From Surf Wiki (app.surf) — the open knowledge base
Hexanitrobenzene
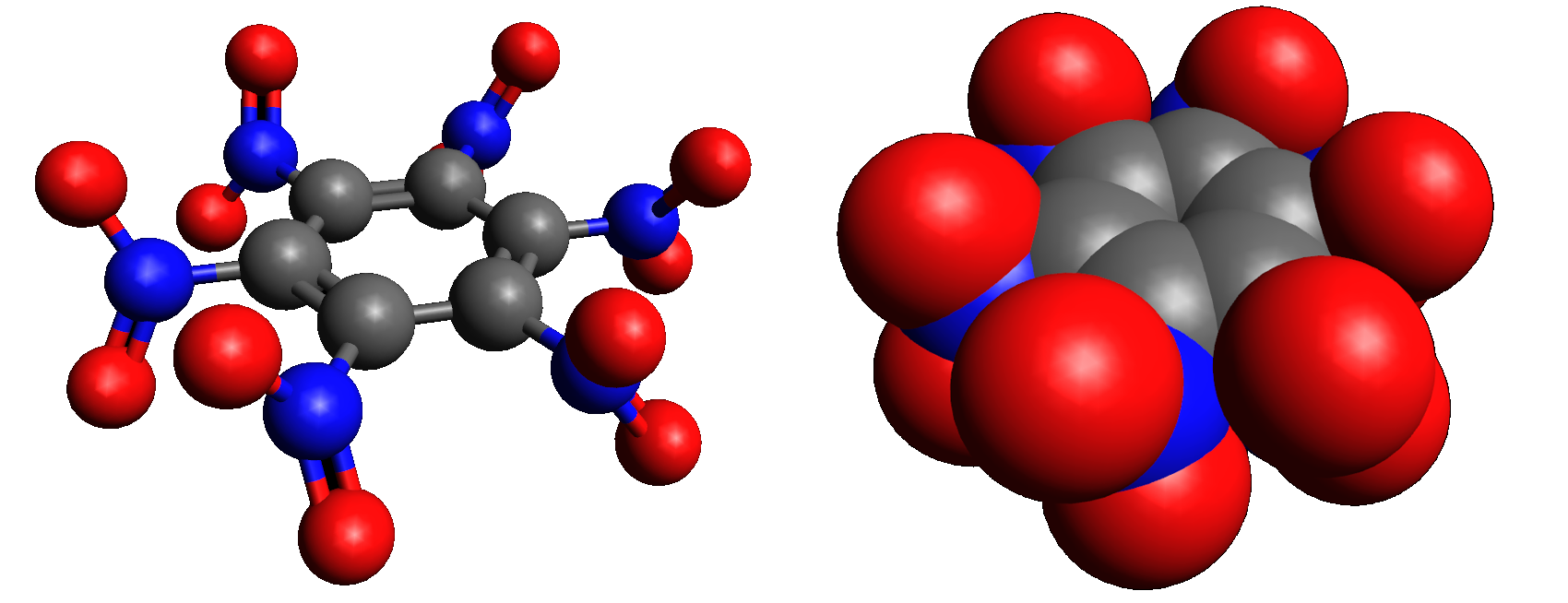
Hexanitrobenzene, also known as HNB, is a nitrobenzene compound in which six nitro groups are bonded to all six positions of a central benzene ring. It is a high-density explosive compound with chemical formula , obtained by oxidizing the amine group of pentanitroaniline with hydrogen peroxide in sulfuric acid.
Properties

The stable conformation of this molecule has the nitro groups rotated out of the plane of the central benzene ring. The molecule adopts a propeller-like conformation in which the nitro groups are rotated about 53° from planar.
HNB has the undesirable property of being moderately sensitive to light and, therefore, hard to utilize safely. As of 2021, it is not used in any production explosives applications, though it is used as a precursor chemical in one method of production of TATB, another explosive.
HNB was experimentally used as a gas source for explosively pumped gas dynamic laser. In this application, HNB and tetranitromethane are preferred to more conventional explosives because the explosion products and are a simple enough mixture to simulate gas dynamic processes and quite similar to conventional gas dynamic laser medium. The water and hydrogen products of many other explosives could interfere with vibrational states of in this type of laser.
Preparation
During World War II, a method of synthesis of hexanitrobenzene was suggested in Germany, and the product was supposed to be manufactured on a semi-industrial scale according to the following scheme: : (partial reduction) : (nitration) : (oxidation) Complete nitration of benzene is practically impossible because the nitro groups are deactivating groups for further nitration.
Additional properties
- Chapman-Jouget detonation pressure: 43 GPa
- Crystal Density: 2.01
Notes
References
References
- [http://www.osti.gov/energycitations/servlets/purl/61699-SYl1FI/webviewable/61699.pdf Accurate determination of pair potentials for a CwHxNyOz system of molecules: A semiempirical method], Thiel et al., 1995
- (1968). "The crystal structure of a modification of hexaphenylbenzene". Acta Crystallographica Section B.
- [http://www.freepatentsonline.com/4099142.html Condensed explosive gas dynamic laser], United States Patent 4099142
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Hexanitrobenzene — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report