From Surf Wiki (app.surf) — the open knowledge base
Helminthiasis
Any macroparasitic disease caused by helminths
Any macroparasitic disease caused by helminths
| Field | Value |
|---|---|
| name | Helminthiasis |
| image | Ascaris infection in the x-ray image- ascaris arranged tidily along the long axis of the small bowel (South Africa) (16424840021).jpg |
| caption | Ascaris worms (one type of helminth) in the small bowel of an infected person (X-ray image with barium as contrast medium) |
| pronounce | |
| synonyms | Worm infection, helminthosis, helminthiases, helminth infection |
Helminthiasis, also known as worm infection, is any macroparasitic disease of humans and other animals in which a part of the body is infected with parasitic worms, known as helminths. There are numerous species of these parasites, which are broadly classified into tapeworms, flukes, and roundworms. They often live in the gastrointestinal tract of their hosts, but they may also burrow into other organs, where they induce physiological damage.
Soil-transmitted helminthiasis and schistosomiasis are the most important helminthiases, and are among the neglected tropical diseases. These group of helminthiases have been targeted under the joint action of the world's leading pharmaceutical companies and non-governmental organizations through a project launched in 2012 called the London Declaration on Neglected Tropical Diseases, which aimed to control or eradicate certain neglected tropical diseases by 2020.
Helminthiasis has been found to result in poor birth outcome, poor cognitive development, poor school and work performance, poor socioeconomic development, and poverty. Chronic illness, malnutrition, and anemia are further examples of secondary effects.
Soil-transmitted helminthiases are responsible for parasitic infections in as much as a quarter of the human population worldwide. One well-known example of soil-transmitted helminthiases is ascariasis.
Types of parasitic helminths

Of all the known helminth species, the most important helminths with respect to understanding their transmission pathways, their control, inactivation and enumeration in samples of human excreta from dried feces, faecal sludge, wastewater, and sewage sludge are:
- soil-transmitted helminths, including Ascaris lumbricoides (the most common worldwide), Trichuris trichiura, Necator americanus, Strongyloides stercoralis and Ancylostoma duodenale
- Hymenolepis nana
- Taenia saginata
- Enterobius
- Fasciola hepatica
- Schistosoma mansoni
- Toxocara canis
- Toxocara cati
Helminthiases are classified as follows (the disease names end with "-sis" and the causative worms are in brackets):
Roundworm infection (nematodiasis)
- Filariasis (filarial worms: Wuchereria bancrofti, Brugia malayi infection)
- Onchocerciasis (Onchocerca volvulus infection)
- Soil-transmitted helminthiasis – this includes ascariasis (Ascaris lumbricoides infection), trichuriasis (Trichuris infection), and hookworm infection (includes necatoriasis and Ancylostoma duodenale infection)
- Trichostrongyliasis (Trichostrongylus spp. infection)
- Dracunculiasis (guinea worm infection)
- Baylisascaris (raccoon roundworm, may be transmitted to pets, livestock, and humans)
Tapeworm infection (cestodiasis)
- Echinococcosis (Echinococcus infection)
- Hymenolepiasis (Hymenolepis infection)
- Taeniasis/cysticercosis (Taenia infection)
- Coenurosis (T. multiceps, T. serialis, T. glomerata, and T. brauni infection)
Trematode infection (trematodiasis)
- Amphistomiasis (amphistomes infection)
- Clonorchiasis (Clonorchis sinensis infection)
- Fascioliasis (Fasciola infection)
- Fasciolopsiasis (Fasciolopsis buski infection)
- Opisthorchiasis (Opisthorchis infection)
- Paragonimiasis (Paragonimus infection)
- Schistosomiasis/bilharziasis (Schistosoma infection)
Acanthocephala infection
- Moniliformis infection
Signs and symptoms
_(15806559913).jpg)
The signs and symptoms of helminthiasis depend on a number of factors including: the site of the infestation within the body; the type of worm involved; the number of worms and their volume; the type of damage the infesting worms cause; and, the immunological response of the body. Where the burden of parasites in the body is light, there may be no symptoms.
