From Surf Wiki (app.surf) — the open knowledge base
List of psychoactive plants
List of plant species with reported psychoactive properties
List of plant species with reported psychoactive properties
Main article: Psychoactive plant
A list of plant species that, when consumed by humans, are known or suspected to produce psychoactive effects: changes in nervous system function that alter perception, mood, consciousness, cognition or behavior. Many of these plants are used intentionally as psychoactive drugs, for medicinal, religious, and/or recreational purposes. Some have been used ritually as entheogens for millennia.
The plants are listed according to the specific psychoactive chemical substances they contain; many contain multiple known psychoactive compounds.
Cannabinoids

Main article: Cannabis
Species of the genus Cannabis, known colloquially as marijuana, including Cannabis sativa and Cannabis indica, are popular psychoactive plants that are often used medically and recreationally. The principal psychoactive substance in Cannabis, tetrahydrocannabinol (THC), contains no nitrogen, unlike many (but not all) other psychoactive substances and is not an indole, tryptamine or phenethylamine. THC is just one of more than 100 identified cannabinoid compounds in Cannabis, which also include cannabinol (CBN) and cannabidiol (CBD).
Cannabis plants vary widely, with different strains producing dynamic balances of cannabinoids (THC, CBD, etc.) and yielding markedly different effects. Popular strains are often hybrids of C. sativa and C. indica.
The medicinal effects of cannabis are widely studied, and are active topics of research both at universities and private research firms. Many jurisdictions have laws regulating or prohibiting the cultivation, sale and/or use of medical and recreational cannabis.
Tryptamines
_Delosperma_cooperi_-_Flower.jpg)

Many of the psychedelic plants contain dimethyltryptamine (DMT), or other tryptamines, which are either snorted (Virola, Yopo snuffs), vaporized, or drunk with MAOIs (Ayahuasca). It cannot simply be eaten as it is not orally active without an MAOI, and must be extremely concentrated in order to be vaporized.
[[Acanthaceae]]
"Species*, *Alkaloid content, where given, refers to dried material"
- Fittonia albivenis, a common ornamental plant from South America.
[[Aceraceae]]
- Acer saccharinum (silver maple) was found to contain the indole alkaloid gramine (not active and extremely toxic) 0.05% in the leaves, so it is possible that other members of this plant family contain active compounds.
[[Aizoaceae]]
- Delosperma acuminatum, DMT, 5-MeO-DMT
- Delosperma cooperi, DMT, 5-MeO-DMT
- Delosperma ecklonis, DMT
- Delosperma esterhuyseniae, DMT
- Delosperma hallii, 5-MeO-DMT
- Delosperma harazianum, DMT, 5-MeO-DMT
- : Delosperma harazianum Shibam, DMT
- Delosperma hirtum, DMT
- : Delosperma hallii aff. litorale
- Delosperma lydenbergense, DMT, 5-MeO-DMT
- Delosperma nubigenum, 5-MeO-DMT
- Delosperma pageanum, DMT, 5-MeO-DMT
- Delosperma pergamentaceum, Traces of DMT
- Delosperma tradescantioides, DMT
[[Apocynaceae]]
- Prestonia amazonica: DMT{{cite web |access-date=2008-04-19 |archive-url=https://web.archive.org/web/20020522235849/http://paranoia.lycaeum.org/psychedelics/tryptamines/dmt.info |archive-date=2002-05-22
- Voacanga africana: Up to 10% Iboga alkaloids
[[Asteraceae]]
- Pilosella officinarum
[[Erythroxylaceae]]
- Erythroxylum pungens: DMT
[[Fabaceae]] (Leguminosae)













- Acacia acuminata, Up to 1.5% alkaloids, mainly consisting of dimethyltryptamine in bark & leaf{{cite web |access-date=2008-02-23 |archive-url=https://web.archive.org/web/20071012182142/http://leda.lycaeum.org/?ID=15928 |archive-date=2007-10-12
- Acacia alpina, Active principles in leaf
- Acaciella angustissima, β-methyl-phenethylamine,{{cite book
- Vachellia aroma, Tryptamine alkaloids. Significant amount of tryptamine in the seeds.
- Acacia auriculiformis, 5-MeO-DMT in stem bark
- Acacia baileyana, 0.02% tryptamine and β-carbolines, in the leaf, Tetrahydroharman
- Acacia beauverdiana, Psychoactive Ash used in Pituri.
- Senegalia berlandieri, DMT, phenethylamine, mescaline, nicotine
- Senegalia catechu, DMT and other tryptamines in leaf, bark
- Vachellia caven, Psychoactive
- Senegalia chundra, DMT and other tryptamines in leaf, bark
- Acacia colei, DMT
- Acacia complanata, 0.3% alkaloids in leaf and stem, almost all N-methyl-tetrahydroharman, with traces of tetrahydroharman, some of tryptamine
- Acacia confusa, DMT & NMT in leaf, stem & bark 0.04% NMT and 0.02% DMT in stem. Also N,N-dimethyltryptamine N-oxideNMR spectral assignments of a new chlorotryptamine alkaloid and its analogues from Acacia confusa Malcolm S. Buchanan, Anthony R. Carroll, David Pass, Ronald J. Quinn Magnetic Resonance in Chemistry Volume 45, Issue 4, pp. 359–361. John Wiley & Sons, Ltd.
- Vachellia cornigera, Psychoactive, Tryptamines DMT according to C. Rastch.
- Acacia cultriformis, Tryptamine, in the leaf, stem and seeds. Phenethylamine in leaf and seeds
- Acacia cuthbertsonii, Psychoactive
- Acacia decurrens, Psychoactive, but less than 0.02% alkaloids
- Acacia delibrata, Psychoactive
- Acacia falcata, Psychoactive, but less than 0.02% alkaloids
- Vachellia farnesiana, Traces of 5-MeO-DMT in fruit. β-methyl-phenethylamine, flower. Ether extracts about 2–6% of the dried leaf mass. Alkaloids are present in the bark and leaves.
- Acacia flavescens, Strongly Psychoactive, Bark
- Acacia floribunda, Tryptamine, phenethylamine,{{cite book |access-date=2008-02-23 |archive-url=https://web.archive.org/web/20071012182159/http://leda.lycaeum.org/?ID=16001 |archive-date=2007-10-12
- Acacia georginae, Psychoactive, plus deadly toxins
- Vachellia horrida, Psychoactive
- Acacia implexa, Psychoactive
- Mimosa jurema, DMT, NMT
- Vachellia karroo, Psychoactive
- Senegalia laeta, DMT, in the leaf
- Acacia longifolia, 0.2% tryptamine in bark, leaves, some in flowers, phenylethylamine in flowers, 0.2% DMT in plant.{{cite web |access-date=2008-02-23 |archive-url=https://web.archive.org/web/20070418113901/http://leda.lycaeum.org/?ID=15799 |archive-date=2007-04-18
- Acacia sophorae, Tryptamine in leaves, bark
- Acacia macradenia, Tryptamine
- Acacia maidenii, 0.6% NMT and DMT in about a 2:3 ratio in the stem bark, both present in leaves
- Acacia mangium, Psychoactive
- Acacia melanoxylon, DMT, in the bark and leaf, but less than 0.02% total alkaloids
- Senegalia mellifera, DMT, in the leaf
- Vachellia nilotica, DMT, in the leaf
- Vachellian ilotica subsp. adstringens, Psychoactive, DMT in the leaf
- Acacia neurophylla DMT in bark, Harman in leaf.
- Acacia obtusifolia, Tryptamine, DMT, NMT, other tryptamines, 0.4–0.5% in dried bark,0.15–0.2% in leaf, 0.07% in branch tips.
- Vachellia oerfota, Less than 0.1% DMT in leaf, NMT
- Acacia penninervis, Psychoactive
- Acacia phlebophylla, 0.3% DMT in leaf, NMT
- Acacia podalyriifolia, Tryptamine in the leaf, 0.5% to 2% DMT in fresh bark, phenethylamine, trace amounts. Although this species is claimed to contain 0.5% to 2% DMT in fresh bark the reference for this is invalid as there is no reference to Acacia Podalyriffolia anywhere in the reference article. Additionally, well known and proven extraction techniques for DMT have failed to produce any DMT or alkaloids from fresh bark or the leaves on multiple sample taken at various seasons. Should DMT actually exist in this species of Acacia then it exists in extremely small amounts and have failed to produce any alkaloids with Acid/Base extraction techniques using HCl/NaOH. On the same note, more academic research is definitely required into the DMT content of this and other Australian Acacia species with proper chemical analysis of sample.
- Senegalia polyacantha, DMT in leaf and other tryptamines in leaf, bark
- Senegalia polyacantha ssp. campylacantha, Less than 0.2% DMT in leaf, NMT; DMT and other tryptamines in leaf, bark{{cite web |access-date = 2008-02-23 |archive-url = https://web.archive.org/web/20071012181801/http://hortipedia.org/de/index.php?title=Acacia_campylacantha |archive-date = 2007-10-12
- Senegalia rigidula: Phenethylamine, tryptamine, tyramine, and β-Methylphenethylamine.
- Acacia sassa, Psychoactive
- Vachellia schaffneri, β-methyl-phenethylamine, Phenethylamine
- Senegalia senegal, Less than 0.1% DMT in leaf, NMT, other tryptamines. DMT in plant, DMT in bark.
- Vachellia seyal, DMT, in the leaf. Ether extracts about 1–7% of the dried leaf mass.
