From Surf Wiki (app.surf) — the open knowledge base
Intestinal permeability
Medical term
Medical term
Intestinal permeability is a term describing the control of material passing from inside the gastrointestinal tract through the cells lining the gut wall, into the rest of the body. The intestine normally exhibits some permeability, which allows nutrients to pass through the gut, while also maintaining a barrier function to keep potentially harmful substances (such as antigens) from leaving the intestine and migrating to the body more widely. In a healthy human intestine, small particles (
Physiology

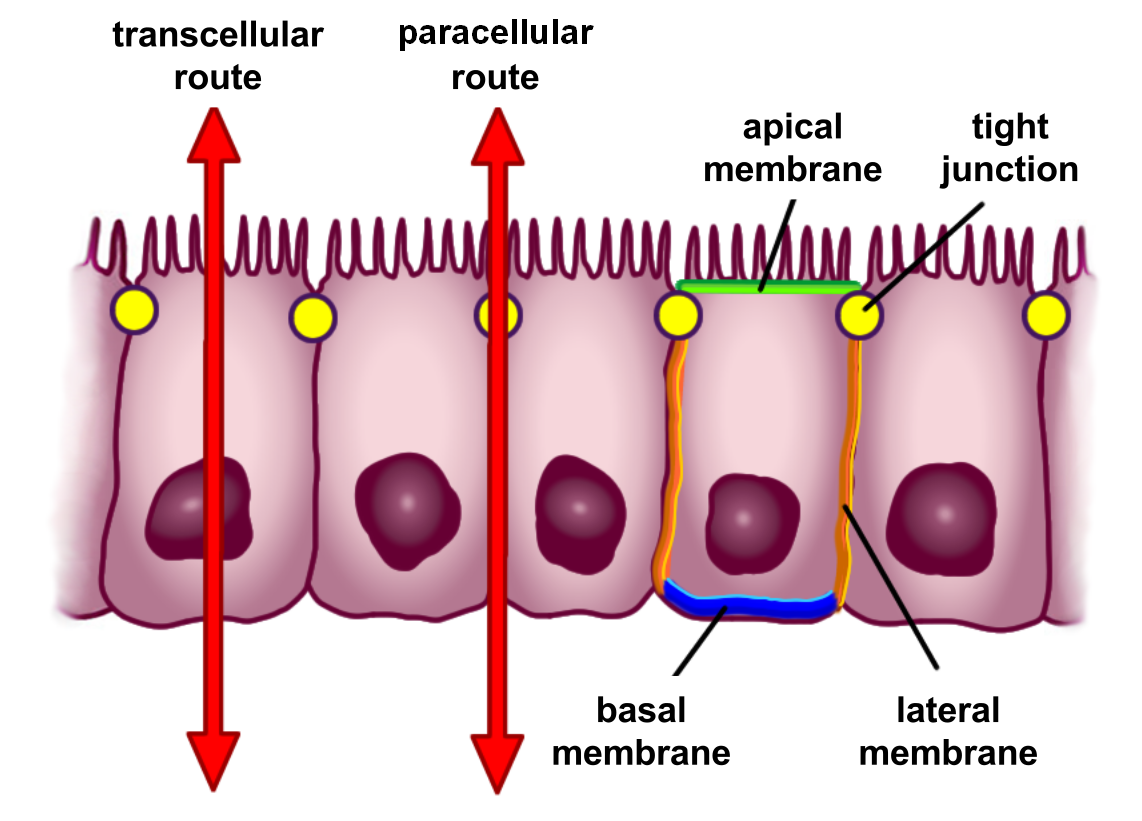
The barrier formed by the intestinal epithelium separates the external environment (the contents of the intestinal lumen) from the body and is the most extensive and important mucosal surface of the body. However, the intestinal mucin can also be barriers for the host antimicrobial peptides, thus plays a bidirectional barrier for host-microbial interaction. The intestinal epithelium is composed of a single layer of cells and serves two crucial functions. First, it acts as a barrier, preventing the entry of harmful substances such as foreign antigens, toxins and microorganisms. Second, it acts as a selective filter which facilitates the uptake of dietary nutrients, electrolytes, water and various other beneficial substances from the intestinal lumen. Selective permeability is mediated via two major routes:
- Transepithelial or transcellular permeability. This consists of specific transport of solutes across the epithelial cells. It is predominantly regulated by the activities of specialised transporters that translocate specific electrolytes, amino acids, sugars, short chain fatty acids and other molecules into or out of the cell.
