From Surf Wiki (app.surf) — the open knowledge base
Mercury(II) sulfate
insoluble in ethanol, acetone, ammonia | NFPA-H = 3 | NFPA-F = 0 | NFPA-R = 1
Mercury(II) sulfate, commonly called mercuric sulfate, is the chemical compound HgSO4. It is an odorless salt that forms white granules or crystalline powder. In water, it separates into an insoluble basic sulfate with a yellow color and sulfuric acid.
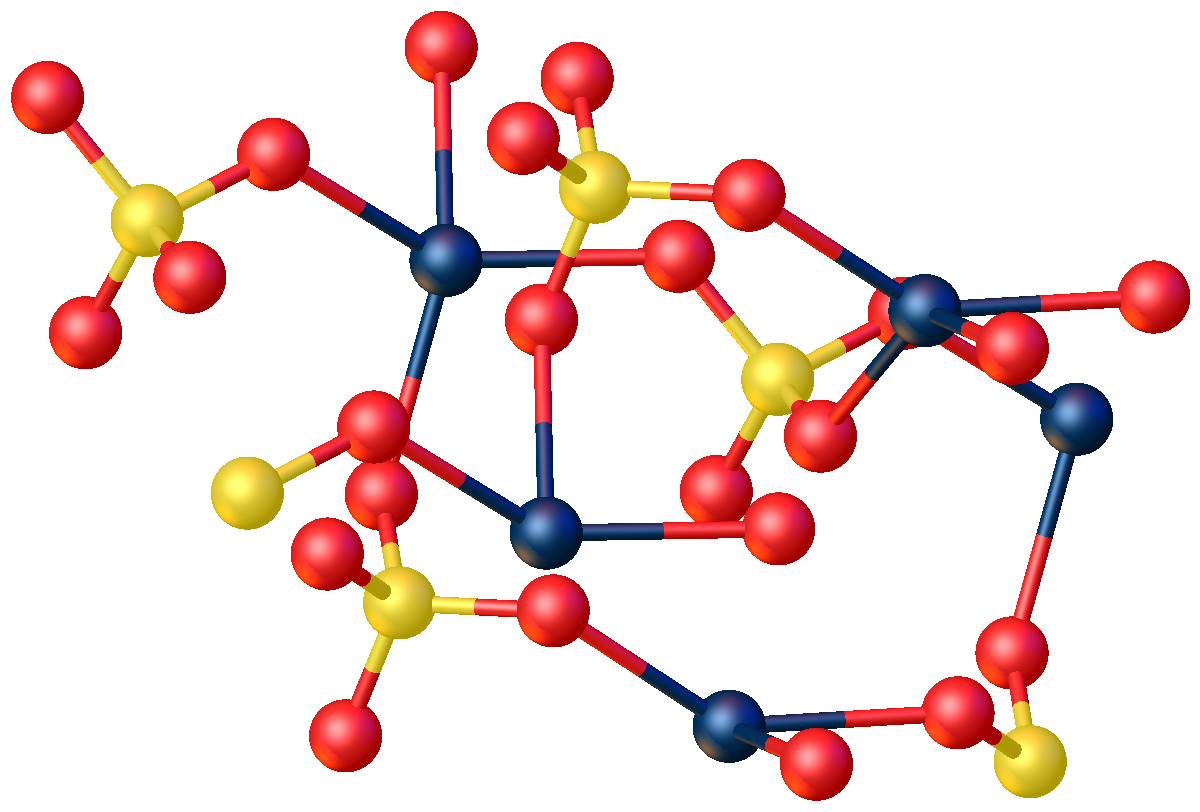
Structure

The anhydrous compound features Hg2+ in a highly distorted tetrahedral HgO4 environment. Two Hg–O distances are 2.22 Å and the others are 2.28 and 2.42 Å. In the monohydrate, Hg2+ adopts a linear coordination geometry with Hg–O (sulfate) and Hg–O (water) bond lengths of 2.179 and 2.228 Å respectively. Four weaker bonds are also observed with Hg–O distances 2.5 Å.