- Vachellia sieberiana, DMT, in the leaf
- Acacia simplex, DMT and NMT, in the leaf, stem and trunk bark, 0.81% DMT in bark, MMT
- Vachellia tortilis, DMT, NMT, and other tryptamines
- Acacia vestita, Tryptamine, in the leaf and stem, but less than 0.02% total alkaloids
- Acacia victoriae, tryptamines, 5-MeO-alkyltryptamine
- List of acacia species having little or no alkaloids in the material sampled:
(0% \le C \le 0.02%, Concentration of alkaloids)
- Acacia acinacea
- Acacia baileyana
- Acacia decurrens
- Acacia dealbata
- Acacia mearnsii
- Acacia drummondii
- Acacia elata
- Acacia falcata
- Acacia leprosa
- Acacia linearis
- Acacia melanoxylon
- Acacia pycnantha
- Acacia retinodes
- Acacia saligna
- Acacia stricta
- Acacia verticillata
- Acacia vestita
- Pseudalbizzia inundata leaves contain DMT.
- Anadenanthera colubrina, Bufotenin, Beans, Bufotenin oxide, Beans, N,N-Dimethyltryptamine, Beans, pods,
- Anadenanthera colubrina var. cebil – Bufotenin and Dimethyltryptamine have been isolated from the seeds and seed pods, 5-MeO-DMT from the bark of the stems. The seeds were found to contain 12.4% bufotenine, 0.06% 5-MeO-DMT and 0.06% DMT.{{cite web |access-date=2008-02-23
- Anadenanthera peregrina, 1,2,3,4-Tetrahydro-6-methoxy-2,9-dimethyl-beta-carboline, Plant, 1,2,3,4-Tetrahydro-6-methoxy-2-methyl-beta-carboline, Plant, 5-Methoxy-N,N-dimethyltryptamine, Bark, 5-Methoxy-N-methyltryptamine, Bark, Bufotenin, plant, beans, Bufotenin N-oxide, Fruit, beans, N,N-Dimethyltryptamine-oxide, Fruit
- Anadenanthera peregrina var. peregrina, Bufotenine is in the seeds.
- Desmanthus illinoensis, 0–0.34% DMT in root bark, highly variable. Also NMT, N-hydroxy-N-methyltryptamine, 2-hydroxy-N-methyltryptamine, and gramine (toxic). |access-date=2008-05-08
- Desmanthus leptolobus, 0.14% DMT in root bark, more reliable than D. illinoensis
- Desmodium caudatum (syn. Ohwia caudata), Roots: 0.087% DMT,
- Codariocalyx motorius(syn. Desmodium gyrans), DMT, 5-MeO-DMT, leaves, roots
- Desmodium racemosum, 5-MeO-DMT
- Desmodium triflorum, 0.0004% DMT-N-oxide, roots, less in stems and trace in leaves.
- Lespedeza capitata
- Lespedeza bicolor, DMT, Lespedamine, and 5-MeO-DMT in leaves and roots
- Lespedeza bicolor var. japonica, DMT, 5-MeO-DMT in leaves and root bark
- Mimosa ophthalmocentra, Dried root: DMT 1.6%, NMT 0.0012% and hordenine 0.0065%
- Mimosa scabrella, tryptamine, NMT, DMT and N-methyltetrahydrocarboline in bark |access-date=2008-05-07
- Mimosa somnians, tryptamines and MMT
- Mimosa tenuiflora (syn. "Mimosa hostilis"), 1-1.7% DMT (dry root bark).
- Mimosa verrucosa, DMT |archive-url=https://web.archive.org/web/20070708155740/http://www.unodc.org/unodc/en/bulletin/bulletin_1969-01-01_4_page004.html |archive-date=2007-07-08
- Mucuna pruriens, the seeds of the plant contain about 3.1–6.1% L-DOPA.
- Petalostylis casseoides, 0.4–0.5% tryptamine, DMT, etc. in leaves and stems
- Petalostylis labicheoides var. casseoides, DMT in leaves and stems; 0.4–0.5% alkaloids in leaves and stems; Tryptamines in leaves and stems, MAO's up to 0.5%
- Phyllodium pulchellum(syn. Desmodium pulchellum), DMT; 0.2% 5-MeO-DMT, small quantities of DMT DMT (dominates in seedlings and young plants), 5-MeO-DMT (dominates in mature plant), whole plant, roots, stems, leaves, flowers;
- Erythrina flabelliformis, other Erythrina species, seeds contain the alkaloids erysodin and erysovin |access-date=2008-04-18
- Zornia latifolia, the flavones genistein, apigenin and syzalterin may explain the cannabis-like effects
[[Lauraceae]]
- Nectandra megapotamica, NMT
[[Malpighiaceae]]
- Diplopterys cabrerana: McKenna et al. (1984) assayed and found the leaves contain 0.17% DMT |url-access = limited
[[Myristicaceae]]
- Horsfieldia superba: 5-MeO-DMT, Horsfiline, and beta-carbolines
- Iryanthera macrophylla: 5-MeO-DMT in bark;
- Iryanthera ulei: 5-MeO-DMT in bark
- Osteophloem platyspermum: DMT, 5-MeO-DMT in bark
- Virola calophylla, Leaves 0.149% DMT, leaves 0.006% MMT 5-MeO-DMT in bark |access-date = 2008-04-11 |archive-url = https://web.archive.org/web/20041110182051/http://sun.ars-grin.gov:8080/npgspub/xsql/duke/plantdisp.xsql?taxon=1065 |archive-date = 2004-11-10
- Virola calophylloidea, DMT, 5-MeO-DMT
- Virola carinata, DMT in leaves; DMT, 5-MeO-DMT
- Virola cuspidata, DMT
- Virola divergens, DMT in leaves
- Virola elongata(syn. Virola theiodora), DMT, 5-MeO-DMT in bark, roots, leaves and flowers
- Virola melinonii, DMT in bark; DMT, 5-MeO-DMT
- Virola multinervia, DMT, 5-MeO-DMT in bark and roots
- Virola pavonis, DMT in leaves
- Virola peruviana, DMT, 5-MeO-DMT; 5-MeO-DMT, traces of DMT and 5-MeO-tryptamine in bark
- Virola rufula, Alkaloids in bark and root, 95% of which is MeO-DMT 0.190% 5-MeO-DMT in bark, 0.135% 5-MeO-DMT in root, 0.092% DMT in leaves.
- Virola sebifera, The bark contains 0.065% to 0.25% alkaloids, most of which are DMT and 5-MeO-DMT.
- Virola venosa, DMT, 5-MeO-DMT in roots, leaves DMT
[[Ochnaceae]]
- Testulea gabonensis: 0.2% 5-MeO-DMT, small quantities of DMT, DMT in bark and root bark, NMT
[[Pandanaceae]]
- Genus Pandanus (Screw Pine): DMT in nuts
[[Poaceae]] (Gramineae)
Some Graminae (grass) species contain gramine, which can cause brain damage, other organ damage, central nervous system damage and death in sheep. |access-date=2008-04-20
- Arundo donax, 0.0057% DMT in dried rhizome, no stem, 0.026% bufotenine, 0.0023% 5-MeO-MMT
- Phalaris aquatica, 0.0007–0.18% Total alkaloids, 0.100% DMT, 0.022% 5-MeO-DMT, 0.005% 5-OH-DMT
- Phalaris arundinacea, 0.0004–0.121% Total alkaloids
- Phalaris brachystachys, aerial parts up to 3% total alkaloids, DMT present
- Phalaris coerulescens, Coerulescine and 2-methyl-1,2,3,4-Tetrahydro-β-carboline in rhizome.
- Phragmites australis, DMT, 5-MeO-DMT, bufotenine and gramine in the rhizome.
None of the above alkaloids are said to have been found in Phalaris californica, Phalaris canariensis, Phalaris minor and hybrids of P. arundinacea together with P. aquatica.
[[Polygonaceae]]
- Eriogonum : DMT
[[Rubiaceae]]
- Psychotria carthagenensis, 0.2% average DMT in dried leaves.
- Psychotria colorata, Presence of mu opioid receptor(MOR) agonist and NMDA antagonist: hodgkinsine, psychotridine. Also mentioned in The Encyclopedia of Psychoactive Plants: Ethnopharmacology and Its Applications.
- Psychotria expansa, DMT
- Psychotria forsteriana, DMT
- Psychotria insularum, DMT
- Psychotria poeppigiana, DMT
- Psychotria rostrata, DMT
- Psychotria rufipilis, DMT
- Psychotria viridis, DMT 0.1–0.61% dried mass.
[[Rutaceae]]
Source:
- Dictyoloma incanescens, 5-MeO-DMT in leaves, 0.04% 5-MeO-DMT in bark
- Dutaillyea drupacea, 0.4% 5-MeO-DMT in leaves
- Dutaillyea oreophila, 5-MeO-DMT in leaves
- Tetradium ruticarpum (syn. Evodia rutaecarpa), 5-MeO-DMT in leaves, fruit and roots
- Limonia acidissima, traces of DMT; 5-MeO-DMT in stems
- Euodia leptococca (formerly Melicope), 0.2% total alkaloids, 0.07% 5-MeO-DMT; 5-MeO-DMT in leaves and stems, also "5-MeO-DMT-Oxide and a beta-carboline"
- Pilocarpus organensis, DMT, 5-MeO-DMT in leaves (Might also contain pilocarpine)
- Vepris ampody, up to 0.2% DMT in leaves and branches
- Zanthoxylum arborescens, traces of DMT; DMT in leaves
- Zanthoxylum procerum, DMT in leaves
- Citrus limon, DMT, N-Methylated tryptamine derivative in leaves
- Citrus sinesis, DMT, N-Methylated tryptamine derivative
- Citrus bergamia, DMT, N-Methylated tryptamine derivative
- Mandarin orange, traces of N-methylated tryptamine derivative in leaf.