Certain worms may cause particular constellations of symptoms. For instance, taeniasis can lead to seizures due to neurocysticercosis.
Mass and volume
In extreme cases of intestinal infestation, the mass and volume of the worms may cause the outer layers of the intestinal wall, such as the muscular layer, to tear. This may lead to peritonitis, volvulus, and gangrene of the intestine.
Immunological response
As pathogens in the body, helminths induce an immune response. Immune-mediated inflammatory changes occur in the skin, lung, liver, intestine, central nervous system, and eyes. Signs of the body's immune response may include eosinophilia, edema, and arthritis. An example of the immune response is the hypersensitivity reaction that may lead to anaphylaxis. Another example is the migration of Ascaris larvae through the bronchi of the lungs causing asthma.
Secondary effects
Immune changes
In humans, T helper cells and eosinophils respond to helminth infestation. It is well established that T helper 2 cells are the central players of protective immunity to helminths, while the roles for B cells and antibodies are context-dependent. Inflammation leads to encapsulation of egg deposits throughout the body. Helminths excrete into the intestine toxic substances after they feed. These substances then enter the circulatory and lymphatic systems of the host body.
Chronic immune responses to helminthiasis may lead to increased susceptibility to other infections such as tuberculosis, HIV, and malaria. There is conflicting information about whether deworming reduces HIV progression and viral load and increases CD4 counts in antiretroviral naive and experienced individuals, although the most recent Cochrane review found some evidence that this approach might have favorable effects. Helminth infection also lowers the immune responses to vaccination for other diseases such as BCG, measles, and Hepatitis B.
Chronic illness
Chronic helminthiasis may cause severe morbidity. Helminthiasis has been found to result in poor birth outcome, poor cognitive development, poor school and work performance, decreased productivity, poor socioeconomic development, and poverty.
Malnutrition
Helminthiasis may cause chronic illness through malnutrition including vitamin deficiencies, stunted growth, anemia, and protein-energy malnutrition. Worms compete directly with their hosts for nutrients, but the magnitude of this effect is likely minimal as the nutritional requirements of worms is relatively small. In pigs and humans, Ascaris has been linked to lactose intolerance and vitamin A, amino acid, and fat malabsorption. Impaired nutrient uptake may result from direct damage to the intestinal mucosal wall or from more subtle changes such as chemical imbalances and changes in gut flora. Alternatively, the worms' release of protease inhibitors to defend against the body's digestive processes may impair the breakdown of other nutrients. In addition, worm induced diarrhoea may shorten gut transit time, thus reducing absorption of nutrients.
Malnutrition due to worms can give rise to anorexia. A study of 459 children in Zanzibar revealed spontaneous increases in appetite after deworming. Anorexia might be a result of the body's immune response and the stress of combating infection. Specifically, some of the cytokines released in the immune response to worm infestation have been linked to anorexia in animals.
Anemia
Helminths may cause iron-deficiency anemia. This is most severe in heavy hookworm infections, as Necator americanus and Ancylostoma duodenale feed directly on the blood of their hosts. Although the daily consumption of an individual worm (0.02–0.07 ml and 0.14–0.26 ml respectively) is small, the collective consumption under heavy infection can be clinically significant. Intestinal whipworm may also cause anemia. Anemia has also been associated with reduced stamina for physical labor, a decline in the ability to learn new information, and apathy, irritability, and fatigue. A study of the effect of deworming and iron supplementation in 47 students from the Democratic Republic of the Congo found that the intervention improved cognitive function. Another study found that in 159 Jamaican schoolchildren, deworming led to better auditory short-term memory and scanning and retrieval of long-term memory over a period of nine-weeks.
Cognitive changes
Malnutrition due to helminths may affect cognitive function leading to low educational performance, decreased concentration and difficulty with abstract cognitive tasks. Iron deficiency in infants and preschoolers is associated with "lower scores ... on tests of mental and motor development ... [as well as] increased fearfulness, inattentiveness, and decreased social responsiveness". Studies in the Philippines and Indonesia found a significant correlation between helminthiasis and decreased memory and fluency. Large parasite burdens, particularly severe hookworm infections, are also associated with absenteeism, under-enrollment, and attrition in school children.
