From Surf Wiki (app.surf) — the open knowledge base
Ethchlorvynol
Group of stereoisomers
Group of stereoisomers
| Field | Value | |||
|---|---|---|---|---|
| Verifiedfields | changed | |||
| verifiedrevid | 408562693 | |||
| IUPAC_name | 1-chloro-3-ethylpent-1-en-4-yn-3-ol | |||
| image | Ethchlorvynol.svg | |||
| image_class | skin-invert-image | |||
| alt | Structural formula | |||
| image2 | Ethchlorvynol molecule ball.png | |||
| image_class2 | bg-transparent | |||
| alt2 | Ball-and-stick model of the ethchlorvynol molecule | |||
| width | 190 | |||
| pronounce | ||||
| Drugs.com | ||||
| legal_BR | B1 | |||
| legal_BR_comment | ||||
| legal_CA | Schedule IV | |||
| legal_DE | Anlage II | |||
| legal_US | Schedule IV | |||
| legal_UK | Class C | |||
| legal_US_comment | ||||
| routes_of_administration | Oral | |||
| protein_bound | 35–50% | |||
| IUPHAR_ligand | 7180 | |||
| CAS_number_Ref | ||||
| CAS_number | 113-18-8 | |||
| ATC_prefix | N05 | |||
| ATC_suffix | CM08 | |||
| PubChem | 5281077 | |||
| DrugBank_Ref | ||||
| DrugBank | DB00189 | |||
| ChemSpiderID_Ref | ||||
| ChemSpiderID | 4444534 | |||
| UNII_Ref | ||||
| UNII | 6EIM3851UZ | |||
| KEGG_Ref | ||||
| KEGG | D00704 | |||
| ChEMBL_Ref | ||||
| ChEMBL | 591 | |||
| C | 7 | H=9 | Cl=1 | O=1 |
| smiles | Cl[C@H]=CC(C#C)(O)CC | |||
| StdInChI_Ref | ||||
| StdInChI | 1S/C7H9ClO/c1-3-7(9,4-2)5-6-8/h1,5-6,9H,4H2,2H3/b6-5+ | |||
| StdInChIKey_Ref | ||||
| StdInChIKey | ZEHYJZXQEQOSON-AATRIKPKSA-N |
| Drugs.com = | elimination_half-life =
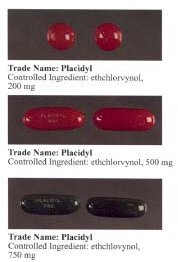
Ethchlorvynol is a GABA-ergic hypnotic sedative medication first developed by Pfizer in the 1950s. In the United States, it was sold by Abbott Laboratories under the trade name Placidyl. Placidyl was available in 200 mg, 500 mg, and 750 mg strength gel filled capsules. While the 500 mg and 750 mg strength capsules were for use in reducing sleep latency, the 200 mg strength capsules were intended to be used to re-induce sleep in case of early awakening. Abbott discontinued production in 1999 due to problems of the widespread abuse of minor tranquilizers, and non-barbiturate sedatives largely being replaced by the benzodiazepine family (with drugs such as Librium and Valium); by the end of 2001, ethchlorvynol reserves had dried up in the United States.
Theoretically, ethchlorvynol could be manufactured for sale in the United States by another pharmaceutical company (subject to FDA approval of such manufacture), though no pharmaceutical company has chosen to do so. Individuals with a valid prescription for the substance may legally transport a reasonable amount of ethchlorvynol with them into the United States. The availability of ethchlorvynol resembles similar barbiturate-like, GABA-ergic sedatives, such as glutethimide (Doriden, which ceased production in U.S. by 1993), ethinamate (Valmid), and methyprylon (vended as Noludar, Noctan, and Dimerin), which were voluntarily withdrawn in favor of benzodiazepine hypnotics in 1965, and supplies widely eliminated by 1969. These are all controlled substances, but are not illegal.
Use and effects

Ethchlorvynol was indicated to treat insomnia, and was widely prescribed in the 1960s and 1970s; prescriptions had fallen significantly by 1990, however, as other hypnotics that were considered safer and less dangerous in overdose became more common.
Availability
Ethchlorvynol is no longer prescribed in the United States due to unavailability, but is still available in some countries, and would still be considered legal to possess and use with a valid prescription.
