From Surf Wiki (app.surf) — the open knowledge base
Hemiaminal
Organic compound or group with a hydroxyl and amine attached to the same carbon
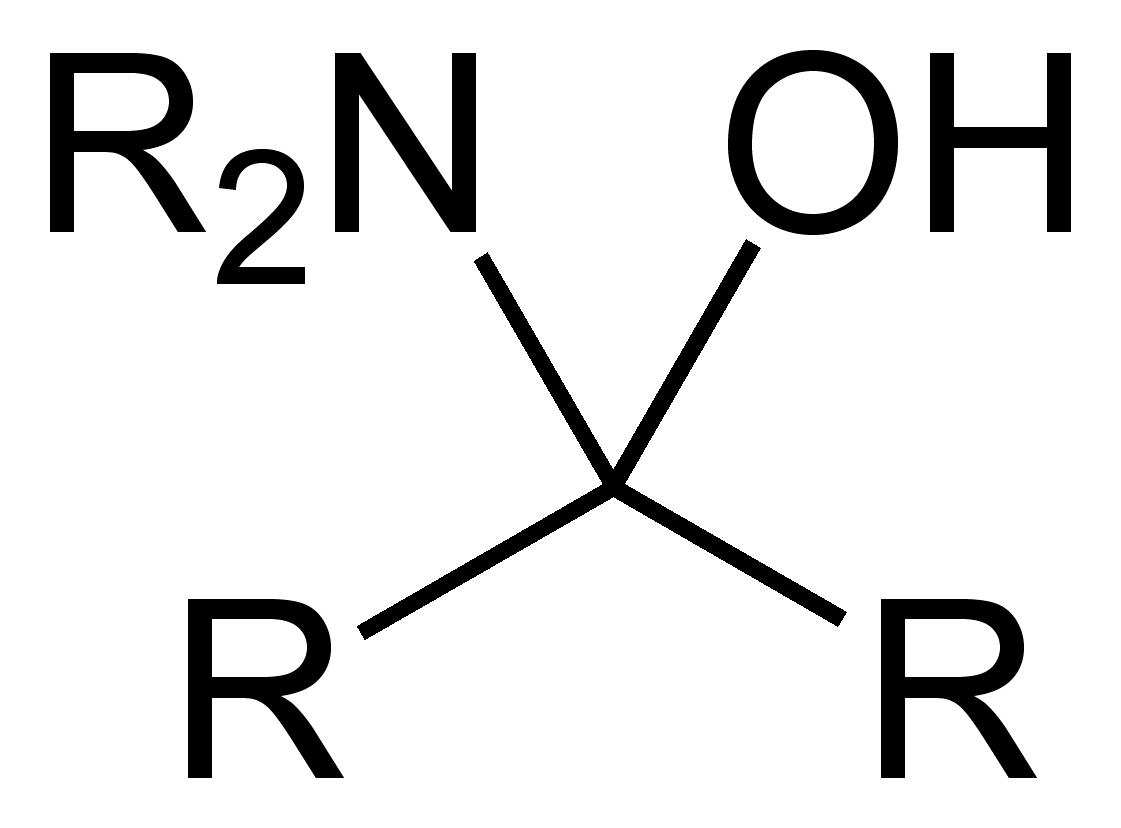
Organic compound or group with a hydroxyl and amine attached to the same carbon

In organic chemistry, a hemiaminal (also carbinolamine) is a functional group or type of chemical compound that has a hydroxyl group and an amine attached to the same carbon atom: . R can be hydrogen or an alkyl group. Hemiaminals are intermediates in imine formation from an amine and a carbonyl by alkylimino-de-oxo-bisubstitution. Hemiaminals can be viewed as a blend of aminals and geminal diol. They are a special case of amino alcohols.
Classification according to amine precursor
Hemiaminals form from the reaction of an amine and a ketone or aldehyde. The hemiaminal is sometimes isolable, but often they spontaneously dehydrate to give imines.
Addition of ammonia
The adducts formed by the addition of ammonia to aldehydes have long been studied. Compounds containing both a primary amino group and a hydroxyl group bonded to the same carbon atom are rarely stable ("The hemiaminal [derived from primary amines] is, except in very special cases... not observed"), as they tend to dehydrate to form imines which polymerise to hexamethylenetetramine. A rare stable example is the adduct of ammonia and hexafluoroacetone, .