Transmission
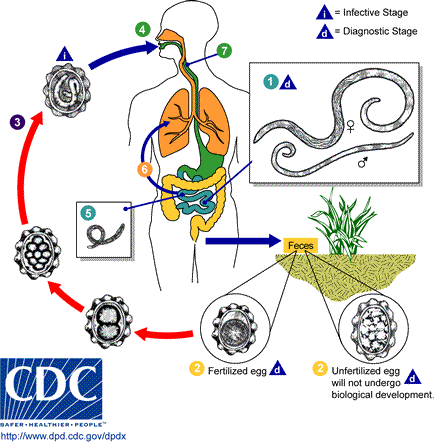
Helminths are transmitted to the final host in several ways. The most common infection is through ingestion of contaminated vegetables, drinking water, and raw or undercooked meat. Contaminated food may contain eggs of nematodes such as Ascaris, Enterobius, and Trichuris; cestodes such as Taenia, Hymenolepis, and Echinococcus; and trematodes such as Fasciola. Raw or undercooked meats are the major sources of Taenia (pork, beef and venison), Trichinella (pork and bear), Diphyllobothrium (fish), Clonorchis (fish), and Paragonimus (crustaceans). Schistosomes and nematodes such as hookworms (Ancylostoma and Necator) and Strongyloides can penetrate the skin directly.
The roundworm, Dracunculus has a complex mode of transmission: it is acquired from drinking infested water or eating frogs and fish that contain (had eaten) infected crustaceans (copepods); and can also be transmitted from infected pets (cats and dogs). Roundworms such as Brugia, Wuchereria and Onchocerca are directly transmitted by mosquitoes. In the developing world, the use of contaminated water is a major risk factor for infection. Infection can also take place through the practice of geophagy, which is not uncommon in parts of sub-Saharan Africa. Soil is eaten, for example, by children or pregnant women to counteract a real or perceived deficiency of minerals in their diet.
Diagnosis

Specific helminths can be identified through microscopic examination of their eggs (ova) found in faecal samples. The number of eggs is measured in units of eggs per gram. Sophisticated tests such as serological assays, antigen tests, and molecular diagnosis are also available;
Prevention
Disrupting the cycle of the worm will prevent infestation and re-infestation. Prevention of infection can largely be achieved by addressing the issues of WASH—water, sanitation and hygiene. The reduction of open defecation is particularly called for, as is stopping the use of human waste as fertilizer.
Further preventive measures include adherence to appropriate food hygiene, wearing of shoes, regular deworming of pets, and the proper disposal of their feces.
Scientists are also searching for a vaccine against helminths, such as a hookworm vaccine.
Treatment
Medications
Main article: Anthelmintic
Broad-spectrum benzimidazoles (such as albendazole and mebendazole) are the first line treatment of intestinal roundworm and tapeworm infections. Macrocyclic lactones (such as ivermectin) are effective against adult and migrating larval stages of nematodes. Praziquantel is the drug of choice for schistosomiasis, taeniasis, and most types of food-borne trematodiases. Oxamniquine is also widely used in mass deworming programmes. Pyrantel is commonly used for veterinary nematodiasis. Artemisinins and derivatives are proving to be candidates as drugs of choice for trematodiasis.
Mass deworming
Main article: Mass deworming
In regions where helminthiasis is common, mass deworming treatments may be performed, particularly among school-age children, who are a high-risk group. Most of these initiatives are undertaken by the World Health Organization (WHO) with positive outcomes in many regions. Deworming programs can improve school attendance by 25 percent. Although deworming improves the health of an individual, outcomes from mass deworming campaigns, such as reduced deaths or increases in cognitive ability, nutritional benefits, physical growth, and performance, are uncertain or not apparent.
Surgery
If complications of helminthiasis, such as intestinal obstruction occur, emergency surgery may be required. Patients who require non-emergency surgery, for instance for removal of worms from the biliary tree, can be pre-treated with the anthelmintic drug albendazole.
