From Surf Wiki (app.surf) — the open knowledge base
Lithium iodide
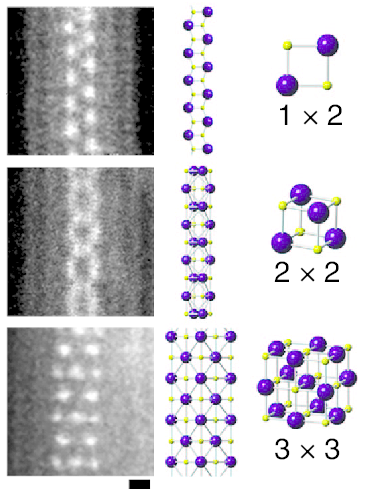
3.494 g/cm3 (trihydrate) 1670 g/L (25 °C) 4330 g/L (100 °C) | NFPA-H = 2 | NFPA-F = 0 | NFPA-R = 0 Lithium chloride Lithium bromide Lithium astatide Potassium iodide Rubidium iodide Caesium iodide Francium iodide Lithium iodide, or LiI, is a compound of lithium and iodine. When exposed to air, it becomes yellow in color, due to the oxidation of iodide to iodine. It crystallizes in the NaCl motif. It can participate in various hydrates.
Applications

Lithium iodide is used as a solid-state electrolyte for high-temperature batteries. It is also the standard electrolyte in artificial pacemakers due to the long cycle life it enables. The solid is used as a phosphor for neutron detection. It is also used, in a complex with Iodine, in the electrolyte of dye-sensitized solar cells.
In organic synthesis, LiI is useful for cleaving C-O bonds. For example, it can be used to convert methyl esters to carboxylic acids: : Similar reactions apply to epoxides and aziridines.
Lithium iodide was used as a radiocontrast agent for CT scans. Its use was discontinued due to renal toxicity. Inorganic iodine solutions suffered from hyperosmolarity and high viscosities. Current iodinated contrast agents are organoiodine compounds.
It is also useful in MALDI imaging mass spectrometry of lipids by adding lithium salts to the matrix solution.
References
References
- Patnaik, Pradyot (2002) ''Handbook of Inorganic Chemicals''. McGraw-Hill, {{ISBN. 0-07-049439-8
- "Lithium iodide". ESPI Corp. MSDS.
- Wells, A.F. (1984) ''Structural Inorganic Chemistry'', Oxford: Clarendon Press. {{ISBN. 0-19-855370-6.
- Wietelmann, Ulrich and Bauer, Richard J. (2005) "Lithium and Lithium Compounds" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH: Weinheim. {{doi. 10.1002/14356007.a15_393.
- (2015). "Single-atom electron energy loss spectroscopy of light elements". Nature Communications.
- Holmes, C.. (2007-09-28). "The Lithium/Iodine-Polyvinylpyridine Pacemaker Battery - 35 years of Successful Clinical Use". ECS Transactions.
- Hanif, Maryam. (2008). "The Pacemaker Battery - Review Article". UIC Bioengineering Student Journal.
- (1955). "Some lithium iodide phosphors for slow neutron detection". Br. J. Appl. Phys..
- Charette, André B.; Barbay, J. Kent and He, Wei (2005) "Lithium Iodide" in ''Encyclopedia of Reagents for Organic Synthesis'', John Wiley & Sons. {{doi. 10.1002/047084289X.rl121.pub2
- (2013). "X-ray-Computed Tomography Contrast Agents". Chemical Reviews.
- (2011). "MALDI imaging mass spectrometry of lipids by adding lithium salts to the matrix solution". Analytical and Bioanalytical Chemistry.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Lithium iodide — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report