From Surf Wiki (app.surf) — the open knowledge base
Ergine
Chemical compound
.png)
Chemical compound
| Field | Value | |||
|---|---|---|---|---|
| Watchedfields | verified | |||
| verifiedrevid | 443731816 | |||
| image | Ergine.svg | |||
| image_class | skin-invert-image | |||
| width | 175px | |||
| image2 | Ergine 3D.png | |||
| image_class2 | bg-transparent | |||
| width2 | 200px | |||
| pregnancy_category | X | |||
| routes_of_administration | Oral, intramuscular injection, subcutaneous injection | |||
| class | Serotonin receptor agonist; Serotonergic psychedelic; Hallucinogen; Sedative | |||
| ATC_prefix | None | |||
| legal_AU | ||||
| legal_BR | F2 | |||
| legal_BR_comment | ||||
| legal_CA | ||||
| legal_DE | NpSG | |||
| legal_NZ | ||||
| legal_UK | Class A | |||
| legal_US | Schedule III | |||
| legal_status | Illegal in France | |||
| metabolism | Hepatic | |||
| onset | 0.3–3h (morn. glory seeds) | |||
| duration_of_action | 4–10h (morn. glory seeds) | |||
| excretion | Urine | |||
| CAS_number_Ref | ||||
| CAS_number | 478-94-4 | |||
| UNII_Ref | ||||
| UNII | 073830XH10 | |||
| PubChem | 442072 | |||
| ChemSpiderID_Ref | ||||
| ChemSpiderID | 390611 | |||
| ChEBI_Ref | ||||
| ChEBI | 4819 | |||
| ChEMBL_Ref | ||||
| ChEMBL | 227213 | |||
| synonyms | Lysergic acid amide; LSA; LAA; ᴅ-Lysergic acid amide; ᴅ-Lysergamide, *d*-Lysergamide; LA-111; "Lacy"; 6-Methyl-9,10-didehydroergoline-8β-carboxamide | |||
| IUPAC_name | (6*aR*,9*R*)-7-methyl-6,6a,8,9-tetrahydro-4*H*-indolo[4,3-fg]quinoline-9-carboxamide | |||
| C | 16 | H=17 | N=3 | O=1 |
| SMILES | O=C(N)[C@@H]1C=C2C3=CC=CC4=C3C(C[C@@]2([H])N(C1)C)=CN4 | |||
| StdInChI_Ref | ||||
| StdInChI | 1S/C16H17N3O/c1-19-8-10(16(17)20)5-12-11-3-2-4-13-15(11)9(7-18-13)6-14(12)19/h2-5,7,10,14,18H,6,8H2,1H3,(H2,17,20)/t10-,14-/m1/s1 | |||
| StdInChIKey_Ref | ||||
| StdInChIKey | GENAHGKEFJLNJB-QMTHXVAHSA-N | |||
| melting_point | 135 | |||
| melting_notes | Decomposes |
| elimination_half-life =
Ergine, also known as lysergic acid amide (LSA or LAA) as well as LA-111, is a psychoactive compound of the ergoline and lysergamide families related to lysergic acid diethylamide (LSD). Ergine is an ergoline alkaloid found in fungi such as Claviceps paspali (ergot) and Periglandula species such as Periglandula clandestina, which are permanently connected with many morning glory vines. Ergine induces relatively mild psychedelic effects as well as pronounced sedative effects.
The most common sources of ergine for use as a drug are the seeds of morning glory species including Ipomoea tricolor (tlitliltzin), Ipomoea corymbosa (ololiuhqui), and Argyreia nervosa (Hawaiian baby woodrose). Morning glory seeds have a history of entheogenic use in Mesoamerica dating back at least hundreds of years. They have also since been used by many Westerners. In addition to ergine, morning glory seeds contain other ergolines such as lysergic acid hydroxyethylamide (LSH), lysergic acid propanolamide (ergonovine), and isoergine. Some of these compounds are pharmacologically active and are thought to contribute to the effects of the seeds as well. There has been debate about the role of ergine in causing the psychedelic effects of morning glory seeds.
Ergine was first described by Sidney Smith and Geoffrey Timmis after they isolated it from ergot in 1932. It was first synthesized subsequent to its isolation in the 1930s. Albert Hofmann, the discoverer of LSD's psychedelic effects in 1943, evaluated the effects of ergine in humans in 1947 and described the results many years later. He and his colleagues also isolated ergine from morning glory seeds in 1960. Morning glory seeds started to become frequently used as a recreational drug that same year and have been widely used since. Recreational use of morning glory seeds may be increasing due to their inexpensiveness, widespread availability, and lack of legal restrictions. Ergine has been encountered as a novel designer drug in Europe. Ergine, though not morning glory seeds, has become a controlled substance in various places in the world.