Epidemiology
Areas with the highest prevalence of helminthiasis are tropical and subtropical areas including sub-Saharan Africa, central and east Asia, and the Americas.
Neglected tropical diseases
Some types of helminthiases are classified as neglected tropical diseases. They include:
- Soil-transmitted helminthiases
- Roundworm infections such as lymphatic filariasis, dracunculiasis, and onchocerciasis
- Trematode infections, such as schistosomiasis, and food-borne trematodiases, including fascioliasis, clonorchiasis, opisthorchiasis, and paragonimiasis
- Tapeworm infections such as cysticercosis, taeniasis, and echinococcosis
Prevalence
The soil-transmitted helminths (A. lumbricoides, T. trichiura, N. americanus, A. duodenale), schistosomes, and filarial worms collectively infect more than a quarter of the human population worldwide at any one time, far surpassing HIV and malaria together. Schistosomiasis is the second most prevalent parasitic disease of humans after malaria.
In 2014–15, the WHO estimated that approximately 2 billion people were infected with soil-transmitted helminthiases, 56 million people with food-borne trematodiasis, 120 million with lymphatic filariasis, 37 million people with onchocerciasis, and 1 million people with echinococcosis. Another source estimated a much higher figure of 3.5 billion infected with one or more soil-transmitted helminths.
In 2014, only 148 people were reported to have dracunculiasis because of a successful eradication campaign for that particular helminth, which is easier to eradicate than other helminths as it is transmitted only by drinking contaminated water.
Because of their high mobility and lower standards of hygiene, school-age children are particularly vulnerable to helminthiasis. Most children from developing nations will have at least one infestation. Multi-species infections are very common.
As with other infectious diseases, climate change will have an effect on the distribution and the risk of transmission of helminthiasis. However, it depends on the geographical region, disease-carrying species, the exact climate change scenario, and many other factors whether there will be a decrease or increase in helminthiasis.
In domestic animals, climate change will also have an impact on helminthiasis. For example, in Europe helminths have now spread further towards the poles, with higher survival rate and higher reproductive capacity (fecundity). Detailed long-term records of both livestock diseases and various agricultural interventions in Europe mean that demonstrating the role of climate change in the increased helminth burden in livestock is actually easier than attributing the impact of climate change on diseases which affect humans.
The most common intestinal parasites in the United States are Enterobius vermicularis, Giardia lamblia, Ancylostoma duodenale, Necator americanus, and Entamoeba histolytica.
In a developing country like Bangladesh, the most common species are round worm (Ascaris lumbricoides), whipworm (Tricurias tricuras) and hookworm (Ancylostoma duodenalis).
Variations within communities
Even in areas of high prevalence, the frequency and severity of infection is not uniform within communities or families. A small proportion of community members harbour the majority of worms, and this depends on age. The maximum worm burden is at five to ten years of age, declining rapidly thereafter. Individual predisposition to helminthiasis for people with the same sanitation infrastructure and hygiene behavior is thought to result from differing immunocompetence, nutritional status, and genetic factors. Because individuals are predisposed to a high or a low worm burden, the burden reacquired after successful treatment is proportional to that before treatment.
Disability-adjusted life years
It is estimated that intestinal nematode infections cause 5 million disability-adjusted life years (DALYS) to be lost, of which hookworm infections account for more than 3 million DALYS and ascaris infections more than 1 million. There are also signs of progress: The Global Burden of Disease Study published in 2015 estimates a 46 percent (59 percent when age standardised) reduction in years lived with disability (YLD) for the 13-year time period from 1990 to 2013 for all intestinal/nematode infections, and even a 74 percent (80 percent when age standardised) reduction in YLD from ascariasis.
Deaths
As many as 135,000 die annually from soil transmitted helminthiasis.
