From Surf Wiki (app.surf) — the open knowledge base
Knorr pyrrole synthesis
Chemical reaction

Chemical reaction
The Knorr pyrrole synthesis is a widely used chemical reaction that synthesizes substituted pyrroles (3). The method involves the reaction of an α-amino-ketone (1) and a compound containing an electron-withdrawing group (e.g. an ester as shown) α to a carbonyl group (2).

Method
The mechanism requires zinc and acetic acid as catalysts. It will proceed at room temperature. Because α-aminoketones self-condense very easily, they must be prepared in situ. The usual way of doing this is from the relevant oxime, via the Neber rearrangement.
The original Knorr synthesis employed two equivalents of ethyl acetoacetate, one of which was converted to ethyl 2-oximinoacetoacetate by dissolving it in glacial acetic acid, and slowly adding one equivalent of saturated aqueous sodium nitrite, under external cooling. Zinc dust was then stirred in, reducing the oxime group to the amine. This reduction consumes two equivalents of zinc and four equivalents of acetic acid.

Modern practice is to add the oxime solution resulting from the nitrosation and the zinc dust gradually to a well-stirred solution of ethyl acetoacetate in glacial acetic acid. The reaction is exothermic, and the mixture can reach the boiling point, if external cooling is not applied. The resulting product, diethyl 3,5-dimethylpyrrole-2,4-dicarboxylate, has been called Knorr's Pyrrole ever since. In the Scheme above, R2 = COOEt, and R1 = R3 = Me represent this original reaction.
Knorr's pyrrole can be derivatized in a number of useful manners. One equivalent of sodium hydroxide will saponify the 2-ester selectively. Dissolving Knorr's pyrrole in concentrated sulfuric acid, and then pouring the resulting solution into water will hydrolyze the 4-ester group selectively. The 5-methyl group can be variously oxidized to chloromethyl, aldehyde, or carboxylic acid functionality by the use of stoichiometric sulfuryl chloride in glacial acetic acid. Alternatively, the nitrogen atom can be alkylated. The two ester positions can be more smoothly differentiated by incorporating benzyl or tert-butyl groups via the corresponding acetoacetate esters. Benzyl groups can be removed by catalytic hydrogenolysis over palladium on carbon, and tertiary-butyl groups can be removed by treatment with trifluoroacetic acid, or boiling aqueous acetic acid. R1 and R3 (as well as R2 and "Et") can be varied by the application of appropriate β-ketoesters readily made by a synthesis emanating from acid chlorides, Meldrum's acid, and the alcohol of one's choice. Ethyl and benzyl esters are easily made thereby, and the reaction is noteworthy in that even the highly hindered tert-butyl alcohol gives very high yields in this synthesis.
Levi and Zanetti extended the Knorr synthesis in 1894 to the use of acetylacetone (2,4-pentanedione) in reaction with ethyl 2-oximinoacetoacetate. The result was ethyl 4-acetyl-3,5-dimethylpyrrole-2-carboxylate, where "OEt" = R1 = R3 = Me, and R2 = COOEt. The 4-acetyl group could easily be converted to a 4-ethyl group by Wolff-Kishner reduction (hydrazine and alkali, heated); hydrogenolysis, or the use of diborane. Benzyl or tert-butyl acetoacetates also work well in this system, and with close temperature control, the tert-butyl system gives a very high yield (close to 80%). N,N-dialkyl pyrrole-2- and/or 4-carboxamides may be prepared by the use of N,N-dialkyl acetoacetamides in the synthesis. Even thioesters have been successfully prepared, using the method. As for the nitrosation of β-ketoesters, despite the numerous literature specifications of tight temperature control on the nitrosation, the reaction behaves almost like a titration, and the mixture can be allowed to reach even 40 °C without significantly impacting the final yield.
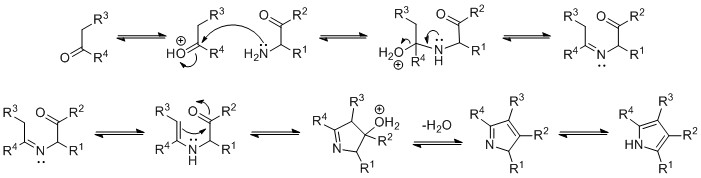
The mechanism of the Knorr pyrrole synthesis begins with condensation of the amine and ketone to give an imine. The imine then tautomerizes to an enamine, followed by cyclization, elimination of water, and isomerization to the pyrrole.
References
References
- (1884). "Synthese von Pyrrolderivaten". [[Berichte der deutschen chemischen Gesellschaft]].
- Knorr, Ludwig. (1886). "Synthetische Versuche mit dem Acetessigester". [[Annalen der Chemie]].
- (1902). "Ueber die Bildung von Pyrrolderivaten aus Isonitrosoketonen". [[Berichte der deutschen chemischen Gesellschaft]].
- Corwin, Alsoph Henry. (1950). "Heterocyclic Compounds". [[John Wiley & Sons]].
- Fischer, Hans. (1935). "2,4-Dimethyl-3,5-dicarbethoxypyrrole (2,4-Pyrroledicarboxylic acid, 3,5-dimethyl-, diethyl ester)".
- Fischer, Hans. (1941). "Kryptopyrrole (Pyrrole, 2,4-dimethyl-3-ethyl)".
- (1942). "Structural Investigations upon a Substituted Dipyrrylmethane. An Unusual Melting Point-Symmetry Relationship". [[Journal of the American Chemical Society]].
- (1978). "Meldrum's acid in organic synthesis. 2. A general and versatile synthesis of β-keto esters". [[The Journal of Organic Chemistry]].
- (1894). "Sintesi di composti pirrolici dai nitrosochetoni". [[La Gazzetta Chimica Italiana]].
- (1954). "''tert''-Butylester von Pyrrolcarbonsäuren". [[Chemische Berichte]].
- (1966). "Preparation and reactions of some pyrrylthiol esters". [[Canadian Journal of Chemistry]].
- (1948). "Über eine neue Pyrrolsynthese". [[Zeitschrift für Physiologische Chemie]].
- Kleinspehn, George G.. (1955). "A Novel Route to Certain 2-Pyrrolecarboxylic Esters and Nitriles". [[Journal of the American Chemical Society]].
- (1985). "Pyrrole chemistry. An improved synthesis of ethyl pyrrole-2-carboxylate esters from diethyl aminomalonate". [[The Journal of Organic Chemistry]].
- (1958). "287. A synthesis of coproporphyrin III". [[Journal of the Chemical Society (Resumed)]].
- (1987). "Mechanism of the formation of ''N'',''N''-dialkyl-2-pyrrolecarboxamides from 1,3-diketones and ''N'',''N''-dialkyloximinoacetoacetamides". [[The Journal of Organic Chemistry]].
- (1971). "Mechanism of a modified Knorr pyrrole condensation". [[The Journal of Organic Chemistry]].
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Knorr pyrrole synthesis — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report