- Chinotto Tree, N-Methylated tryptamine derivative in leaf
- Citrus medica, N-Methylated tryptamine derivative in leaf
Phenethylamines
Main article: Psychoactive cactus

MESCALINE CACTI
Species, Alkaloid Content (Fresh) – Alkaloid Content (Dried)
- Coryphantha contains various phenethylamine alkaloids including macromerine, coryphanthine, O-methyl-candicine, corypalmine, and N-methyl-corypalmine.
- Cylindropuntia echinocarpa (syn. Opuntia echinocarpa), Mescaline 0.01%, DMPEA 0.01%, 4-hydroxy-3-5-dimethoxyphenethylamine 0.01%
- Cylindropuntia spinosior (syn. Opuntia spinosior), Mescaline 0.00004%, 3-methoxytyramine 0.001%, tyramine 0.002%, 3-4-dimethoxyphenethylamine.
- Echinopsis lageniformis (syns Echinopsis scopulicola, Trichocereus bridgesii), Mescaline 0.025%, also DMPEA
- Echinopsis macrogona (syn. Trichocereus macrogonus), 0.01–0.05% Mescaline
- [[File:Echinopsis pachanoi (7996941451).jpg|thumb|Echinopsis pachanoi]]Echinopsis pachanoi (syn. Trichocereus pachanoi), Mescaline 0.006–0.12%, 0.05% Average; Mescaline 0.01%–2.375%
- Echinopsis peruviana (syn. Trichocereus peruvianus), Mescaline 0.0005%–0.12%; Mescaline
- Echinopsis spachiana (syn. Trichocereus spachianus), Mescaline; Mescaline
- Echinopsis tacaquirensis subsp. taquimbalensis (syn. Trichocereus taquimbalensis), 0.005–0.025% mescaline
- Echinopsis terscheckii (syn. Trichocereus terscheckii, Trichocereus werdemannianus) 0.005–0.025% Mescaline; mescaline 0.01%–2.375%
- Echinopsis valida, 0.025% mescaline
- Lophophora williamsii (Peyote), 0.4% Mescaline; 3–6% Mescaline
- Opuntia acanthocarpa Mescaline
- Opuntia basilaris Mescaline 0.01%, plus 4-hydroxy-3-5-dimethoxyphenethylamine
- Pelecyphora aselliformis, mescaline
OTHER
Eria Jarens- N,N-Dimethylphenethylamine
Beta-carbolines




Beta-carbolines are "reversible" MAO-A inhibitors. They are found in some plants used to make Ayahuasca. In high doses the harmala alkaloids are somewhat hallucinogenic on their own. β-carboline is a benzodiazepine receptor inverse agonist and can therefore have convulsive, anxiogenic and memory enhancing effects.
[[Apocynaceae]]
- Amsonia tabernaemontana, harman
- Aspidosperma exalatum, beta-carbolines
- Aspidosperma polyneuron, beta-carbolines
- Apocynum cannabinum, harmalol
- Ochrosia nakaiana, harman
- Pleiocarpa mutica, beta-carbolines
[[Bignoniaceae]]
- Newbouldia laevis, harman
[[Calycanthaceae]]
- Calycanthus occidentalis, harman; Harmine
[[Chenopodiaceae]]
- Hammada leptoclada, harman; Tetrahydroharman, etc.
- Kochia scoparia, harman; Harmine, etc.
[[Combretaceae]]
- Guiera senegalensis, tetrahydroharmine; Harman, etc.
[[Cyperaceae]]
- Carex brevicollis, harmine, etc.
- Carex parva, beta-carbolines
[[Elaeagnaceae]]
- Elaeagnus angustifolia, harman, etc.
- Elaeagnus commutata, beta-carbolines
- Elaeagnus hortensis, tetrahydroharman, etc.
- Elaeagnus orientalis, tetrahydroharman
- Elaeagnus spinosa, tetrahydroharman
- Hippophae rhamnoides, harman, etc.
- Shepherdia argentea, tetrahydroharmol
- Shepherdia canadensis, tetrahydroharmol
[[Gramineae]]
- Arundo donax, tetrahydroharman, etc.
- Festuca arundinacea, harman, etc.
- Lolium perenne (perennial ryegrass), harman, etc.
- Phalaris aquatica, beta-carbolines
- Phalaris arundinacea, beta-carbolines
[[Lauraceae]]
- Nectandra megapotamica, beta-carbolines
[[Leguminosae]]
- Acacia baileyana, tetrahydroharman
- Acacia complanata, tetrahydroharman, etc.
- Burkea africana, harman, etc.
- Desmodium gangeticum, beta-carbolines
- Desmodium gyrans, beta-carbolines
- Mucuna pruriens, 6-methoxyharman, dihydroharman, harman
- Petalostylis labicheoides, tetrahydroharman; MAOs up to 0.5%
- Prosopis nigra, harmalicin, harman, etc.
- Shepherdia pulchellum, beta-carbolines
[[Loganiaceae]]
- Strychnos melinoniana, beta-carbolines
- Strychnos usambarensis, harman
[[Malpighiaceae]]
- Banisteriopsis argentia, 5-methoxytetrahydroharman, (−)-N(6)-methoxytetrahydroharman, dimethyltryptamine-N(6)-oxide
- Banisteriopsis caapi, Harmine 0.31–0.84%, tetrahydroharmine, telepathine, dihydroshihunine, 5-MeO-DMT in bark{{cite web |access-date = 2008-04-11 |archive-url = https://web.archive.org/web/20041121010104/http://sun.ars-grin.gov:8080/npgspub/xsql/duke/chemdisp.xsql |archive-date = 2004-11-21
- Banisteriopsis inebrians, beta-carbolines
- Banisteriopsis lutea, harmine, telepathine
- Banisteriopsis metallicolor, harmine, telepathine
- Banisteriopsis muricata, harmine up to 6%, harmaline up to 4%, plus DMT |access-date=2008-04-18
- Diplopterys cabrerana, beta-carbolines
- Cabi pratensis, beta-carbolines
- Callaeum antifebrile(syn. Cabi paraensis), harmine
- Tetrapterys methystica
- Tetrapterys mucronata
[[Myristicaceae]]
- Gymnacranthera paniculata, beta-carbolines
- Horsfieldia superba, beta-carbolines
- Virola cuspidata, 6-methoxyharman
- Virola rufula, beta-carbolines
- Virola theiodora, beta-carbolines
[[Ochnaceae]]
- Testulea gabonensis, beta-carbolines
[[Palmae]]
- Plectocomiopsis geminiflora, beta-carbolines
[[Papaveraceae]]
- Meconopsis horridula, beta-carbolines
- Meconopsis napaulensis, beta-carbolines
- Meconopsis paniculata, beta-carbolines
- Meconopsis robusta, beta-carbolines
- Meconopsis rudis, beta-carbolines
- Papaver rhoeas, beta-carbolines
- Papaver Bracteatum ~ tefamine
- Papaver Paeoniflorum ~ morphine
- Papaver Setigerum ~ morphine
- Papaver somniferum ~ morphine, codeine, thebaine, papaverine, noscapine, narcotine, narceine
[[Passifloraceae]]

- Passiflora actinia, harman
- Passiflora alata, harman
- Passiflora alba, harman
- Passiflora bryonoides, harman
- Passiflora caerulea, harman
- Passiflora capsularis, harman
- Passiflora decaisneana, harman
- Passiflora edulis, harman, 0–7001 ppm in fruit
- Passiflora eichleriana, harman
- Passiflora foetida, harman
- Passiflora incarnata (with bee), harmine, harmaline, harman, etc. 0.03%. Alkaloids in rind of fruit 0.25%
- Passiflora quadrangularis, harman
- Passiflora ruberosa, harman
- Passiflora subpeltata, harman
- Passiflora warmingii, harman
[[Polygonaceae]]
- Calligonum minimum, beta-carbolines
- Leptactinia densiflora, tetrahydroharmine, etc.
- Ophiorrhiza japonica, harman
- Pauridiantha callicarpoides, harman
- Pauridiantha dewevrei, harman
- Pauridiantha lyalli, harman
- Pauridiantha viridiflora, harman
- Simira klugei, harman
- Simira rubra, harman
[[Rubiaceae]]
- Borreria verticillata, beta-carbolines
- Leptactinia densiflora, beta-carbolines
- Nauclea diderrichii, beta-carbolines
- Ophiorrhiza japonica, beta-carbolines
- Pauridiantha callicarpoides, beta-carbolines
- Pauridiantha dewevrei, beta-carbolines
- Pauridiantha yalli, neta-carbolines
- Pauridiantha viridiflora, Beta-carbolines
- Pavetta lanceolata, beta-carbolines
- Psychotria carthagenensis, beta-carbolines
- Psychotria viridis, beta-carbolines
- Simira klugei, beta-carbolines
- Simira rubra, beta-carbolines
- Uncaria attenuata, beta-carbolines
- Uncaria canescens, beta-carbolines
- Uncaria orientalis, beta-carbolines
[[Rutaceae]]
- Tetradium (syn. Evodia) species: some contain carbolines
- Euodia leptococca beta-carboline
- Araliopsis tabouensis, beta-carbolines
- Flindersia laevicarpa, beta-carbolines
- Xanthoxylum rhetsa, beta-carbolines
[[Sapotaceae]]
- Chrysophyllum lacourtianum, norharman etc.
- Scutellaria nana
[[Simaroubaceae]]
- Ailanthus malabarica, beta-carbolines. (See also Nag Champa)
- Perriera madagascariensis, beta-carbolines
- Picrasma ailanthoides, beta-carbolines
- Picrasma crenata, beta-carbolines
- Picrasma excelsa, beta-carbolines
- Picrasma javanica, beta-carbolines
[[Solanaceae]]
- Vestia foetida, (Syn V. lycioides) beta-carbolines
[[Symplocaceae]]
- Symplocos racemosa, harman
[[Tiliaceae]]
- Grewia mollis, beta-carbolines
[[Zygophyllaceae]]
- Fagonia cretica, harman
- Nitraria schoberi, beta-carbolines
- Peganum harmala, (Syrian Rue), The seeds contain about 2–6% alkaloids, most of which is harmaline. Peganum harmala is also an abortifacient.