Use
Ergine is most commonly used as a drug in the form of morning glory seeds, including those of Ipomoea tricolor (tlitliltzin), Ipomoea corymbosa (ololiuhqui), and Argyreia nervosa (Hawaiian baby woodrose). They may be consumed whole and intact, crushed or ground up, or drunk as an extract following soaking of the seeds in water. A hallucinogenic dose (~0.5–1mg) is 150 to 200seeds (3–6g) of Ipomoea tricolor (0.02% ergine by dry weight) or 5 to 10seeds (0.5–1g) of Argyreia nervosa (0.14% ergine by dry weight). The onset is 0.3 to 3hours and the duration is 4 to 10hours.
Ergine may be used as a drug in pure or purified form as well, either isolated or synthesized. Albert Hofmann and colleagues found that a 0.5 to 2mg dose by intramuscular or subcutaneous injection produced relatively weak but significant hallucinogenic effects as well as marked sedation. Another study described the effects of pure ergine by injection but the doses were not clearly provided (although appeared to be around 0.1–1mg). Based on the preceding studies, Alexander Shulgin describes pure ergine as having a dosage of 0.5 to 1mg and being 10-fold less potent than LSD, but as being "not hallucinogenic". Hofmann also stated that ergine was 10- to 40-fold less potent than LSD and that it had qualitatively different effects. Robert Oberlender has stated that ergine is about 30-fold less potent than LSD in humans. Heim and colleagues assessed ergine at higher doses of 3 to 6mg orally and observed toxic-like effects, whereas isoergine at 2 to 5mg orally produced notable hallucinogenic effects, including some euphoria, synaesthesia, and altered time perception.
Per Shulgin in his 1997 book TiHKAL (Tryptamines I Have Known and Loved) however, both ergine and isoergine are "probably correctly dismissed" as not contributing to the effects of morning glory seeds. The poorly-stable lysergic acid hydroxyethylamides (LSHs) might alternatively be involved in the psychedelic effects of morning glory seeds per Shulgin.
Sleepy grass (Achnatherum robustum) and Claviceps paspali (ergot) have similar ergoline constituents as morning glory seeds and have also been used to produce psychoactive effects, albeit rarely.
Effects
Subjective effects
Ergine has only been given a minuscule amount of attention. Albert Hofmann and his colleagues self-administered ergine. In addition, it was assessed in two clinical studies by other researchers. Synthetic ergine was used in all of these cases. Hofmann stated that ergine induces a "psychotomimetic" effect with "a marked narcotic component": "Tired, dreamy, incapable of clear thoughts. Very sensitive to noises which give an unpleasant sensation." There are parallels between Hofmann's comments and the ones in the two trials:
| Hofmann (1963) | Heim et al. (1968) | Solms (1956) |
|---|---|---|
| "dysphoria" | "irritative depressive moods" | |
| "incapable of clear thoughts" | "impairment of concentration" | "impaired concentration" |
| "Desire to lie down and sleep. Genuine physical and mental tiredness, which is not experienced as an unpleasant sensation. Slept for 3 hours." | "test subject SB [...] had to go to bed after an antineoplastic injection and did not recover until the following day." | "and an immediate desire to sleep, after which he slept for three hours during the day". |
| "a feeling of mental emptiness and of the unreality and complete meaninglessness of the outside world". [isoergine] | "In the test subject PS (5 mg), severe nausea with a drop in blood pressure suddenly occurred after 3½ hours, which was controlled with analeptics and antinausea after about 30 minutes. At the same time, the test subject experienced a feeling of total annihilation and fear of death, which subsided after vomiting about 60 minutes later, but only completely subsided during the course of the night." [isoergine] | "Indifference" |
Heim 1968 also noted "paraesthesia", "synesthesia" and an "overestimation of the time that had passed" (isoergine), but also concluded, "our experiments with ᴅ-lysergic acid amide also confirm the results that Sᴏʟᴍꜱ had made with this substance, namely a predominantly sedative intoxication." Hofmann emphasized this sedative effect:
"Furthermore there is not only a quantitative difference between the principles of Ipomoea [tricolor] and Turbina corymbosa and LSD; there is likewise a qualitative one, LSD being a very specific hallucinogen, whereas the psychic effects of lysergic acid amide and the total alkaloids of these two plants are characterized by a pronounced narcotic component (Hofmann, 1968)."
"A substance very closely related to LSD, the monoethylamide of lysergic acid (LAE-32), in which an ethyl group is replaced by a hydrogen atom on the diethylamide residue of LSD, proved to be some ten times less psychoactive than LSD. The hallucinogenic effect is also qualitatively different: it is characterized by a narcotic component. This narcotic effect is yet more pronounced in lysergic acid amide (LA-111), in which both ethyl groups of LSD are displaced by hydrogen atoms. These effects, which I established in comparative self-experiments with LA-111 and LAE-32, were corroborated by subsequent clinical investigations."
"The experience had some strong narcotic effect, but at the same time there was a very strange sense of voidness. In this [void], everything loses its meaning. It is a very mystical experience."
Physiological effects
While its physiological effects vary from person to person, the following symptoms have been attributed to the consumption of ergine or ergine containing seeds: sedation, visual hallucinations, auditory hallucinations, euphoria, loss of motor control, nausea, vasoconstriction, delusions, anxiety, paranoia, and irregular heartbeats.