The 1990–2013 Global Burden of Disease Study estimated 5,500 direct deaths from schistosomiasis, while more than 200,000 people were estimated in 2013 to die annually from causes related to schistosomiasis. Another 20 million have severe consequences from the disease. It is the most deadly of the neglected tropical diseases.
| Helminth genera | Common name | Infections (million per year) | Direct deaths per year | Regions where common | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Soil transmitted helminthiasis (STH) (classified as neglected tropical disease): | |||||||||||
| *Ascaris lumbricoides* | Roundworm | 1000 to 1450 | 20,000 | vauthors = Jiménez B | title = Irrigation in developing countries using wastewater. | journal = International Review for Environmental Strategies. | date = March 2006 | volume = 6 | issue = 2 | pages = 229–50 | url = https://www.iges.or.jp/en/publication_documents/pub/peer/en/1199/IRES_Vol.6-2_229.pdf }} |
| *Trichuris trichiura* | Whipworm | 500 | url=https://web.archive.org/web/20141017235811/http://www.susana.org/en/resources/library/details/1004 | date=2014-10-17 }}. World Health Organization (WHO), Geneva, Switzerland | |||||||
| *Ancylostoma duodenale* | Hookworm | 900 to 1300 | In tropical and subtropical countries (Sub-Saharan Africa) | ||||||||
| *Necator americanus* | |||||||||||
| *Strongyloides stercoralis* | Hookworm, pinworm | 50 to 100 | Thousands | In moist rainy areas of the tropics and subtropics, in some areas of southern and eastern Europe and of the United States of America | |||||||
| All STH together | 1500 to 2000 | 135,000 | Tropical and subtropical areas, in particular sub-Saharan Africa, the Americas, China and east Asia. | ||||||||
| Not transmitted via soil but classified as neglected tropical disease: | |||||||||||
| *Schistosoma mansoni* | Blood fluke | All types of *Schistosoma *together:* *160 to 200 | 12,000 150,000 deaths from kidney failure | In tropical and subtropical regions | |||||||
| *Schistosoma haematobium* | url=https://web.archive.org/web/20141207140442/http://ebooks.benthamscience.com/book/9781608051489/ | date=2014-12-07 }}, Bentham e books, Vol 2, pp. 93–124, | |||||||||
| *Echinococcus granulosus* | 3 | Developing countries | |||||||||
| Not transmitted via soil and not classified as neglected tropical disease: | |||||||||||
| *Toxocara canis* | Dog roundworm | 50 | Many regions of South-east Asia, Africa, and Central and South America | ||||||||
| *Taenia solium* | Pork tapeworm | 50 | South America, Southeast Asia, West Africa and East Africa | ||||||||
| *Taenia saginata* | Beef tapeworm | 50 | |||||||||
| *Hymenolepis nana* | Dwarf tapeworm | 100 | |||||||||
| *Hymenolepis diminuta* | Rat tapeworm | ||||||||||
| *Fasciola hepatica*, | |||||||||||
| *Fascioloides magna* | Liver fluke | 50 | Largely in southern and eastern Asia but also in central and eastern Europe | ||||||||
| *Fasciolopsis buski* | Giant intestinal fluke | ||||||||||
| *Dracunculus medinensis* | Guinea worm | date=February 2016 | title=Dracunculiasis (guinea-worm disease) Fact sheet N°359 (Revised) | url=https://www.who.int/mediacentre/factsheets/fs359/en | archive-url=https://web.archive.org/web/20130730230030/http://www.who.int/mediacentre/factsheets/fs359/en/ | archive-date=July 30, 2013 | access-date=7 February 2016 | website=World Health Organization}} | Formerly widespread in India, west Africa and southern Sudan | ||
| *Trichostrongylus orientalis* | Roundworm | 1–3 ("several") | Rural communities in Asia | ||||||||
| Other | 100 | Worldwide | |||||||||
| Total (number of infections) | Approx. 3.5 billion | Worldwide |
References
References
- (6 June 2011). "Neglected Tropical Diseases".
- London Declaration. (30 January 2012). "London Declaration on Neglected Tropical Diseases".
- Report of a WHO Expert Committee (1987). [http://whqlibdoc.who.int/trs/WHO_TRS_749.pdf Prevention and Control of Intestinal Parasitic Infections]. World Health Organization, Technical Report Series 749.