- Peganum nigellastrum, harmine
- Tribulus terrestris, harmine etc.; Harman
- Zygophyllum fabago, harmine etc.; Harman
Opiates
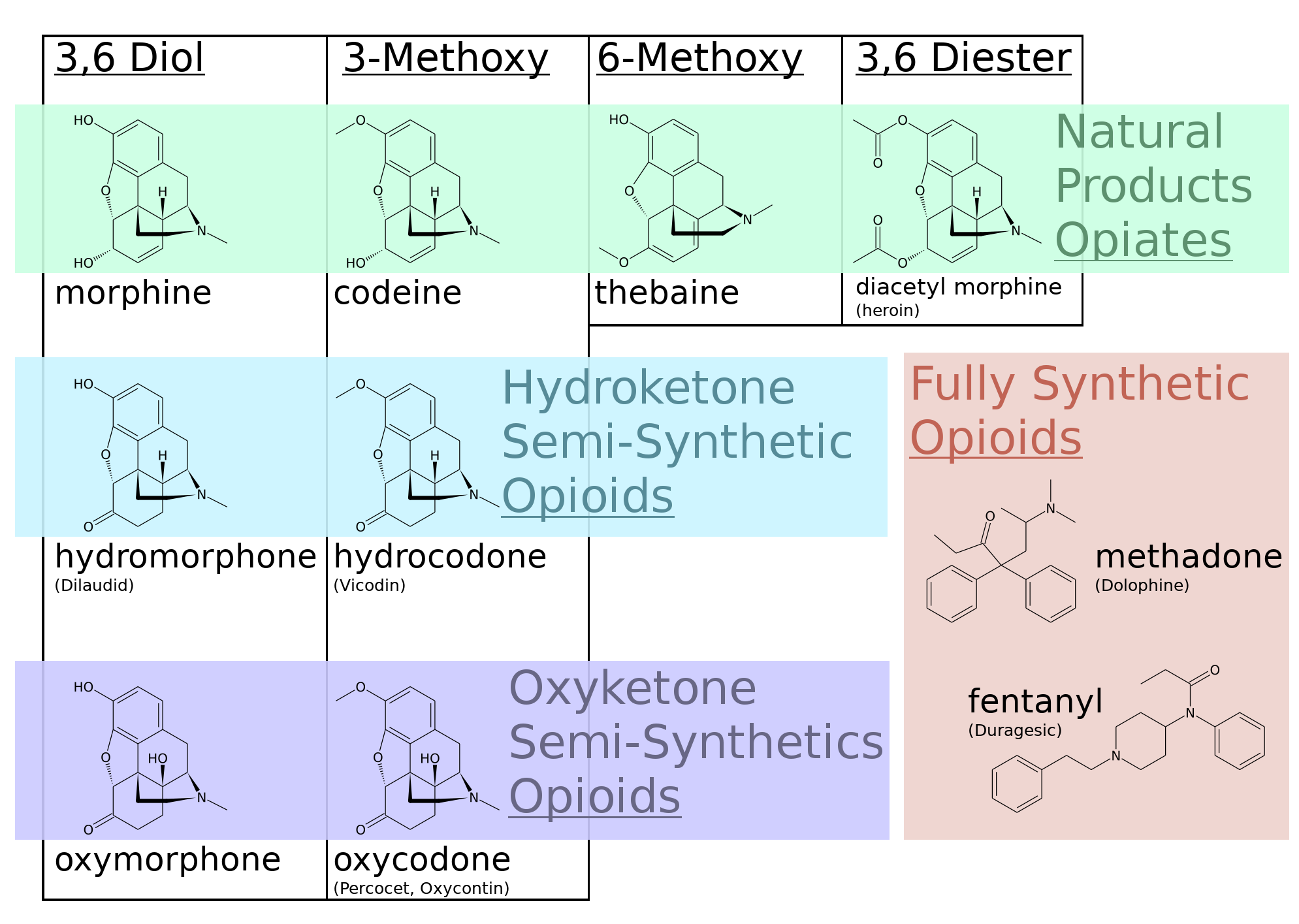
Opiates are the natural products of many plants, the most famous and historically relevant of which is Papaver somniferum. Opiates are defined as natural products (or their esters and salts that revert to the natural product in the human body), whereas opioids are defined as semi-synthetic or fully synthetic compounds that trigger the Opioid receptor of the mu sub-type. Other opiate receptors, such as kappa- and delta-opiate receptors are part of this system but do not cause the characteristic behavioral depression and analgesia which is mostly mediated through the mu-opiate receptor.
An opiate, in classical pharmacology, is a substance derived from opium. In more modern usage, the term opioid is used to designate all substances, both natural and synthetic, that bind to opioid receptors in the brain (including antagonists). Opiates are alkaloid compounds naturally found in the Papaver somniferum plant (opium poppy). The psychoactive compounds found in the opium plant include morphine, codeine, and thebaine. Opiates have long been used for a variety of medical conditions with evidence of opiate trade and use for pain relief as early as the eighth century AD. Opiates are considered drugs with moderate to high abuse potential and are listed on various "Substance-Control Schedules" under the Uniform Controlled Substances Act of the United States of America.
In 2014, between 13 and 20 million people used opiates recreationally (0.3% to 0.4% of the global population between the ages of 15 and 65). According to the CDC, from this population, there were 47,000 deaths, with a total of 500,000 deaths from 2000 to 2014. In 2016, the World Health Organization reported that 27 million people suffer from Opioid use disorder. They also reported that in 2015, 450,000 people died as a result of drug use, with between a third and a half of that number being attributed to opioids.

[[Papaver somniferum]]
The plant contains a latex that thickens into opium when it is dried. Opium contains approximately 40 alkaloids, which are summarized as opium alkaloids. The main psychoactive alkaloids are:
- Morphine: 3 to 20% in opium
- Codeine 0.1 to 4% in opium
- Thebaine 0.1 to 4% in opium
- Noscapine 1 to 11% in opium
- Oripavine
[[Atherospermataceae]]
Laurelia novae-zelandiae ~ pukateine
- Cnidium officinale
[[Mitragyna speciosa]]
- Mitragynine: Approx. 0.33% in dried leaves
- 7-Hydroxymitragynine
[[Picralima nitida]]
- Akuammine
- Akuammicine
- Pericine It may also have convulsant effects.
- Akuammiline
- Akuammidine
- N-Formylakuammiline
- Picraline
- Picraphylline
- Dihydroakuammine & Other alkaloids
[[Psychotria colorata]]
- Hodgkinsine
[[Aspidosperma|Aspidosperma spp.]]
- Akuammicine
Plants containing other psychoactive substances
+---------------------------------------------------------------------------------------------- The third column of this table has info of various types: plant common name, part of plant containing the substance, the substance, general comment ... this is sloppy; entries should be in parallel structure +----------------------------------------------------------------------------------------------
| Substance(s) | Plant | Comments | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [[File:Alpha-Asaron.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Acorus calamus1.jpg | 150px]] | |||||||||||||||||||||||||||||
| Toxic. | ||||||||||||||||||||||||||||||
| [[File:Yohimbine structure.svg | 150px]] | |||||||||||||||||||||||||||||
| *Alchornea floribunda* | α2-adrenergic receptor antagonist. | |||||||||||||||||||||||||||||
| [[File:Arecoline.svg | 150px]] [[File:Arecaidine.svg | 150px]] | ||||||||||||||||||||||||||||
| [[File:Beetle palm with nut bunch.jpg | 150px]] | |||||||||||||||||||||||||||||
| GABA uptake inhibitor, stimulant. | ||||||||||||||||||||||||||||||
| [[File:Protopine structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Stachelmohn.JPG | 150px]] | Used by Chinese residents of Mexico during the early 20th century as a legal substitute for opium and currently smoked as a marijuana substitute. | ||||||||||||||||||||||||||||
| [[File:Ergine.png | 150px]] | |||||||||||||||||||||||||||||
| [[File:Starr 050107-2974 Argyreia nervosa.jpg | 150px]] | last1=Chen | first1=W. | last2=De Wit-Bos | first2=L. | title=Risk assessment of Argyreia nervosa | date=2020 | doi=10.21945/rivm-2019-0210}} | ||||||||||||||||||||||
| [[File:Α-thujone.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Artemisia absinthium P1210748.jpg | 150px]] | |||||||||||||||||||||||||||||
| Also called "wormwood". GABA receptor antagonist. | ||||||||||||||||||||||||||||||
| Quinoline & Aporphine alkaloids | [[File:Asimina triloba3.jpg | 150px]] | ||||||||||||||||||||||||||||
| Unknown | ||||||||||||||||||||||||||||||
| [[File:L-Scopolamin.svg | 150px]] [[File:Atropine.svg | 150px]] [[File:Hyoscyamine.svg | 150px]] | |||||||||||||||||||||||||||
| [[File:Atropa belladonna - Köhler–s Medizinal-Pflanzen-018.jpg | 150px]] | |||||||||||||||||||||||||||||
| last=Kennedy | first=David O. | year=2014 | chapter=The Deliriants – The Nightshade (*Solanaceae*) Family | chapter-url=https://books.google.com/books?id=YUNDAgAAQBAJ&pg=PA131 | title=Plants and the Human Brain | location=New York | publisher=Oxford University Press | pages=131–137 | isbn=978-0-19-991401-2 | lccn=2013031617}} | ||||||||||||||||||||
| [[File:L-Scopolamin.svg | 150px]] [[File:Atropine.svg | 150px]] [[File:Hyoscyamine.svg | 150px]] | |||||||||||||||||||||||||||
| [[File:Brugmansia.jpg | 150px]] | |||||||||||||||||||||||||||||
| Commonly known as 'angel's trumpets'. An anticholinergic deliriant. | ||||||||||||||||||||||||||||||
| [[File:Harmine structure.svg]] [[File:Brunfelsamidine.svg]] [[File:L-Scopolamin.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Brunfelsia pauciflora ( Cham. & Schltdl. ) Benth. flower close-up.jpg | 150px]] | Known to cause delirium, sustained mental confusion, and possible blindness. | ||||||||||||||||||||||||||||
| Unknown | [[File:Calea zacatechichi cutting.jpg | 150px]] | Produces vivid dreams after smoking. It is also employed by the Chontal people as a medicinal herb against gastrointestinal disorders, and is used as an appetizer, cathartic anti-dysentery remedy, and as a fever-reducing agent. Its psychedelic properties do not become apparent until the user is asleep. Reports describe rituals that involve drinking it as a tea to induce divinatory or lucid dreams due to its properties as an oneirogen. | |||||||||||||||||||||||||||
| [[File:Caffeine structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Csinensis.jpg | 150px]] | |||||||||||||||||||||||||||||
| Tea leaves, tea, native to Asia. | ||||||||||||||||||||||||||||||
| [[File:S-Cathinone.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Catha edulis.jpg | 150px]] | Khat, commonly chewed, produces a stimulant effect. | ||||||||||||||||||||||||||||
| [[File:Vincristine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Catharanthus roseus24 08 2012 (1).JPG | 150px]] | |||||||||||||||||||||||||||||
| Catharanthus roseus is (perhaps unpleasantly) "hallucinogenic." | ||||||||||||||||||||||||||||||
| Unknown | [[File:Cestrum nocturnum (2464189820).jpg | 150px]] | ||||||||||||||||||||||||||||
| Commonly referred to as 'night-blooming jasmine', 'lady of the night', and 'poisonberry'. It has an unknown mechanism of action. | ||||||||||||||||||||||||||||||
| [[File:Caffeine structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Starr 070308-5472 Coffea arabica.jpg | 150px]] | |||||||||||||||||||||||||||||
| Coffee beans, coffee, native to Africa. | ||||||||||||||||||||||||||||||
| [[File:Caffeine structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Cola acuminata - Köhler–s Medizinal-Pflanzen-190.jpg | 150px]] | |||||||||||||||||||||||||||||
| Cola or kola nut, traditional additive to cola, native to Africa. | ||||||||||||||||||||||||||||||
| Salviorin A | [[File:Coleus-1.jpg | 150px]] | Trace amounts of Salviorin A have been discovered in a specific variety: Electric Lime | |||||||||||||||||||||||||||
| [[File:Bulbocapnine skeletal.svg | 150px]] | [[File:Corydalis ambigua.jpg | 150px]] | Bulbocapnine, Nantenine, Tetrahydropalmatine | ||||||||||||||||||||||||||
| [[File:L-Scopolamin.svg | 150px]] [[File:Atropine.svg | 150px]] | ||||||||||||||||||||||||||||