- Paracellular permeability. It depends on transport through the spaces that exist between epithelial cells. It is regulated by cellular junctions that are localized in the laminal membranes of the cells. Disruption of the tight junction barrier can be a trigger for the development of intestinal diseases.
Modulation
One way in which intestinal permeability is modulated is via CXCR3 receptors in cells in the intestinal epithelium, which respond to zonulin.
Gliadin (a glycoprotein present in wheat) activates zonulin signaling in all people who eat gluten, irrespective of the genetic expression of autoimmunity. This leads to increased intestinal permeability to macromolecules. Bacterial infections such as cholera, select enteric viruses, parasites, and stress can all modulate intestinal tight junction structure and function, and these effects may contribute to the development of chronic intestinal disorders. So called absorption modifying excipients, investigated for the possibility of increasing intestinal drug absorption, can increase the gut permeability.
Clinical significance
Most people do not experience adverse symptoms, but the opening of intercellular tight junctions (increased intestinal permeability) can act as a trigger for diseases that can affect any organ or tissue depending on genetic predisposition.
Increased intestinal permeability is a factor in several diseases, such as celiac disease, irritable bowel syndrome, type 1 diabetes, type 2 diabetes, rheumatoid arthritis, spondyloarthropathies, inflammatory bowel disease, schizophrenia, certain types of cancer, fatty liver, atopy and allergic diseases,
A well studied model is celiac disease, in which increased intestinal permeability appears secondary to the abnormal immune reaction induced by gluten and allows fragments of gliadin protein to get past the intestinal epithelium, triggering an immune response at the intestinal submucosa level that leads to diverse gastrointestinal or extra-gastrointestinal symptoms. Other environmental triggers may contribute to alter permeability in celiac disease, including intestinal infections and iron deficiency.
Research directions
In normal physiology, glutamine plays a key role in signalling in enterocytes that are part of the intestinal barrier, but it is not clear if supplementing the diet with glutamine is helpful in conditions where there is increased intestinal permeability.
Prebiotics and certain probiotics such as E. coli strain Nissle 1917 have been found to reduce increased intestinal permeability. Lactobacillus rhamnosus, Lactobacillus reuteri, and Faecalibacterium prausnitzii have also been shown to significantly reduce increased intestinal permeability.
Larazotide acetate (previously known as AT-1001) is a zonulin receptor antagonist that has been probed in clinical trials. It seems to be a drug candidate for use in conjunction with a gluten-free diet in people with celiac disease, with the aim to reduce the intestinal permeability caused by gluten and its passage through the epithelium, and therefore mitigating the resulting cascade of immune reactions.
Genetic disruption of arginase-2 in mouse attenuates the onset of senescence and extends lifespan. Arginase inhibitors have been developed to reduce the effect of NO on intestinal permeability.
Leaky gut syndrome
Main article: Leaky gut syndrome
"Leaky gut syndrome" is a hypothetical, medically unrecognized condition.
Exercise-induced stress
Exercise-induced stress can diminish intestinal barrier function. In humans, the level of physical activity modulates the gastrointestinal microbiota, an increased intensity and volume of exercise may lead to gut dysbiosis, and supplementation may keep gut microbiota in biodiversity, especially with intense exercise. In mice, exercise reduced the richness of the microbial community, but increased the distribution of bacterial communities.
References
References
- (31 March 2002). "Inflammatory Bowel Disease: A Clinical Case Approach to Pathophysiology, Diagnosis, and Treatment". Springer.
- (2012). "Physiology of the Gastrointestinal Tract". Academic Press.
- Fasano, A.. (February 2012). "Leaky Gut and Autoimmune Diseases". Clinical Reviews in Allergy & Immunology.
- (July 2009). "Intestinal barrier function: molecular regulation and disease pathogenesis". The Journal of Allergy and Clinical Immunology.
- (2010-01-01). "Regulation of Gastrointestinal Mucosal Growth". Morgan & Claypool Life Sciences.