The C-substituted derivatives are obtained by reaction of aldehydes and ammonia: :3 RCHO + 3 NH3 - (RCHNH)3 + 3 H2O
Addition of primary amines
N-substituted derivatives are somewhat stable. They are invoked but rarely observed as intermediates in the Mannich reaction. These N,N',N''-trisubstituted hexahydro-1,3,5-triazines arise from the condensation of the amine and formaldehyde as illustrated by the route to 1,3,5-trimethyl-1,3,5-triazacyclohexane: :3 CH2O + 3 H2NMe - (CH2NMe)3 + 3 H2O
Although adducts generated from primary amines or ammonia are usually unstable, the hemiaminals have been trapped in a cavity.
Addition of secondary amines: carbinolamines (hemiaminals) and bisaminomethanes
One of the simplest reactions entails condensation of formaldehyde and dimethylamine. This reaction produces first the carbinolamine (a hemiaminal) and bis(dimethylamino)methane (): :Me2NH + CH2O - Me2NCH2OH :Me2NH + Me2NCH2OH - Me2NCH2NMe2 + H2O
The reaction of formaldehyde with carbazole, which is weakly basic, proceed similarly: :[[File:CarbazoleFormaldehydeReaction.png|400px|reaction of carbazole with formaldehyde to Carbazol-9-yl-methanol]] Again, this carbinol converts readily to the methylene-linked bis(carbazole).
Hemiaminal ethers
Hemiaminal ethers have the following structure: R‴-C(NR'2)(OR")-R⁗. The glycosylamines are examples of cyclic hemiaminal ethers. Methanolamine.svg|methanolamine, an intermediate in the reaction of ammonia with formaldehyde OC(NHCH2OH)2.png|Bis(hydroxymethyl)urea is a commercially useful hemiaminal CF3-stabilizedHemiaminal.svg|An unusual example of an isolable, acyclic hemiaminal: the adduct of ammonia and hexafluoroacetone Hemiaminal ether aldehyde.png| Hemiaminal ether derived from an aldehyde Hemiaminal ether ketone.png| Hemiaminal ether derived from a ketone Bredereck's reagent.svg|Tert-Butoxybis(dimethylamino)methane
Use in total synthesis
Hemiaminal formation is a key step in an asymmetric total synthesis of saxitoxin:
:[[File:Hemiaminalformationinsaxitoxinsynthesis.png|400px|Hemiaminal formation in saxitox in synthesis]] In this reaction step the alkene group is first oxidized to an intermediate acyloin by action of osmium(III) chloride, oxone (sacrificial catalyst) and sodium carbonate (base).
References
References
- (2000). "Carbinolamines and Geminal Diols in Aqueous Environmental Organic Chemistry". Journal of Chemical Education.
- (1995). "Comprehensive Organic Functional Group Transformations".
- Justus Liebig "Ueber die Producte der Oxydation des Alkohols" Annalen der Pharmacie 1835, Volume 14, pp 133–167. {{doi. 10.1002/jlac.18350140202
- (2007). "Stabilization of Labile Carbonyl Addition Intermediates by a Synthetic Receptor". Science.
- W. J. Middleton, H. D. Carlson. (1970). "Hexafluoroacetone imine". Org. Syntheses.
- (1973). "Structure and Chemistry of the Aldehyde Ammonias. 1-Amino-1-Alkanols, 2,4,6-Trialkyl-1,3,5-Hexahydrotriazines, and N,N-Dialkylidene-1,1-Diaminoalkanes". J. Org. Chem..
- (2007). "Stabilization of Labile Carbonyl Addition Intermediates by a Synthetic Receptor". Science.
- Hellmann Heinrich, Günter Opitz. (1960). "Aminoalkylierung".
- (1974). "Thermochemistry of Carbonyl Addition Reactions. II. Enthalpy of Addition of Dimethylamine to Formaldehyde". The Journal of Physical Chemistry.
- ''Carbazol-9-yl-methanol'' Milata Viktor, Kada Rudolf, Lokaj Ján [[Molbank]] 2004, M354 [[Open access (publishing). open access]] publication [http://www.mdpi.net/molbank/molbank2004/m0354.htm] {{Webarchive. link. (2018-09-26)
- (2007). "(+)-Saxitoxin: A First and Second Generation Stereoselective Synthesis". Journal of the American Chemical Society.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Hemiaminal — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report