| [[File:Sacred datura (Datura wrightii) (14212557338).jpg | 150px]] | title=Atropine | url=https://www.drugs.com/monograph/atropine.html | publisher=The American Society of Health-System Pharmacists | access-date=Aug 13, 2015 | url-status=live | archive-url=https://web.archive.org/web/20150712191611/http://www.drugs.com/monograph/atropine.html | archive-date=2015-07-12}} which produce hallucinogenic and deliriant effects. It has an extensive history of being used recreationally. | ||||||||||||||||||||||
| [[File:(-)-Cytisine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Calia secundiflora flowers.jpg | 150px]] | |||||||||||||||||||||||||||||
| last1=Dallanoce | first1=Clelia | last2=Frigerio | first2=Fabio | last3=Martelli | first3=Giuliana | last4=Grazioso | first4=Giovanni | last5=Matera | first5=Carlo | last6=Pomè | first6=Diego Yuri | last7=Pucci | first7=Luca | last8=Clementi | first8=Francesco | last9=Gotti | first9=Cecilia | last10=Amici | first10=Marco De | display-authors=6 | title=Novel tricyclic Δ2-isoxazoline and 3-oxo-2-methyl-isoxazolidine derivatives: Synthesis and binding affinity at neuronal nicotinic acetylcholine receptor subtypes | journal=Bioorganic & Medicinal Chemistry | volume=18 | issue=12 | year=2010 | pages=4498–4508 | issn=0968-0896 | doi=10.1016/j.bmc.2010.04.065 | pmid=20478710 | name-list-style=vanc }} |
| Unknown | [[File:Desfontainia spinosa.jpg | 150px]] | ||||||||||||||||||||||||||||
| Causes visions. | ||||||||||||||||||||||||||||||
| [[File:Nicotine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Duboisia hopwoodii.jpg | 150px]] | |||||||||||||||||||||||||||||
| Pituri | ||||||||||||||||||||||||||||||
| Unknown | [[File:Entada rheedii05.jpg | 150px]] | ||||||||||||||||||||||||||||
| African dream herb. | ||||||||||||||||||||||||||||||
| [[File:Ephedrine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Ephedra sinica alexlomas.jpg | 150px]] | |||||||||||||||||||||||||||||
| Ephedra | ||||||||||||||||||||||||||||||
| [[File:Cocaine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Erythroxylum coca - Köhler–s Medizinal-Pflanzen-204.jpg | 150px]] | |||||||||||||||||||||||||||||
| Coca. Widely used illegal stimulant, produces hallucination in overdose, native to South America. | ||||||||||||||||||||||||||||||
| Unknown | [[File:Colpfl25.jpg | 150px]] | ||||||||||||||||||||||||||||
| Nerve or mosaic plant, said to produce vision of eyeballs | ||||||||||||||||||||||||||||||
| [[File:Himbacine.svg | 150px]] | |||||||||||||||||||||||||||||
| *Galbulimima belgraveana* | *Galbulimima belgraveana* is rich in alkaloids and twenty-eight alkaloids have been isolated including himbacine. | |||||||||||||||||||||||||||||
| [[File:Glaucine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Glaucium flavum03.jpg | 150px]] | Hallucinogenic effects. | ||||||||||||||||||||||||||||
| [[File:Cryogenine.svg | 150px]] | |||||||||||||||||||||||||||||
| *Heimia myrtifolia* | Auditory | |||||||||||||||||||||||||||||
| [[File:Cryogenine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Heimia salicifolia flowers by Jules Jardinier.jpg | 150px]] | |||||||||||||||||||||||||||||
| url=http://www.erowid.org/plants/sinicuichi/sinicuichi_faq.shtml | title=Erowid Sinicuichi Vault: FAQ (heimia salicifolia Frequently Asked Questions) | publisher=Erowid.org | access-date=14 January 2015}} | |||||||||||||||||||||||||||
| [[File:Lobeline structure.svg | 150px]] [[File:Nicotine.svg | 150px]] | ||||||||||||||||||||||||||||
| [[File:Hippobroma longiflora Belize 2018 2.jpeg | 150px]] | |||||||||||||||||||||||||||||
| Star of Bethlehem | ||||||||||||||||||||||||||||||
| [[File:Hyperforin2DACS.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Saint John's wort flowers.jpg | 150px]] | |||||||||||||||||||||||||||||
| Saint John's wort | ||||||||||||||||||||||||||||||
| Tropane alkaloids | [[File:Henbane1.JPG | 150px]] | ||||||||||||||||||||||||||||
| Henbane | ||||||||||||||||||||||||||||||
| [[File:Caffeine structure.svg | 150px]] [[File:Theobromine.svg | 150px]] | ||||||||||||||||||||||||||||
| [[File:View of Ilex guayusa from above.jpg | 150px]] | |||||||||||||||||||||||||||||
| *Ilex guayusa* is used as an additive to some versions of Ayahuasca. According to the Ecuadorian indigenous, it is also slightly hallucinogenic on its own, when drunk in high enough quantities. | ||||||||||||||||||||||||||||||
| [[File:Ergine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Ipomoea violacea.jpg | 150px]] | |||||||||||||||||||||||||||||
| Ergine and isoergine in seeds; up to 0.12% ergine total | ||||||||||||||||||||||||||||||
| Lactucarium | [[File:Lactuca virosa - Köhler–s Medizinal-Pflanzen-213.jpg | 150px]] | Lactucarium | |||||||||||||||||||||||||||
| [[File:Lagochilin.png | 150px]] | [[File:Lagochilus inebrians.jpg | 150px]] | |||||||||||||||||||||||||||
| Lagochilin is thought to be responsible for the sedative, hypotensive and hemostatic effects of this plant. | ||||||||||||||||||||||||||||||
| [[File:Pukateine Structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Old Rimu in Kaitoke Park.jpg | 150px]] | Pukateine | ||||||||||||||||||||||||||||
| [[File:RolliniaDeliciosa.jpg | 150px]] | |||||||||||||||||||||||||||||
| Corexamine inhibits the enzyme dopamine β-hydroxylase, which converts dopamine to norepinephrine.Reticuline acts as a central nervous system depressant in rats and mice. | ||||||||||||||||||||||||||||||
| [[File:Leonurine structure.png | 150px]] | |||||||||||||||||||||||||||||
| [[File:Leonotis leonurus flower.jpg | 150px]] | Both leaves and flowers (where most concentrated) contain Leonurine. (Effects reminiscent of marijuana) | ||||||||||||||||||||||||||||
| [[File:Nicotine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Leucas aspera plant.jpg | 150px]] | |||||||||||||||||||||||||||||
| *Leucas aspera* | Nicotine | |||||||||||||||||||||||||||||
| [[File:Leonurine structure.png | 150px]] | |||||||||||||||||||||||||||||
| [[File:Leonotis nepetifolia1.jpg | 150px]] | Both leaves and flowers (where most concentrated) contain Leonurine and several compounds. (Effects reminiscent of marijuana) | ||||||||||||||||||||||||||||
| [[File:Lobeline.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Lobelia inflata - Köhler–s Medizinal-Pflanzen-218.jpg | 150px]] | |||||||||||||||||||||||||||||
| Indian tobacco | ||||||||||||||||||||||||||||||
| Unknown | [[File:Sweetbay Magnolia Magnolia virginiana Comparison 4400px.jpg | 150px]] | url=https://books.google.com/books?id=Rs5rAwAAQBAJ&q=Magnolia+virginiana+psychoactive&pg=PT2377 | title=The Encyclopedia of Psychoactive Plants: Ethnopharmacology and Its Applications | first=Christian | last=Rätsch | date=25 April 2005 | publisher=Inner Traditions/Bear | access-date=22 December 2017 | via=Google Books | isbn=978-1-59477-662-5}} | |||||||||||||||||||
| [[File:L-Scopolamin.svg | 150px]] [[File:Atropine.svg | 150px]] [[File:Hyoscyamine.svg | 150px]] | |||||||||||||||||||||||||||
| [[File:Mandragora officinarum 002.JPG | 150px]] | |||||||||||||||||||||||||||||
| Mandrake has deliriant and anticholinergic properties. | ||||||||||||||||||||||||||||||
| [[File:Ergine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:2006-10-18Mirabilis jalapa10.jpg | 150px]] | |||||||||||||||||||||||||||||
| Possibly contains ergine, a hallucinogen. | ||||||||||||||||||||||||||||||
| [[File:Mitragynine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Mitragyna speciosa111.JPG | 150px]] | |||||||||||||||||||||||||||||
| Usually referred to as kratom. Has opioid-like and stimulant properties. | ||||||||||||||||||||||||||||||
| [[File:Myristicin.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Myristica fragrans - Köhler–s Medizinal-Pflanzen-097.jpg | 150px]] | |||||||||||||||||||||||||||||
| Nutmeg | ||||||||||||||||||||||||||||||
| [[File:Aporphine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Sacred lotus Nelumbo nucifera.jpg | 150px]] | |||||||||||||||||||||||||||||
| Sacred lotus | ||||||||||||||||||||||||||||||
| [[File:Nepetalactone.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Catnip flowers.jpg | 150px]] | |||||||||||||||||||||||||||||
| Catnip | ||||||||||||||||||||||||||||||
| [[File:Nicotine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Tabak P9290021.JPG | 150px]] | |||||||||||||||||||||||||||||
| Tobacco. Can cause hallucinations in very large doses. | ||||||||||||||||||||||||||||||