- Del Rosso, Joy Miller and Tonia Marek (1996). [https://books.google.com/books?id=XeY4-Y15cCgC&dq=class%20action%3A%20improving%20school%20performance&pg=PP1 Class Action: Improving School Performance in the Developing World through Better Health and Nutrition]. The [[World Bank]], Directions in Development.
- (2012). "Research priorities for helminth infections". World Health Organization Technical Report Series.
- (May 2015). "Soil-transmitted helminth infections".
- (October 2012). "Viability of six species of larval and non-larval helminth eggs for different conditions of temperature, pH and dryness". Water Research.
- (2012). "Neurocysticercosis: a review". TheScientificWorldJournal.
- (February 1996). "Surgical management of worm volvulus". South African Journal of Surgery. Suid-Afrikaanse Tydskrif vir Chirurgie.
- (October 2012). "Anaphylaxis caused by helminths: review of the literature". European Review for Medical and Pharmacological Sciences.
- (2006). "Markell and Vogue's Medical Parasitology, 9th Edition". Saunders Elsevier Press.
- (September 2017). "Th2 and Th1 Responses: Clear and Hidden Sides of Immunity Against Intestinal Helminths". Trends in Parasitology.
- (May 2021). "Context-dependent roles of B cells during intestinal helminth infection". PLOS Neglected Tropical Diseases.
- (2007). "Chronic helminth infections induce immunomodulation: consequences and mechanisms". Immunobiology.
- (June 2012). "Status of medical parasitology in South Africa: new challenges and missed opportunities". Trends in Parasitology.
- (2000). "Eradication of helminthic infections may be essential for successful vaccination against HIV and tuberculosis". Bulletin of the World Health Organization.
- (August 2014). "Empiric deworming and CD4 count recovery in HIV-infected Ugandans initiating antiretroviral therapy". PLOS Neglected Tropical Diseases.
- (April 2016). "Antihelminthics in helminth-endemic areas: effects on HIV disease progression". The Cochrane Database of Systematic Reviews.
- (17 June 2022). "The effect of helminth infection on vaccine responses in humans and animal models: A systematic review and meta-analysis". Parasite Immunology.
- (1996). "Medical Microbiology". Galveston (TX): The University of Texas Medical Branch at Galveston.
- Crompton, D. W. T. (1993). [https://books.google.com/books?id=oT6BrfK7XQYC&dq=crompton%20Human%20Nutrition%20and%20Parasitic%20Infection.&pg=PA5202 Human Nutrition and Parasitic Infection]. Cambridge University Press.
- (1992). "Nutrition, Health, and Learning: Current Issues and Trends".
- (March 1997). ""Stupidity or worms": do intestinal worms impair mental performance?". Psychological Bulletin.
- The World Bank. "World Development Report 1993: Investing in Health".
- (February 2004). "Low dose daily iron supplementation improves iron status and appetite but not anemia, whereas quarterly anthelminthic treatment improves growth, appetite and anemia in Zanzibari preschool children". The Journal of Nutrition.
- (April 1993). "Improvements in cognitive performance for schoolchildren in Zaire, Africa, following an iron supplement and treatment for intestinal parasites". Journal of Pediatric Psychology.
- (February 1992). "Parasitic helminth infection and cognitive function in school children". Proceedings. Biological Sciences.
- (May 2005). "Helminth infection and cognitive impairment among Filipino children". The American Journal of Tropical Medicine and Hygiene.
- (May 1999). "Evidence for an association between hookworm infection and cognitive function in Indonesian school children". Tropical Medicine & International Health.
- (August 2019). "Dracunculiasis: water-borne anthroponosis vs. food-borne zoonosis". Journal of Helminthology.
- (October 2017). "The Current Status of Molecular Xenomonitoring for Lymphatic Filariasis and Onchocerciasis". Trends in Parasitology.
- (January 1989). "Clinical and laboratory aspects of filariasis". Clinical Microbiology Reviews.
- (July 2022). "Contaminated drinking water".
