From Surf Wiki (app.surf) — the open knowledge base
Malaria culture
Method for growing malaria parasites outside the body
Method for growing malaria parasites outside the body

Malaria culture is a method for growing malaria parasites outside the body, i.e., in an ex vivo environment. Although attempts for propagation of the parasites outside of humans or animal models reach as far back as 1912, the success of the initial attempts was limited to one or just a few cycles. The first successful continuous culture was established in 1976. Initial hopes that the ex vivo culture would lead quickly to the discovery of a vaccine were premature. However, the development of new drugs was greatly facilitated.
Method

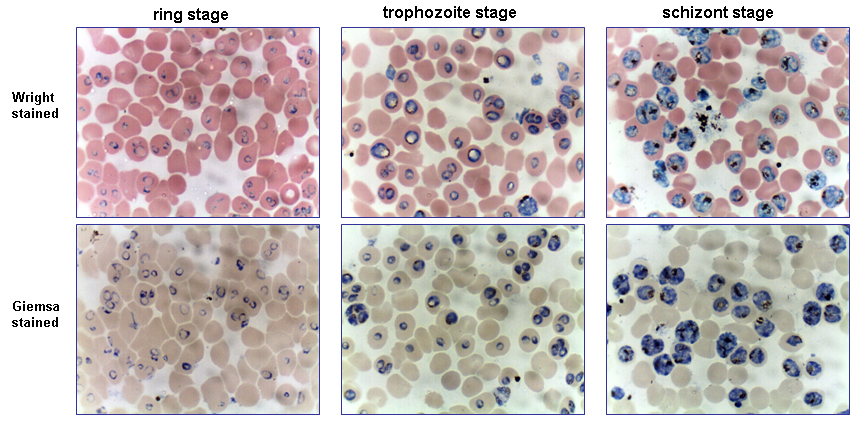
Infected human red blood cells are incubated in a culture dish or flask at 37 °C together with a nutrient medium and plasma, serum or serum substitutes. A special feature of the incubation is the special gas mixture filled with nitrogen (90-92 %), CO2 (5 %) and oxygen (3-5 %), allowing the parasites to grow at 37 °C in a cell incubator. An alternative to gassing the cultures with the exact gas mixture, is the use of a candlejar. The candlejar is an airtight container in which the cultures and a lit candle are placed. The burning candle consumes some of the oxygen and produces carbon dioxide (CO2), which acts as a fire extinguisher. Carbon dioxide content in fresh air varies between 0.036 % and 0.039 %. Once the CO2 concentration reaches approximately 5 %, the candle stops burning. The number of parasites increased by a factor 5 approximately every 48 hours (one cycle). The parasitemia can be determined via blood film, to keep it within the wanted limits, the culture can be thinned out with healthy red blood cells.

The original method for the successful ex vivo propagation of P. falciparum described culture of the parasite under static conditions (Trager-Jensen method). Despite this, the practice of culturing the parasite under static conditions remains widespread. The greatest value of the candlejar method is that it can be used in laboratories almost anywhere in the world where there is an incubator, a candle and a desiccator. Around 60% parasitized cells can be obtained using optimized culturing conditions. Recent studies of P. falciparum isolated directly from infected patients indicate that alternative parasite biological states occur in the natural host that are not observed with ex vivo cultivated parasites.
Concentration of infected cells


To achieve synchronization and/or concentration of the parasites in culture several methods have been developed. A discontinuous Percoll gradient procedure can be used to isolate infected red blood cells because red cells containing plasmodia are less dense than normal ones. Young trophozoites coincided with erythrocytes in a broad band corresponding to densities from 1.075 to 1.100 g/ml, whereas schizonts were concentrated at a density approximating 1.062 g/ml. There are studies, however, that suggest that some strains of P.falciparum are affected in their capacity of invasion after being exposed to this chemical. The difference between diamagnetic low-spin oxyhemoglobin in uninfected red blood cells and paramagnetic hemozoin in infected red blood cells can also be used for isolation. Magnetic columns have shown to be less harmful for the parasite and are simple and adjustable to the needs of the researcher. The column is mounted in a potent magnet holder and the culture flowed through it. The column traps the erythrocytes infected with the latest stages of the parasites, which can then be eluted when the column is removed from the magnet. It is a simple method that does not need expensive equipment and it does not seem to affect the parasites as to their invasion capabilities afterwards.
References
References
- [[Charles C. Bass. (1912). "The Cultivation of Malarial Plasmodia (Plasmodium vivax and Plasmodium Falciparum) ''in vitro''". J. Exp. Med..
- (1976). "Human malaria parasites in continuous culture". Science.
- (1997). "Continuous culture of Plasmodium falciparum: its impact on malaria research". Int. J. Parasitol..
- Basco LK. (2003). "Molecular epidemiology of malaria in Cameroon. XV. Experimental studies on serum substitutes and supplements and alternative culture media for in vitro drug sensitivity assays using fresh isolates of Plasmodium falciparum". Am. J. Trop. Med. Hyg..
- Trigg PI. (1985). "Recent advances in malaria parasite cultivation and their application to studies on host-parasite relationships: a review". Bull. World Health Organ..
- Schuster FL. (2002). "Cultivation of plasmodium spp". Clin. Microbiol. Rev..
- (1982). "The use of percoll gradients, elutriator rotor elution, and mithramycin staining for the isolation and identification of intraerythrocytic stages of plasmodium berghei". Z. Parasitenkd..
- (2010). "''Plasmodium falciparum'' culture: The benefits of shaking". Mol. Biochem. Parasitol..
- Sherman, I. W.. (2010). "Magic Bullets to Conquer Malaria. From Quinine to Qinghaosu". ASM Press.
- (2009). "Synchronous culture of Plasmodium falciparum at high parasitemia levels". Nat. Protoc..
- (2009). "Plasmodium falciparum biology: analysis of in vitro versus in vivo growth conditions". Trends Parasitol..
- (2013). "Separation of Plasmodium falciparum Late Stage-infected Erythrocytes by Magnetic Means". J. Vis. Exp..
- (February 2019). "Separation and concentration of schizonts of Plasmodium falciparum by Percoll gradients". J. Protozool..
- (2010). "Improved methods for magnetic purification of malaria parasites and haemozoin". Malar. J..
- (2010). "Optimized high gradient magnetic separation for isolation of Plasmodium-infected red blood cells". Malar. J..
- (2011). "Comparison of the in vitro invasive capabilities of Plasmodium falciparum schizonts isolated by Percoll gradient or using magnetic based separation". Malar. J..
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Malaria culture — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report