| [[File:Aporphine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Nymphaea caerulea.jpg | 150px]] | Blue lotus or lily. Recent studies have shown *Nymphaea caerulea* to have psychedelic properties, and may have been used as a sacrament in ancient Egypt and certain ancient South American cultures. Dosages of 5 to 10 grams of the flowers induces slight stimulation, a shift in thought processes, enhanced visual perception, and mild closed-eye visuals. *Nymphaea caerulea* is unrelated to *Nelumbo nucifera* the Sacred Lotus, with Nymphaea in the Nymphales, one of the oldest and most basal linegages of flowering plants and with Nelumbo in Proteales one of the core eudicots. Their morphological similarities being entirely convergent evolution, however they apparently have convergently evolved similar biochemistry. Both *Nymphaea caerulea* and *Nelumbo nucifera* contain the alkaloids nuciferine and apomorphine, which have been recently isolated by independent labs. | ||||||||||||||||||||||||||||
| [[File:Ginsenoside Rg1.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Panax quinquefolius.jpg | 150px]] | |||||||||||||||||||||||||||||
| Ginseng | ||||||||||||||||||||||||||||||
| [[File:Morphin - Morphine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Papaver somniferum - Köhler–s Medizinal-Pflanzen-102.jpg | 150px]] | |||||||||||||||||||||||||||||
| url=https://www.drugs.com/dict/opium.html | title=Opium definition | publisher=Drugs.com | access-date=28 April 2022}} | |||||||||||||||||||||||||||
| Unknown | [[File:Pokeweed bush in Northumberland County, Pennsylvania.JPG | 150px]] | ||||||||||||||||||||||||||||
| url=https://www.academia.edu/1103613 | title=Psychoactive Plants Described in a Brazilian Literary Work and their Chemical Compounds | first1=Rafaela | last1=Denise Otsuka | first2=Rafaela Denise | last2=Otsuka | first3=Joao Henrique Ghilardi | last3=Lago | first4=Lucia | last4=Rossi | first5=Jose Carlos Fernandes | last5=Galduroz | first6=Eliana | last6=Rodrigues | journal=Central Nervous System Agents in Medicinal Chemistry | year=2010 | volume=10 | issue=3 | pages=218–237 | doi=10.2174/1871524911006030218 | pmid=20557283 | via=www.academia.edu}} | |||||||||
| [[File:Yohimbine structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Pau de cabinda.jpg | 150px]] | |||||||||||||||||||||||||||||
| α2-adrenergic receptor antagonist. | ||||||||||||||||||||||||||||||
| Unknown | [[File:Pedicularis densiflora mt. diablo.JPG | 150px]] | ||||||||||||||||||||||||||||
| Indian warrior | ||||||||||||||||||||||||||||||
| [[File:Kavalactone-general-numbered.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Starr 040318-0058 Piper methysticum.jpg | 150px]] | author=Pittler MH, Ernst E | title=Kava extract for treating anxiety | journal=Cochrane Database of Systematic Reviews | issue=1 | article-number=CD003383 | year=2003 | volume=2003 | pmid=12535473 | doi=10.1002/14651858.CD003383 | url=http://www.cochrane.org/CD003383/DEPRESSN_kava-extract-for-treating-anxiety | editor1-last=Pittler | editor1-first=Max H | pmc=6999799 }} and hypnotic. Often advertised as a 'healthier' alternative to alcohol. | ||||||||||||||||
| [[File:Ergine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Rivea corymbosa 1838.jpg | 150px]] | |||||||||||||||||||||||||||||
| Seeds contain ergine, isoergine, lysergol, and turbicoryn; lysergic acid alkaloids up to 0.03% | ||||||||||||||||||||||||||||||
| [[File:Salvinorin A structure.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Salvia divinorum - Herba de Maria.jpg | 150px]] | Salvinorin A, 0.89–3.87 mg/g, also Salvinorin B and Salvinorin C | ||||||||||||||||||||||||||||
| [[File:Mesembrine.svg | 150px]] | |||||||||||||||||||||||||||||
| Mesembrine | [[File:Sceletium tortuosum 01102003 Afrique du sud 2.JPG | 150px]] | ||||||||||||||||||||||||||||
| Kanna | ||||||||||||||||||||||||||||||
| [[File:Baicalein.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Scutellaria pekinensis Yamatatsunamisou in Ibukiyama 2002-6-9.jpg | 150px]] | |||||||||||||||||||||||||||||
| Known commonly as 'skullcaps'. Baicalein is a positive allosteric modulator of GABAA receptor. | ||||||||||||||||||||||||||||||
| Unknown | [[File:Sessea brasiliensis.jpg | 150px]] | S. brasiliensis poisoning is described as very similar to that of Cestrum laevigatum; a species used to induce hallucinations by the Krahô tribe for spiritual purposes. | |||||||||||||||||||||||||||
| Unknown | [[File:Silene capensis (flowering).jpg | 150px]] | author=J. F. Sobiecki | date=2008 | title=A review of plants used in divination in southern Africa and their psychoactive effects | journal=Southern African Humanities | volume=20 | pages=333–351 | s2cid=37305695}} | |||||||||||||||||||||
| Unknown | [[File:Tagetes lemmonii flower.jpg | 150px]] | Anethole, Chavicol, Coumarin, Estragole, Isorhamnetin, Methyleugenol, Quercitin | |||||||||||||||||||||||||||
| [[File:Ibogaine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Iboga.jpg | 150px]] | url=http://www.erowid.org/library/books_online/tihkal/tihkal25.shtml | title=Erowid Online Books: "TIHKAL" – #25 IBOGAINE | publisher=Erowid.org | access-date=14 January 2015}} | |||||||||||||||||||||||||
| [[File:Ibogaine.svg | 150px]] | |||||||||||||||||||||||||||||
| Ibogaine in root leaves. Produces psychedelic and a dissociative effects. | ||||||||||||||||||||||||||||||
| [[File:Voacangine.svg | 150px]] [[File:Ibogaine.svg | 150px]] | ||||||||||||||||||||||||||||
| [[File:Crape_Jasmine.jpg | 150px]] | vauthors=Krengel F, Herrera Santoyo J, Olivera Flores TJ, Chávez Ávila VM, Pérez Flores FJ, Reyes Chilpa R | title=Quantification of Anti-Addictive Alkaloids Ibogaine and Voacangine in In Vivo- and In Vitro-Grown Plants of Two Mexican Tabernaemontana Species | journal=Chemistry & Biodiversity | volume=13 | issue=12 | pages=1730–1737 | date=December 2016 | pmid=27448833 | doi=10.1002/cbdv.201600146 | s2cid=46046257 | url= | issn=}} | |||||||||||||||||
| [[File:Ibogaine.svg | 150px]] | |||||||||||||||||||||||||||||
| Is a psychedelic and a dissociative. Contains ibogaine and similar alkaloids. | ||||||||||||||||||||||||||||||
| [[File:Ibogaine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Tabernaemontana divaricata by kadavoor.jpg | 150px]] | |||||||||||||||||||||||||||||
| Is a psychedelic and a dissociative. | ||||||||||||||||||||||||||||||
| [[File:Theobromine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Matadecacao.jpg | 150px]] | |||||||||||||||||||||||||||||
| Cocoa or cacao bean, chocolate, native to the Americas | ||||||||||||||||||||||||||||||
| [[File:Ibogaine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Confederate Jasmine, Star Jasmine (Trachelospermum jasminoides).jpg | 150px]] | Exhibits psychedelic and dissociative effects. Contains ibogaine, coronaridine, voacangine, apparicine, conoflorine, and [19-epi-voacangarine](19-epi-voacangarine). | ||||||||||||||||||||||||||||
| [[File:Valerenic acid.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Valeriana officinalis - Niitvälja.jpg | 150px]] | |||||||||||||||||||||||||||||
| Possible sedative and anxiolytic effects. Valerenic acid is GABAA receptor positive allosteric modulator, and a [5-HT5A receptor](5-ht-receptor) partial agonist. | ||||||||||||||||||||||||||||||
| [[File:Vincamine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Vinca minor Nashville.jpg | 150px]] | |||||||||||||||||||||||||||||
| Vincamine. | ||||||||||||||||||||||||||||||
| [[File:Voacangine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Voacanga Africana 06.jpg | 150px]] | |||||||||||||||||||||||||||||
| Voacangine is similar in structure to ibogaine. It inhibits AChE. | ||||||||||||||||||||||||||||||
| [[File:Dendrobine.svg | 150px]] | |||||||||||||||||||||||||||||
| [[File:Dendrobium_nobile_-_flower_view_01.jpg | 150px]] | |||||||||||||||||||||||||||||
| Also contains phenanthrenes and dendrobine related alkaloids. | ||||||||||||||||||||||||||||||
| [[File:Genistein.svg | 150px]] [[File:Apigenin.svg | 150px]] | ||||||||||||||||||||||||||||
| [[File:Zornia latifolia Sm.jpg | 150px]] | |||||||||||||||||||||||||||||
| *Zornia latifolia* is sometimes combined with synthetic cannabis. It may produce similar effects to cannabis. It is nicknamed Maconha brava because locals use it as a cannabis substitute. |
Notes
References
Bibliography
References
- Sayin, H. Umit. (2016). "Neuropathology of Drug Addictions and Substance Misuse". Elsevier.
