From Surf Wiki (app.surf) — the open knowledge base
Lindlar catalyst
Catalyst enabling the hydrogenation of alkynes to alkenes
Catalyst enabling the hydrogenation of alkynes to alkenes

A Lindlar catalyst is a heterogeneous catalyst consisting of palladium deposited on calcium carbonate (CaCO3) or barium carbonate (BaCO3) then poisoned with various forms of lead or sulfur. It is used for the hydrogenation of alkynes to alkenes (i.e. without further reduction into alkanes). It is named after its inventor Herbert Lindlar, who discovered it in 1952.
Synthesis
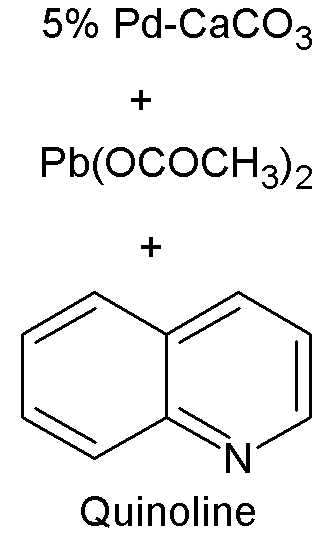
Lindlar catalyst is commercially available but can also be created by reducing palladium chloride in a slurry of calcium carbonate (CaCO3) or barium carbonate (BaCO3) and adding lead acetate. A variety of other "catalyst poisons" have been used, including lead oxide and quinoline. The palladium content of the supported catalyst is usually 5% by weight.
Catalytic properties
The catalyst is used for the hydrogenation of alkynes to alkenes (i.e. without further reduction into alkanes). The lead serves to deactivate the palladium sites, and further deactivation of the catalyst with quinoline or 3,6-dithia-1,8-octanediol enhances its selectivity, preventing formation of alkanes. Thus if a compound contains a double bond as well as a triple bond, only the triple bond is reduced. An example being the reduction of phenylacetylene to styrene.

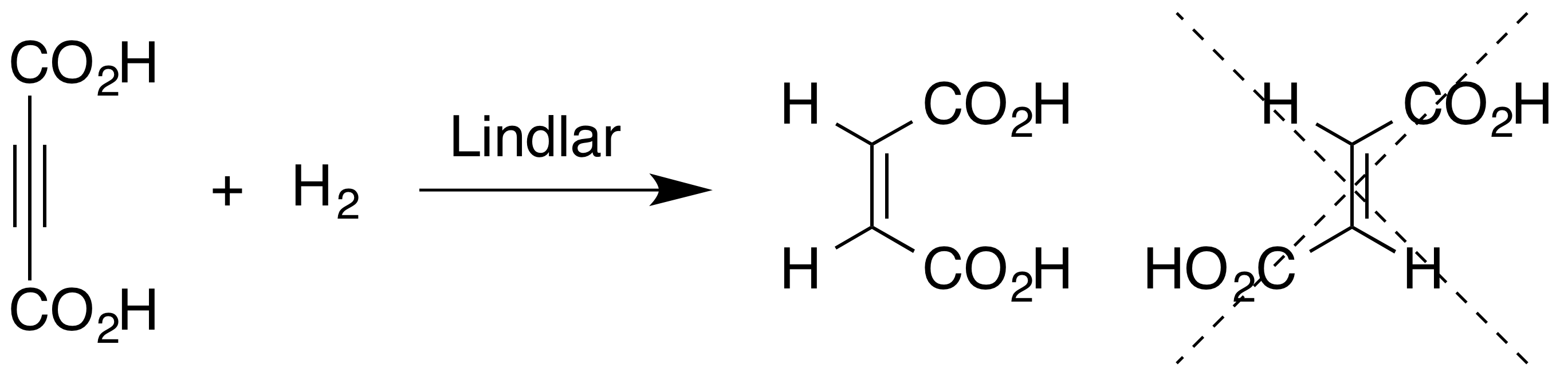
Alkyne hydrogenation is stereospecific, occurring via syn addition to give the cis-alkene. For example the hydrogenation of acetylenedicarboxylic acid using Lindlar catalyst gives maleic acid rather than fumaric acid.
An example of commercial use is the organic synthesis of vitamin A which involves an alkyne reduction with the Lindlar catalyst. These catalysts are also used in the synthesis of dihydrovitamin K1.
References
References
- McEwen, Alan B.. (2002-05-01). "Metallic palladium, the actual catalyst in Lindlar and Rosenmund reductions?".
- (1966). "Palladium Catalyst for Partial Reduction of Acetylenes".
- (1 February 1952). "Ein neuer Katalysator fur selektive Hydrierungen". Helvetica Chimica Acta.
- (1993). "Explanation of a paradoxical selectivity in Friedel-Crafts alkylation under heterogeneous catalysis". [[Pergamon Press]].
- Overman, L. E.. (1993). "(Z)-4-(Trimethylsilyl)-3-Buten-1-ol".
- Fritz Weber, August Rüttimann "Vitamin K" Ullmann's Encyclopedia Of Industrial Chemistry, 2012, Wiley-VCH, Weinheim. {{doi. 10.1002/14356007.o27_o08
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Lindlar catalyst — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report