meiotic crossover and DNA mismatch repair factor, is a Msh2-Msh3-stimulated endonuclease". J. Biol. Chem..
- (2012). "Delineation of joint molecule resolution pathways in meiosis identifies a crossover-specific resolvase". Cell.
- (2007). "Turcot syndrome confirmed with molecular analysis". Eur. J. Neurol..
- (April 2000). "BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures". Genes Dev..
- (August 2001). "The Bloom's syndrome protein (BLM) interacts with MLH1 but is not required for DNA mismatch repair". J. Biol. Chem..
- (August 2001). "Cleavage of the Bloom's syndrome gene product during apoptosis by caspase-3 results in an impaired interaction with topoisomerase IIIalpha". Nucleic Acids Res..
- (November 2001). "Direct association of Bloom's syndrome gene product with the human mismatch repair protein MLH1". Nucleic Acids Res..
- (August 2001). "The interaction of DNA mismatch repair proteins with human exonuclease I". J. Biol. Chem..
- (March 1999). "MED1, a novel human methyl-CpG-binding endonuclease, interacts with DNA mismatch repair protein MLH1". Proc. Natl. Acad. Sci. U.S.A..
- (August 2000). "MSH4 acts in conjunction with MLH1 during mammalian meiosis". FASEB J..
- (February 2003). "Interactions of the DNA mismatch repair proteins MLH1 and MSH2 with c-MYC and MAX". Oncogene.
- (April 2001). "The interacting domains of three MutL heterodimers in man: hMLH1 interacts with 36 homologous amino acid residues within hMLH3, hPMS1 and hPMS2". Nucleic Acids Res..
- (March 1999). "The interaction of the human MutL homologues in hereditary nonpolyposis colon cancer". J. Biol. Chem..
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about MLH1 — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report