From Surf Wiki (app.surf) — the open knowledge base
Melamine cyanurate
(C3H6N6·C3H3N3O3) | NFPA-H = | NFPA-F = | NFPA-R = | NFPA-S =
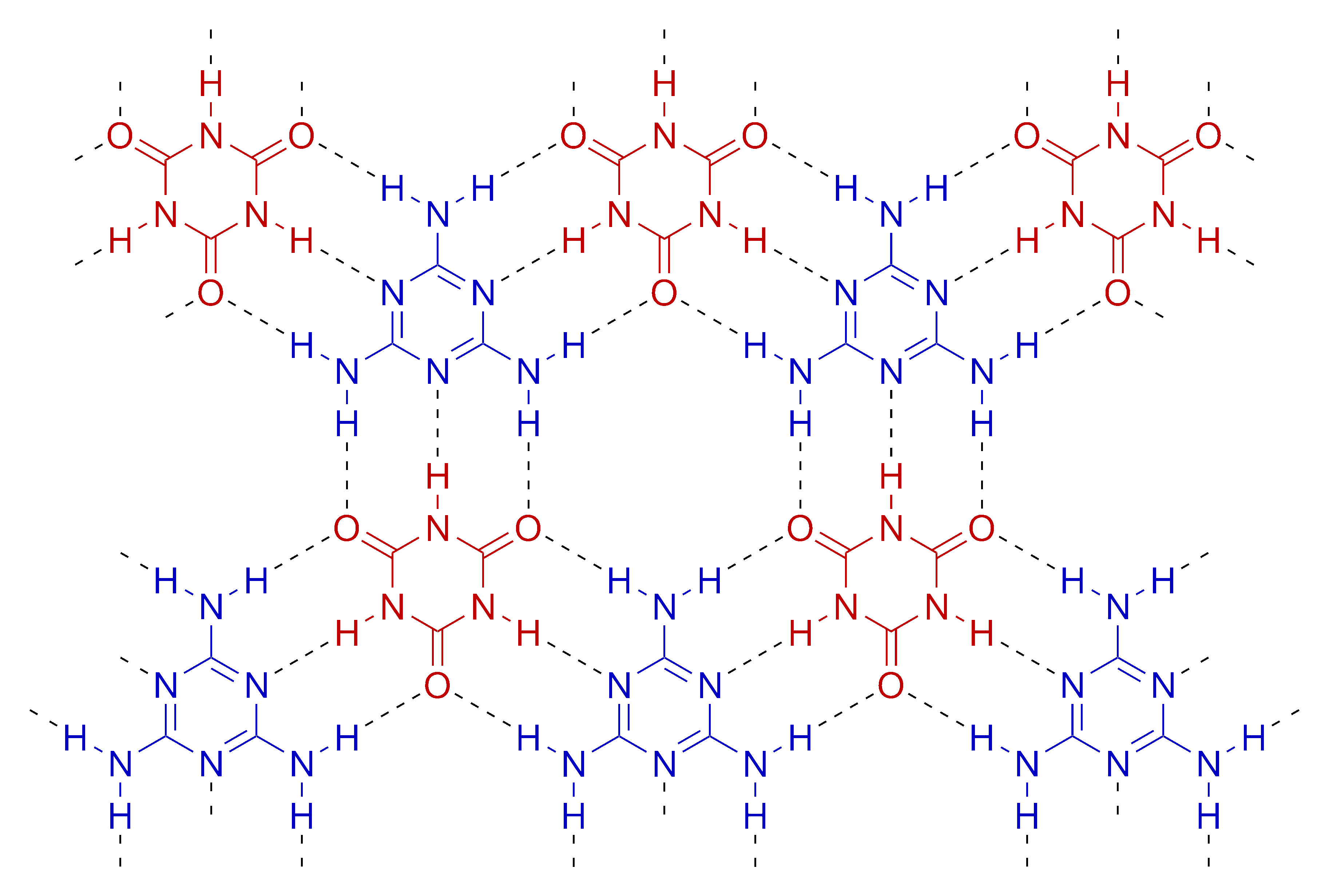
Melamine cyanurate, also known as melamine–cyanuric acid adduct or melamine–cyanuric acid complex, is a crystalline complex formed from a 1:1 mixture of melamine and cyanuric acid. The substance is not a salt despite its non-systematic name melamine cyanurate. The complex is held together by an extensive two-dimensional network of hydrogen bonds between the two compounds, reminiscent of the guanine–cytosine base pairs found in DNA. Melamine cyanurate forms spoke-like crystals from aqueous solutions {{cite journal and has been implicated as a causative agent for toxicity seen in the Chinese protein export contamination and the 2007 pet food recall. This complex is cited as an example of supramolecular chemistry.
Uses
Melamine cyanurate is used as a flame retardant, most commonly in polybutylene terephthalate (PBT), polyamide 6 (nylon 6) and polyamide 6,6 (nylon 6:6). It is also used to fireproof in polyester fabrics.
Toxicity
It has been considered to be more toxic than either melamine or cyanuric acid alone.
** in rats and mice (ingested):**
- 4.1 g/kg – Melamine cyanurate
- 6.0 g/kg – Melamine
- 7.7 g/kg – Cyanuric acid
A toxicology study conducted after recent pet food recalls concluded that the combination of melamine and cyanuric acid in diet does lead to acute kidney injury in cats. A 2008 study produced similar experimental results in rats and characterized the melamine and cyanuric acid in contaminated pet food from the 2007 outbreak.{{cite journal

References
References
- link. (2008-09-22)
- Lehn, J. M.. (1995). "Supramolecular Chemistry: Concepts and Perspectives". VCH.
- (2002). "Differences in the flame retardant mechanism of melamine cyanurate in polyamide 6 and polyamide 66". Polymer Degradation and Stability.
- A.A. Babayan, A.V.Aleksandryan, "Toxicological characteristics of melamine cyanurate, melamine and cyanuric acid", Zhurnal Eksperimental'noi i Klinicheskoi Meditsiny, Vol.25, 345-9 (1985). Original article in Russian.
- Puschner et al. (November 2007). Assessment of melamine and cyanuric acid toxicity in cats. Journal of Veterinary Diagnostic Investigation. Retrieved on 2007-11-16.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Melamine cyanurate — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report