- (2010). "Microbiological and health related perspectives of geophagia: an overview". African Journal of Biotechnology.
- (2012). "An in-depth analysis of a piece of shit: distribution of Schistosoma mansoni and hookworm eggs in human stool". PLOS Neglected Tropical Diseases.
- "Water, Sanitation & Hygiene: Strategy Overview".
- "Water, Sanitation, and Hygiene: Introduction".
- "Goal 7: Ensure Environmental Sustainability".
- (January 2012). "Effect of sanitation on soil-transmitted helminth infection: systematic review and meta-analysis". PLOS Medicine.
- (March 2014). "Water, sanitation, hygiene, and soil-transmitted helminth infection: a systematic review and meta-analysis". PLOS Medicine.
- (April 2013). "The Human Hookworm Vaccine". Vaccine.
- "Anthelmintics".
- "Overview of Anthelmintics". Merck Sharp & Dohme Corp..
- (2012). "Systematic review and meta-analysis of artemisinin based therapies for the treatment and prevention of schistosomiasis". PLOS ONE.
- WHO. (2006). "Preventive chemotherapy in human helminthiasis: coordinated use of anthelminthic drugs in control interventions: a manual for health professionals and programme managers". WHO Press, World Health Organization, Geneva, Switzerland.
- (2012). "A research agenda for helminth diseases of humans: intervention for control and elimination". PLOS Neglected Tropical Diseases.
- (March 2013). "Worms, wisdom, and wealth: why deworming can make economic sense". Trends in Parasitology.
- (March 2008). "Controlling soil-transmitted helminthiasis in pre-school-age children through preventive chemotherapy". PLOS Neglected Tropical Diseases.
- (2004). "Worms: Identifying Impacts on Education and Health in the Presence of Treatment Externalities". Econometrica.
- (2015-08-04). "Economist, World Bank". World Bank.
- (January 2012). "Deworming debunked". BMJ.
- (September 2019). "Public health deworming programmes for soil-transmitted helminths in children living in endemic areas". The Cochrane Database of Systematic Reviews.
- (2015). "When Should Governments Subsidize Health? The Case of Mass Deworming". The World Bank Economic Review.
- Fincham, J., Dhansay, A. (2006). [http://www.susana.org/en/resources/library/details/2110 Worms in SA's children - MRC Policy Brief]. Nutritional Intervention Research Unit of the South African Medical Research Council, South Africa
- "Fact sheets: neglected tropical diseases". WHO Media Centre.
- (2007). "Handbook of Helminthiasis for Public Health". CRC Press, Boca Raton, Florida, US.
- (2012). "A research agenda for helminth diseases of humans: the problem of helminthiases". PLOS Neglected Tropical Diseases.
- WHO. (2013). "Schistosomiasis: progress report 2001 - 2011, strategic plan 2012 - 2020". WHO Press, World Health Organization, Geneva, Switzerland.
- (May 2015). "Malaria". WHO Media Centre.
- (2014). "Foodborne trematode infections". WHO Media Centre.
- (May 2015). "Lymphatic filariasis". WHO Media centre.
- (March 2014). "A global brief on vector-borne diseases". World Health Organization.
- (May 2015). "Echinococcosis". WHO Media Centre.
- (January 2014). "Geohelminths: public health significance". Journal of Infection in Developing Countries.
- Velleman, Y., Pugh, I. (2013). [http://www.susana.org/en/resources/library/details/1794 Under-nutrition and water, sanitation and hygiene - Water, sanitation and hygiene (WASH) play a fundamental role in improving nutritional outcomes. A successful global effort to tackle under-nutrition must include WASH]. WaterAid and Share, UK
- (March 2014). "Dracunculiasis (guinea-worm disease)". WHO Media Centre.
- (2002). "Helminth Control in School-Age Children: A Guide for Managers of Control Programs". World Health Organization.
- (May 2006). "Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm". Lancet.
- Vanalli, Chiara. (25 February 2024). "Helminth ecological requirements shape the impact of climate change on the hazard of infection". Ecology Letters.
