From Surf Wiki (app.surf) — the open knowledge base
Mercury(II) oxide
Montroydite Red mercury 0.0395 g/100 mL (100 °C) |NFPA-H = 4 |NFPA-F = 0 |NFPA-R = 1 Mercury selenide Mercury telluride Cadmium oxide
Mercury(II) oxide, also called mercuric oxide or simply mercury oxide, is the inorganic compound with the formula HgO. It has a red or orange color. Mercury(II) oxide is a solid at room temperature and pressure. The mineral form montroydite is very rarely found.
History
An experiment for the preparation of mercuric oxide was first described by 11th century Arab-Spanish alchemist, Maslama al-Majriti, in Rutbat al-hakim. It was historically called red precipitate (as opposed to white precipitate, mercuric amidochloride).
In 1774, Joseph Priestley discovered that oxygen was released by heating mercuric oxide, although he did not identify the gas as oxygen (rather, Priestley called it "dephlogisticated air," as that was the paradigm that he was working under at the time).
Synthesis and reactions

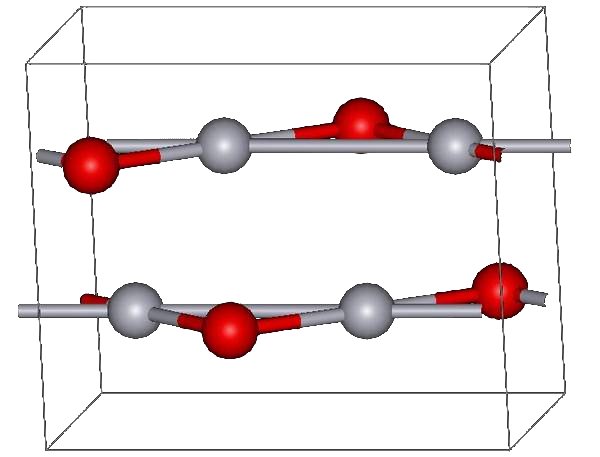
The red form of HgO can be made by heating Hg in oxygen at roughly 350 °C, or by pyrolysis of Hg(NO3)2. The yellow form can be obtained by precipitation of aqueous Hg2+ with alkali. The difference in color is due to particle size; both forms have the same structure consisting of near linear O-Hg-O units linked in zigzag chains with an Hg-O-Hg angle of 108°.
HgO is soluble in many conventional strong acids through protonation of the anion. The exceptions include acids which form insoluble mercury(II) salts, like mercury(II) iodide in the case of hydroiodic acid. Dissolution is also possible through complexation of the cation; e.g. cyanide ligands form stable water soluble mercury(II) complexes.
Structure
Under atmospheric pressure mercuric oxide has two crystalline forms: one is called montroydite (orthorhombic, 2/m 2/m 2/m, Pnma), and the second is analogous to the sulfide mineral cinnabar (hexagonal, hP6, P3221); both are characterized by Hg-O chains. At pressures above 10 GPa both structures convert to a tetragonal form.
Uses
Mercury oxide is sometimes used in the production of mercury as it decomposes quite easily. When it decomposes, oxygen gas is generated.
It is also used as a material for cathodes in mercury batteries.
Health issues

Mercury oxide is a highly toxic substance which can be absorbed into the body by inhalation of its aerosol, through the skin and by ingestion. The substance is irritating to the eyes, the skin and the respiratory tract and may have effects on the kidneys, resulting in kidney impairment. In the food chain important to humans, bioaccumulation takes place, specifically in aquatic organisms. The substance is banned as a pesticide in the EU.
Evaporation at 20 °C is negligible. HgO decomposes on exposure to light or on heating above 500 °C. Heating produces highly toxic mercury fumes and oxygen, which increases the fire hazard. Mercury(II) oxide reacts violently with reducing agents, chlorine, hydrogen peroxide, magnesium (when heated), disulfur dichloride and hydrogen trisulfide. Shock-sensitive compounds are formed with metals and elements such as sulfur and phosphorus.
References
References
- (1999). "Semiconductors · II-VI and I-VII Compounds; Semimagnetic Compounds". Springer-Verlag.
- Zumdahl, Steven S.. (2009). "Chemical Principles 6th Ed.". Houghton Mifflin Company.
- "Laboratory Chemical Safety Summary (LCSS): Mercuric oxide". [[National Center for Biotechnology Information]].
- (2021-12-25). "Safety Data Sheet: Mercury(II) oxide". [[Thermo Fisher Scientific]].
- "Mercuric oxide [ISO]". [[United States National Library of Medicine]].
- Holmyard, E. J.. (1931). "Inorganic chemistry". Рипол Классик.
- Almqvist, Ebbe. (2003). "History of Industrial Gases". Springer.
- {{Greenwood&Earnshaw
- (3 April 2018). "Characteristic Reactions of Mercury Ions (Hg²⁺ and Hg₂²⁺)".
- (1958). "The Structure of Hexagonal Mercury(II)oxide". Acta Chemica Scandinavica.
- Moore, John W.. (2005). "Chemistry: The Molecular Science". Thomson Brooks/Cole.
- Chemicals Regulation Directorate. "Banned and Non-Authorised Pesticides in the United Kingdom".
- "Mercury (II) oxide". International Occupational Safety and Health Information Centre.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Mercury(II) oxide — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report