History
In 1932, the Japanese chemical company Chisso Corporation began using mercury sulfate as the catalyst for the production of acetaldehyde from acetylene and water. Though it was unknown at the time, methylmercury is formed as a side product of this reaction. Exposure and consumption of the mercury waste products, including methylmercury, that were dumped into Minamata Bay by Chisso are believed to be the cause of Minamata disease in Minamata, Japan.
Production
Mercury sulfate can be produced by treating mercury with hot concentrated sulfuric acid: :
Alternatively yellow mercuric oxide reacts also with concentrated sulfuric acid.
Uses
Denigés' reagent
An acidic solution of mercury sulfate is known as Denigés' reagent. It was commonly used throughout the 20th century as a qualitative analysis reagent. If Denigés' reagent is added to a solution containing compounds that have tertiary alcohols, a yellow or red precipitate will form.
Hydration reactions
.png)
Mercury sulfate, as well as other mercury(II) compounds, are commonly used as catalysts in oxymercuration-demercuration, a type of electrophilic addition reaction that results in hydration of an unsaturated compound. The hydration of an alkene gives an alcohol. The regioselectivity is that predicted by Markovnikov's rule. For an alkyne, the result is an enol, which tautomerizes to give the carbonyl. At one time, this chemistry was employed commercially for the preparation of acetaldehyde from acetylene: :
A related and specialized example is the conversion of 2,5-dimethylhexyne-2,5-diol to 2,2,5,5-tetramethyltetrahydrofuran using aqueous mercury sulfate without the addition of acid.
Health issues
Inhalation of HgSO4 can result in acute poisoning: causing tightness in the chest, difficulties breathing, coughing and pain. Exposure of HgSO4 to the eyes can cause ulceration of conjunctiva and cornea. If mercury sulfate is exposed to the skin it may cause sensitization dermatitis. Lastly, ingestion of mercury sulfate will cause necrosis, pain, vomiting, and severe purging. Ingestion can result in death within a few hours due to peripheral vascular collapse.
It was used in the late 19th century to induce vomiting for medical reasons.
References
References
- "Chemicalbook".
- Wu, Shengji. (2011). "Fundamental Study on Decomposition Characteristics of Mercury Compounds over Solid Powder by Temperature-Programmed Decomposition Desorption Mass Spectrometry". Energy & Fuels.
- Lide, David R.. (1998). "Handbook of Chemistry and Physics". CRC Press.
- "Mercuric sulfate".
- (1980). "A Reinvestigation of the Crystal Structures of HgSO4 and CdSO4". Zeitschrift für Kristallographie – Crystalline Materials.
- (1980). "An X-ray and Neutron Diffraction Study of Mercury(II) Sulphate Monohydrate". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry.
- [http://www.bu.edu/sustainability/minamata-disease/ Minamata Disease] {{webarchive. link. (13 November 2019. Boston University. Retrieved 2016-11-10.)
- (2006). "Ullmann's Encyclopedia of Industrial Chemistry".
- Robey, R. F.. (May 1947). "Test for tert-Butyl and Isopropyl Alcohols with Deniges Reagent". Analytical Chemistry.
- Marks, E.M.. (1939). "Reaction of Aliphatic Ethers with Denigés' Reagent". J. Org. Chem..
- (2012). "Organic chemistry". Oxford university press.
- (2006). "Ullmann's Encyclopedia of Industrial Chemistry".
- Wasacz, J. P.. (1982). "A hydration of an alkyne illustrating steam and vacuum distillation". Journal of Chemical Education.
- Hubbard. (1846-06-24). "On the Effects of the Sub-Sulphate of Mercury, (Turpeth Mineral,) As An Emetic". BMJ.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Mercury(II) sulfate — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report