- Parmesan, C., M.D. Morecroft, Y. Trisurat, R. Adrian, G.Z. Anshari, A. Arneth, Q. Gao, P. Gonzalez, R. Harris, J. Price, N. Stevens, and G.H. Talukdarr, 2022: [https://www.ipcc.ch/report/ar6/wg2/downloads/report/IPCC_AR6_WGII_Chapter02.pdf Chapter 2: Terrestrial and Freshwater Ecosystems and Their Services]. In [https://www.ipcc.ch/report/ar6/wg2/ Climate Change 2022: Impacts, Adaptation and Vulnerability] [H.-O. Pörtner, D.C. Roberts, M. Tignor, E.S. Poloczanska, K. Mintenbeck, A. Alegría, M. Craig, S. Langsdorf, S. Löschke,V. Möller, A. Okem, B. Rama (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 257–260
- (March 2004). "Common intestinal parasites". American Family Physician.
- (2011). "Patterns and risk factors for helminthiasis in rural children aged under 2 in Bangladesh". South African Journal of Child Health.
- (2013). "Hunter's tropical medicine and emerging infectious diseases". Saunders.
- (2013). "Hunter's tropical medicine and emerging infectious diseases". Saunders.
- (July 2014). "The global burden of disease study 2010: interpretation and implications for the neglected tropical diseases". PLOS Neglected Tropical Diseases.
- (August 2015). "Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013". Lancet.
- (August 2012). "Determining soil-transmitted helminth infection status and physical fitness of school-aged children". Journal of Visualized Experiments.
- (January 2015). "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013". Lancet.
- (July 2013). "Schistosomiasis chemotherapy". Angewandte Chemie.
- (February 1999). "Mortality due to schistosomiasis mansoni: a field study in Sudan". The American Journal of Tropical Medicine and Hygiene.
- (6 June 2011). "Neglected Tropical Diseases".
- (10 January 2013). "Parasites – Soil-transmitted Helminths (STHs)". Centers for Disease Control and Prevention (CDC).
- (March 2006). "Irrigation in developing countries using wastewater.". International Review for Environmental Strategies..
- (2007). "Helminth ova removal from wastewater for agriculture and aquaculture reuse". Water Science and Technology.
- (March 2009). "Application of Helminth ova infection dose curve to estimate the risks associated with biosolid application on soil". Journal of Water and Health.
- (2007). "Encyclopedia of Water Science". CRC Press.
- (March 2015). "The United Nations world water development report 2015: water for a sustainable world.". UNESCO publishing.
- WHO. (1995). "WHO Model Prescribing Information: Drug Use in Parasitic Diseases". WHO.
- WHO (2006). [http://www.susana.org/en/resources/library/details/1004 WHO Guidelines for the Safe Use of Wastewater, Excreta and Greywater – Volume IV: Excreta and greywater use in agriculture] {{Webarchive. link. (2014-10-17 . World Health Organization (WHO), Geneva, Switzerland)
- (2017-05-02). "Parasites – Hookworm". Centers for Disease Control and Prevention.
- (March 2012). "The global burden of neglected tropical diseases". Public Health.
- (December 2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet.
- Luke F. Pennington and Michael H. Hsieh (2014) [http://ebooks.benthamscience.com/book/9781608051489/ Immune Response to Parasitic Infections] {{Webarchive. link. (2014-12-07 , Bentham e books, Vol 2, pp. 93–124, {{ISBN). 978-1-60805-148-9
- Elisabetta Profumo, Alessandra Ludovisi, Brigitta Buttari, Maria, Angeles Gomez Morales and Rachele Riganò (2014) [http://ebooks.benthamscience.com/book/9781608051489/ Immune Response to Parasitic Infections] {{Webarchive. link. (2014-12-07 , Bentham e books, Bentham Science Publishers, Vol 2, pp. 69–91, {{ISBN). 978-1-60805-148-9
- (2005). "Medical Microbiology". Thieme.
- (February 2016). "Dracunculiasis (guinea-worm disease) Fact sheet N°359 (Revised)".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Helminthiasis — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report