- (July 2013). "Deficiency of intestinal mucin-2 ameliorates experimental alcoholic liver disease in mice". Hepatology.
- (2015-01-01). "Transcriptional regulators of claudins in epithelial tight junctions". Mediators of Inflammation.
- (2019). "The Roles of Peyer's Patches and Microfold Cells in the Gut Immune System: Relevance to Autoimmune Diseases". Frontiers in Immunology.
- Fasano A. (Jan 2011). "Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer". Physiological Reviews.
- (2010). "Possible links between intestinal permeability and food processing: A potential therapeutic niche for glutamine". Clinics.
- (2017). "Celiac Disease and Nonceliac Gluten Sensitivity: A Review". JAMA.
- (December 2017). "Preclinical Effect of Absorption Modifying Excipients on Rat Intestinal Transport of Model Compounds and the Mucosal Barrier Marker 51Cr-EDTA". Molecular Pharmaceutics.
- (Nov 18, 2014). "Intestinal permeability--a new target for disease prevention and therapy". BMC Gastroenterology.
- (2020). "Irritable bowel syndrome and gut microbiota". Current Opinion in Endocrinology, Diabetes & Obesity.
- (2015). "Gut barrier in health and disease: focus on childhood". European Review for Medical and Pharmacological Sciences.
- (Mar 2013). "The role of the microbiome in rheumatic diseases". Current Rheumatology Reports.
- (2016). "Modulatory Effects of Gut Microbiota on the Central Nervous System: How Gut Could Play a Role in Neuropsychiatric Health and Diseases". Journal of Neurogastroenterology and Motility.
- (2016). "Autoimmune diseases, gastrointestinal disorders and the microbiome in schizophrenia: more than a gut feeling". Schizophrenia Research.
- (November 2014). "Gut microbiota and metabolic syndrome". World Journal of Gastroenterology.
- (2004). "A brief evidence-based review of two gastrointestinal illnesses: irritable bowel and leaky gut syndromes". Alternative Therapies in Health and Medicine.
- (September 2012). "Intestinal permeability in coeliac disease: insight into mechanisms and relevance to pathogenesis". Gut.
- (January 2016). "The potential utility of tight junction regulation in celiac disease: focus on larazotide acetate". Therapeutic Advances in Gastroenterology.
- (October 2012). "Intestinal permeability and its regulation by zonulin: diagnostic and therapeutic implications". Clinical Gastroenterology and Hepatology.
- (February 2016). "Glutamine for induction of remission in Crohn's disease". The Cochrane Database of Systematic Reviews.
- (2015). "The therapeutic management of gut barrier leaking: the emerging role for mucosal barrier protectors". European Review for Medical and Pharmacological Sciences.
- (2018). "Causal Relationship between Diet-Induced Gut Microbiota Changes and Diabetes: A Novel Strategy to Transplant Faecalibacterium prausnitzii in Preventing Diabetes". International Journal of Molecular Sciences.
- (Jan 2012). "Non-dietary therapeutic clinical trials in coeliac disease". European Journal of Internal Medicine.
- (2017). "Arginase-II Deficiency Extends Lifespan in Mice". Front Physiol.
- (December 2022). "Impairments of intestinal arginine and NO metabolisms trigger aging-associated intestinal barrier dysfunction and 'inflammaging'". Redox Biol.
- (26 February 2015). "Leaky gut syndrome". [[NHS Choices]].
- (5 January 2016). "Exercise-induced stress behavior, gut-microbiota-brain axis and diet: a systematic review for athletes". Journal of the International Society of Sports Nutrition.
- (1 October 2020). "Exercise and intestinal permeability: another form of exercise-induced hormesis?". American Journal of Physiology. Gastrointestinal and Liver Physiology.
- (December 2012). "Aggravation of Exercise-Induced Intestinal Injury by Ibuprofen in Athletes". Medicine & Science in Sports & Exercise.
- (7 July 2022). "Physical activity induced alterations of gut microbiota in humans: a systematic review". BMC Sports Science, Medicine and Rehabilitation.
- (12 October 2021). "The Relationship among Physical Activity, Intestinal Flora, and Cardiovascular Disease". Cardiovascular Therapeutics.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Intestinal permeability — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report