- (2021). "Ancient psychoactive plants in a global village: The ritual use of cannabis in a self-managed community in Catalonia". Elsevier BV.
- (1959). "Indole Alkaloids of ''Acer saccharinum'' (the Silver Maple), ''Dictyoloma incanescens'', ''Piptadenia colubrina'', and ''Mimosa hostilis''". Journal of Organic Chemistry.
- "Trout's Notes on Some Other Succulents".
- (November 2018). "Isolation, spectral characterization, molecular docking, and cytotoxic activity of alkaloids from Erythroxylum pungens O. E. Shulz". [[Phytochemistry (journal).
- "Plants & Seeds > A > Acacia spp.". Shaman Australis Botanicals.
- Nutritive value assessment of the tropical shrub legume Acacia angustissima: anti-nutritional compounds and ''in vitro'' digestibility. Personal Authors: McSweeney, C. S., Krause, D. O., Palmer, B., Gough, J., Conlan, L. L., Hegarty, M. P.Author Affiliation: CSIRO Livestock Industries, Long Pocket Laboratories, 120 Meiers Road, Indooroopilly, Qld 4068, Australia. Document Title: Animal Feed Science and Technology, 2005 (Vol. 121) (No. 1/2) 175–190
- "Maya Ethnobotanicals – Ayahuasca, Rainforest Plants, Folklore, Incenses, Art & Visions".
- Black Panther. "Akacje". Herbarium.0–700.pl.
- "Lycaeum > Leda > Acacia auriculiformis". Leda.lycaeum.org.
- Australian Bush Food and Native Medicine Forum members. "Australian Bushfood (Bushtucker) and Native Medicine Forum". Bushfood.net.
- "Entheology.org – Preserving Ancient Knowledge". Entheology.org.
- "Ask Dr. Shulgin Online September 26, 2001". Cognitiveliberty.org.
- "Dr Karl's Q&A forum". Abc.net.au.
- "comp phyto". Users.lycaeum.org.
- "acacias and entheogens". Users.lycaeum.org.
- "Lycaeum > Leda > Acacia complanata". Users.lycaeum.org.
- (5 January 2010). "Naturheilpraxis – Fachforum – Die Heilkraft der Akazien – Ein einführender Überblick".
- (1996-07-30). "Caesalpinioideae und Mimosoideae". Springer.
- "Lycaeum > Leda > Acacia cultriformis". Leda.lycaeum.org.
- "Plant Choices – Phytochemeco Databases". Ars-grin.gov.
- Vivid Interactive and Design. "Wattle Seed Workshop Proceedings".
- "www.bpi.da.gov.ph".
- "Acacia farnesiana". Hort.purdue.edu.
- Voogelbreinder, S. "Garden Of Eden" 2009
- "Lista över hallucinogena växter, svampar och djur". Wiki.magiskamolekyler.org.
- (September 2017)
- S. Voogelbreinder "Garden Of Eden" 2009
- "Lista över hallucinogena växter, svampar och djur – Magiska Molekylers Wiki".
- "obtusifolia phyto". Users.lycaeum.org.
- [http://www.drogen-forum.com/wiki/index.php/DMT-haltige_Pflanzen Plants Containing DMT (German)] {{webarchive. link. (2007-06-29)
- (January 2014). "Determination of selected biogenic amines in Acacia rigidula plant materials and dietary supplements using LC-MS/MS methods.". Journal of Pharmaceutical and Biomedical Analysis.
- "Chemistry of Acacias from South Texas".
- "Eins". Factorey.ch.
- (January 1, 1965). "Native hallucinogenic drugs piptadenias". [[United Nations Office on Drugs and Crime]].
- link. (2004-11-10 Phytochemical and Ethnobotanical Databases)
- "Cultivo de Curupay, Cebil colorado (Anadenanthera colubrina) y usos, herbotecnia". Herbotecnia.com.ar.
- link. (2013-02-19 Phytochemical and Ethnobotanical Databases)
- (2013-02-18). "Psychedelics Encyclopedia". Ronin.
- Ott, J.. (July–September 2001). "Pharmañopo-psychonautics: human intranasal, sublingual, intrarectal, pulmonary and oral pharmacology of bufotenine". Journal of Psychoactive Drugs.
- "Erowid Online Books: "Ayahuasca: alkaloids, plants, and analogs" by Keeper of the Trout". Erowid.org.
- {{GRIN. ''Desmodium caudatum''. 311307
- "Trout's Notes on Desmodium".
- (1985). "The alkaloids. Chemistry and pharmacology. Volume 26". Academic Press.
- Enzyklopädie der psychoaktiven Pflanzen. Botanik, Ethnopharmakologie und Anwendungen. Aarau: AT-Verl. p. 15. ISBN 978-3-85502-570-1.
- (2004). "Medical Toxicology - Google Book Search". Lippincott Williams & Wilkins.
- "tryptamines: fungi". Bluezoo.org.
- (September 2017)
- (2018-09-01). "Zornia latifolia: a smart drug being adulterated by Stylosanthes guianensis". International Journal of Legal Medicine.
- (2022). "Cannabis-like activity of Zornia latifolia Sm. detected in vitro on rat cortical neurons: major role of the flavone syzalterin". Drug and Chemical Toxicology.
- (January 2018). "Plants Containing DMT List". Dmt-nexus.com.
- (November 1991). "Horsfiline, an oxindole alkaloid from Horsfieldia superba". The Journal of Organic Chemistry.
- "5-MeO-DMT". Tryptamines.com.
- "Committee for veterinary medicinal products virola sebifera summary report".
- "Erowid Arundo donax Vaults: Trout's Notes on Tryptamine Content of Arundo donax". Erowid.org.
- "Erowid Phalaris Vault: FAQ 2.01". Erowid.org.
- "DMT, Life and the Universe". Nepenthes.lycaeum.org.
- (1999). "Assessment of potential for toxicity of Phalaris spp. via alkaloid content determination: P. coerulescens, a case example". Phytochemical Analysis.
- (1985). "Alkaloids from the Rhizomes of Phragmites australis Cav.". Scientia Pharmaceutica.
- (1972). ""Ayahuasca," the South American Hallucinogenic Drink: An Ethnobotanical and Chemical Investigation". Economic Botany.
- "Psychotria – The Most Important Genera and Species from A to Z – The Encyclopedia of Psychoactive Plants: Ethnopharmacology and Its Applications".
- "Psychotria poeppigiana – Uragoga tomentosa". Discover Life.
- "Amazing Nature". Amazing-nature.com.
- (Sep 2012). "N-methylated tryptamine derivatives in citrus genus plants: identification of N,N,N-trimethyltryptamine in bergamot". Journal of Agricultural and Food Chemistry.
- (May 2013). "Citrus genus plants contain N-methylated tryptamine derivatives and their 5-hydroxylated forms". Journal of Agricultural and Food Chemistry.
- "Erowid Psychoactive Vaults: Tryptamine FAQ". Erowid.org.
- (Jun 2004). "Pilocarpus spp.: A survey of its chemical constituents and biologicalactivities". Brazilian Journal of Pharmaceutical Sciences.
- "Citrus Growers Manufacture Huge Amounts of DMT".
- "Citrus Genus Plants Contain N-Methylated Tryptamine Derivatives and Their 5-Hydroxylated Forms".
- "CitrusGenus Plants Contain N‑Methylated Tryptamine Derivativesand Their 5‑Hydroxylated Forms".