Adverse effects
Along with expected sedative effects (e.g. relaxation and drowsiness), reported adverse effects of ethchlorvynol include gastrointestinal upset (including nausea and vomiting), dizziness, blurred or altered vision, numbness or tingling, and unsteadiness or impaired coordination. Hypersensitivity reactions such as skin rash have also been reported.
Misuse by injection has been associated with serious acute toxicity. Reports describe severe noncardiogenic pulmonary oedema and haemodynamic effects following intravenous injection of ethchlorvynol (Placidyl), and public health sources have warned of cardiovascular or pulmonary injury arising from improper intravenous or intra-arterial injection of the drug.
Overdose
There is no specific antidote for ethchlorvynol overdose; management is primarily supportive and broadly parallels the approach used for other sedative-hypnotic overdoses (including barbiturate toxicity), with particular attention to airway, breathing, and circulation. Reported features of ethchlorvynol overdose include confusion, slurred speech, unsteady gait or impaired coordination, slow or troubled breathing (respiratory depression), and slow heartbeat, along with severe poisoning that may progress to coma and death. In severe cases, enhanced elimination has been reported (e.g. hemodialysis in a case of massive ingestion), though such interventions are case-dependent and adjunctive to supportive care.
Withdrawal
Ethchlorvynol withdrawal following prolonged or high-dose use has been reported to produce a distinct withdrawal syndrome, particularly after abrupt discontinuation. Case reports describe symptoms including confusion and perceptual disturbances, as well as severe presentations involving psychosis, delirium, and seizures. In some cases, withdrawal symptoms have been reported to occur after a delayed onset following discontinuation. Severe ethchlorvynol withdrawal has been described as resembling withdrawal syndromes associated with other sedative-hypnotic drugs and may carry a risk of serious or potentially fatal complications without appropriate medical management.. Secondary pharmacology references have noted that chronic use of ethchlorvynol may lead to physical dependence, with withdrawal effects comparable to those of barbiturate-class sedative-hypnotics, reinforcing the clinical significance of these reports
Chemistry
Ethchlorvynol is a member of the class of sedative-hypnotic carbinols, which includes methylparafynol and tert-amyl alcohol. It is not a benzodiazepine, carbamate, or barbiturate, and its molecular structure is considerably simpler. The systematic name of ethchlorvynol is usually given as ethyl 2-chlorovinyl ethynyl carbinol or 1-chloro-3-ethylpent-1-en-4-yn-3-ol. Its empirical formula is C7H9ClO.
Ethchlorvynol is synthesized by an ethynylation reaction using lithium acetylide and 1-chloro-1-penten-3-one in liquid ammonia, followed by acidic work-up. :[[File:Ethchlorvynol synthesis.png|class=skin-invert-image|500px]]
The analogous compound consisting of a carbamate derived from the hydroxy group was investigated and was shown to have a slower onset, longer duration, and increased potency, but was not developed commercially.
References
References
- Anvisa. (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial". [[Diário Oficial da União]].
- (January 2014). "Annual Statistical Report on Substances Listed in the Convention on Psychotropic Substances of 1971 Form P". International Narcotics Control Board (INCB).
- "Hypnotic Agent and Method of Making the Same".
- (12 January 2007). "Erowid Ethchlorvynol Vault". Erowid.
- (15 January 1997). "Drugs of abuse". Practice Management Information Corp..
- "Ethchlorvynol (oral route)".
- (January 1976). "Ethchlorvynol (Placidyl)-induced pulmonary edema". Annals of Internal Medicine.
- "The Drugs and Their Effects". Health Canada.
- (2024). "StatPearls [Internet].". StatPearls Publishing.
- "Barbiturate intoxication and overdose".
- (November 1966). "Ethchlorvynol (Placidyl) intoxication and its treatment by hemodialysis". Canadian Medical Association Journal.
- (October 1961). "Withdrawal symptoms following ethchlorvynol (Placidyl) dependence". The American Journal of Psychiatry.
- (June 1964). "Two Cases of Withdrawal from Ethchlorvynol". The American Journal of Psychiatry.
- (October 1964). "Psychosis and Convulsions Following Withdrawal from Ethchlorvynol". JAMA.
- (2018). "Goodman & Gilman's The Pharmacological Basis of Therapeutics". McGraw-Hill Education.
- (1955). "Hypnotics and Anticonvulsants. II. Halogenated Tertiary Acetylenic Carbinols". Journal of Organic Chemistry.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Ethchlorvynol — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report