- (1983). "Cactus Alkaloids. LIII. Coryphanthine and O-Methyl-Candicine, Two New Quaternary Alkaloids from Coryphantha greenwoodii". Journal of Natural Products.
- (1 July 2004). "Cactus Alkaloids. LIII. Coryphanthine and O-Methyl-Candicine, Two New Quaternary Alkaloids from Coryphantha greenwoodii". Journal of Natural Products.
- "Cane Cholla (''Cylindropuntia spinosior'' )". Desert-tropicals.com.
- "Partial List of Alkaloids in Trichocereus Cacti". Thenook.org.
- a1b2c3.com. "Trichocereus spp. Information". A1b2c3.com.
- "Partial List of Alkaloids in Trichocereus Cacti". Thennok.org.
- "Descriptions of psychoactive Cacti.". Users.lycaeum.org.
- "Echinopsis tacaquirensis ssp. taquimbalensis". Desert-tropicals.com.
- "Mescaline in Trichocereus — The Mescaline Garden".
- "Cardon Grande (Echinopsis terscheckii)". Desert-tropicals.com.
- [http://entheogen.netfirms.com/articles/articles/Narcotic_Cacti.html Forbidden Fruit Archives] {{webarchive. link. (2005-11-28)
- "Erowid Cacti Vaults: Visionary Cactus Guide – Mescaline from Sawdust". Erowid.org.
- "Archived copy".
- (2007). "From the behavioral pharmacology of beta-carbolines to seizures, anxiety, and memory". ScientificWorldJournal.
- "Cornell University Department of Animal Science". Ansci.cornell.edu.
- (2005). "Phytochemical analyses of Banisteriopsis caapi and Psychotria viridis". Journal of Psychoactive Drugs.
- (2002-09-11). "Directory Of Plants Containing Secondary Metabolites". CRC Press.
- (2021). "Opium Consumption". International Agency for Research on Cancer.
- "Passion Flower". Drugs.com.
- "www.amazing-nature.com".
- (April 2000). "Alkaloids and phenylpropanoids from Peganum nigellastrum". [[Phytochemistry (journal).
- (March 2004). "Antinociceptive effect of 7-hydroxymitragynine in mice: Discovery of an orally active opioid analgesic from the Thai medicinal herb Mitragyna speciosa". Life Sciences.
- Alorah Hippo. (5 November 2021). "What is Akuamma and Where Does it Grow?".
- (1998). "Opioid activity of alkaloids extracted from Picralima nitida (Fam. Apocynaceae)". European Journal of Pharmacology.
- (December 1982). "Detection of pericine, a new CNS-active indole alkaloid from Picralima nitida cell suspension culture by opiate receptor binding studies". Planta Medica.
- (30 June 1998). "Alkaloids: Biochemistry, Ecology, and Medicinal Applications". Springer.
- "Alkaloids of picralima nitida used for treatment of protozoal diseases".
- (March 1998). "Pyrrolidinoindoline Alkaloids from Psychotria colorata1". Journal of Natural Products.
- (December 2000). "Antinociceptive profile of hodgkinsine". Planta Medica.
- (1996). "Alkaloids from ''Aspidosperma'' species from Bolivia". Planta Medica.
- (2013). "Transport of the areca nut alkaloid arecaidine by the human proton-coupled amino acid transporter 1 (hPAT1)". The Journal of Pharmacy and Pharmacology.
- (1975). "Betel nut constituents as inhibitors of γ-aminobutyric acid uptake". Nature.
- (2001). "Arecoline M1 receptor activation is a requirement for arecoline analgesia". Il Farmaco.
- (2020). "Risk assessment of Argyreia nervosa".
- Shulgin, Alexander T.. (1976). "Psychopharmacological Agents: Use, Misuse and Abuse". Academic Press.
- (1975). "Hallucinogenic Agents". Wright-Scientechnica.
- Halpern, J.H.. (2004). "Hallucinogens and dissociative agents naturally growing in the United States". Pharmacology & Therapeutics.
- Olsen, Richard W.. (2000-04-25). "Absinthe and γ-aminobutyric acid receptors". Proceedings of the National Academy of Sciences of the United States of America.
- Kennedy, David O.. (2014). "Plants and the Human Brain". [[Oxford University Press]].
- Cleversley, Keith. (2002-01-01). "Brunfelsia grandiflora - Manaca".
- (2016). "Neuropharmacological characterization of the oneirogenic Mexican plant Calea zacatechichi aqueous extract in mice". Metabolic Brain Disease.
- Al Zarouni, Yousif. (2015). "The Effects of Khat (Catha Edulis)". Yousif Al Zarouni.
- "Protected Blog". Sliceoftheday.
- "Best Kratom Strains".
- (2004). "Plant biochemistry: A naturally decaffeinated arabica coffee". [[Nature (journal).
- "Atropine". The American Society of Health-System Pharmacists.
- (2009). "Plant-derived Natural Products: Synthesis, Function, and Application". Springer Science & Business Media.
- (7 January 2021). "Peculiar plants and fantastic fungi: An ethnobotanical study of the use of hallucinogenic plants and mushrooms in Slovenia". PLOS ONE.
- (2010). "Novel tricyclic Δ2-isoxazoline and 3-oxo-2-methyl-isoxazolidine derivatives: Synthesis and binding affinity at neuronal nicotinic acetylcholine receptor subtypes". Bioorganic & Medicinal Chemistry.
- Schultes, Richard Evans, Iconography of New World Plant Hallucinogens. p. 101
- Rovinskiĭ VI. (Sep 1989). "A case of hallucinogen-like action of glaucine. (Russian)". Klinicheskaia Meditsina (Mosk).
- "Erowid Sinicuichi Vault: FAQ (heimia salicifolia Frequently Asked Questions)". Erowid.org.
- https://www.biosynth.com/p/FC20554/483-45-4-s-coreximine
- (1998). "Central depressant effects of reticuline extracted from Ocotea duckei in rats and mice". Journal of Ethnopharmacology.
- (2006). "Isolation and identification of nicotine from leucas aspera (willd) link". Indian Journal of Pharmaceutical Sciences.
- (2020-11-10). "Toward the identification of a phytocannabinoid-like compound in the flowers of a South African medicinal plant (Leonotis leonurus)". BMC Research Notes.
- Rätsch, Christian. (25 April 2005). "The Encyclopedia of Psychoactive Plants: Ethnopharmacology and Its Applications". Inner Traditions/Bear.
- (2020). "Kratom—Pharmacology, Clinical Implications, and Outlook: A Comprehensive Review". Pain and Therapy.
- Seligman, Sian. (2023-01-13). "Blue Lotus Flower: Smoking, Tea & More".
- "Opium definition". [[Drugs.com]].
- (2010). "Psychoactive Plants Described in a Brazilian Literary Work and their Chemical Compounds". Central Nervous System Agents in Medicinal Chemistry.
- Pittler MH, Ernst E. (2003). "Kava extract for treating anxiety". Cochrane Database of Systematic Reviews.
- Baker, Jonathan D.. (2011-06-01). "Tradition and toxicity: evidential cultures in the kava safety debate". Social Studies of Science.
- "Salvia divinorum Clones". Sagewisdom.org.
- (2016-01-11). "High-mesembrine Sceletium extract (Trimesemine™) is a monoamine releasing agent, rather than only a selective serotonin reuptake inhibitor". Journal of Ethnopharmacology.
- (2021-04-28). "A Chewable Cure "Kanna": Biological and Pharmaceutical Properties of Sceletium tortuosum". Molecules.
- (2000). "Interaction of flavones from the roots of Scutellaria baicalensis with the benzodiazepine site". Planta Med..
- (1965). "Poisoning by Sessea brasiliensis Toledo in cattle". Poisoning by Sessea Brasiliensis Toledo in Cattle.
- (2006). "Plants with possible psychoactive effects used by the Krahô Indians, Brazil". Revista Brasileira de Psiquiatria.
- J. F. Sobiecki. (2008). "A review of plants used in divination in southern Africa and their psychoactive effects". Southern African Humanities.
- "Erowid Online Books: "TIHKAL" – #25 IBOGAINE". Erowid.org.
- (December 2016). "Quantification of Anti-Addictive Alkaloids Ibogaine and Voacangine in In Vivo- and In Vitro-Grown Plants of Two Mexican Tabernaemontana Species". Chemistry & Biodiversity.
- Dr. B. Bös. "Sternjasmin (Trachelospermum jasminoides) im GIFTPFLANZEN.COMpendium - giftpflanzen.com". Giftpflanzen.com.
- "Indole Alkaloids from Trachelospermum jasminoides".
- (2015). "Identification of the putative binding pocket of valerenic acid on GABAA receptors using docking studies and site-directed mutagenesis". Br. J. Pharmacol..
- (2005). "Valerian extract and valerenic acid are partial agonists of the 5-HT receptor in vitro". Molecular Brain Research.
- (2010). "Cytotoxicity ofVinca minor". Pharmaceutical Biology.
- (2008). "Two fast screening methods (GC-MS and TLC-ChEI assay) for rapid evaluation of potential anticholinesterasic indole alkaloids in complex mixtures". Annals of the Brazilian Academy of Sciences.
- (June 2005). "Indole alkaloids from Tabernaemontana australis (Muell. Arg) Miers that inhibit acetylcholinesterase enzyme". [[Bioorganic & Medicinal Chemistry]].
- Duke, James A.. (2017-10-24). "Handbook of Phytochemical Constituent Grass, Herbs and Other Economic Plants: Herbal Reference Library". Routledge.
- (2018-09-01). "''Zornia latifolia'': a smart drug being adulterated by ''Stylosanthes guianensis''". International Journal of Legal Medicine.
- (2011). "Beyond THC: The New Generation of Cannabinoid Designer Drugs". Frontiers in Behavioral Neuroscience.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about List of psychoactive plants — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report