One study found that two of four human subjects experienced cardiovascular dysregulation and the study had to be halted, concluding that the ingestion of seeds containing ergine was less safe than commonly believed. Importantly this may have been a product of other substances within the seeds. The same study also observed that reactions were highly differing in type and intensity between different subjects.
Side effects
A 2016 study showed that penniclavine was the predominant alkaloid in Ipomoea tricolor seeds. Ergoclavines are known to cause convulsive ergotism, the milder form of ergotism. Gangrenous ergotism is caused by ergopeptines: the complex peptide moiety forces persistence at the receptor sites. Ergopeptines are rare in Convolvulaceae, being found in 10 species, not including the three that are commonly ingested, although Paulke 2014 says analytical evidence suggests that A. nervosa contains ergopeptines. Many people desire purified seed extracts, but the efficacy of this is questionable, as even pure ergine and ergonovine have shown toxic effects.
The side effects of ergine have been described as follows: "The expression and behavior of the test subjects changed just 45 minutes after taking the substance: the test subjects appeared to be suffering, their facial expressions were deteriorating as if they had suffered a serious illness, and their movements were noticeably slower. [...] In the self-reports of both test subjects, complaints about vegetative symptoms predominated: unpleasant, flu-like feeling of illness, nausea, sudden onset of nausea, with vomiting that could be stopped with 2 cm3 of Cyclicinum hydrochloricum. In addition, sensations of heat, sweating, dizziness, a feeling of heaviness and general tiredness were observed."
And the side effects of ergonovine have been described as follows: "Walking in this dreamy state was difficult due to leg cramps and slight incoordination. There was always a great desire to lie supine. [...] One of us (J.B.) felt the cramping in the legs as painful and debilitating. [...] We all had a slight hangover the following morning. [...] The mild entheogenic effects of ergonovine are similar to those of LSD. However, in dramatic contrast to LSD, the somatic effects of ergonovine greatly overshadow its psychic effects, so much so that we had no wish to ingest more than 10.0 mg, [...]".
Like other psychedelics, ergine is not considered to be addictive. Additionally, there are no known deaths directly associated with pharmacological effects of ergine consumption. All associated deaths are due to indirect causes, such as self-harm, impaired judgement, and adverse drug interactions. One known case involved a suicide that was reported in 1964 after ingestion of morning glory seeds. Another instance is a death due to falling off of a building after ingestion of Hawaiian baby woodrose seeds and alcohol. A study gave mice 3000mg/kg with no lethal effects.
Chemical coatings on seeds
Garden seeds, in general, may be coated with fungicides et. al. (e.g. neonicotinoids, Thiram, and ApronMaxx). It is rumored that this is the cause of the severe adverse effects that have been observed, but the seeds, themselves, contain toxins, specifically glycoresins and ergoclavines. Some even believe that an emetic chemical is purposely added to the seeds to prevent people from ingesting them, but that has never been proven. One 1964 article states that reported adverse effects must come from the seeds, as the stated insecticide is too "inocuous" to humans to be responsible.
A related rumor is that the seeds contain cyanogenic glycosides. The UseNet post on which this is based contains two references, but neither of them support that claim, and Eckart Eich says that they probably don't occur in many Convolvulaceae. There is a similar claim in a publication from 1973, warning about "a strychnine-like alkaloid", but that is probably just a misapplication of the claim that peyote contains strychnine, which, itself, is a rumor.
Overdose
Cases of overdose of ergine and morning glory seeds and associated toxicity have been reported.
Interactions
The interactions of ergine and of morning glory seeds have been discussed.
Pharmacology
Pharmacodynamics
| Receptor | Affinity (Ki, nM) | Ratio | Ergine (LSA) | LSD |
|---|---|---|---|---|
| [5-HT1A](5-ht1a-receptor) | 73 | 1.6 | 46:1 | |
| [5-HT2A](5-ht2a-receptor) | 28–132 (Ki) | |||
| 1.1–58 () | ||||
| 47–82% () | 0.87–1.1 (Ki) | |||
| 0.52–3.6 (EC50) | ||||
| 60–86% (Emax) | 32–120:1 | |||
| 2–16:1 | ||||
| 0.78–0.95:1 | ||||
| [5-HT2B](5-ht2b-receptor) | 54–115 (EC50) | |||
| 40–55% (Emax) | 0.68–34 (EC50) | |||
| 62–73% (Emax) | 3–79:1 | |||
| 0.65–0.75:1 | ||||
| [5-HT2C](5-ht2c-receptor) | 798 | 7.8 | 102:1 | |
| D1 (pig) | 832 | 87 | 10:1 | |
| D2L | 891 | 155 | 6:1 | |
| D2S | 145 | 25 | 6:1 | |
| D3 | 437 | 65 | 7:1 | |
| D4.4 | 141 | 30 | 5:1 | |
| α1 | 912 | 60 | 15:1 | |
| α2 | 62 | 1.0 | 62:1 | |
| **Notes:** All proteins are human unless otherwise specified. **Sources:** |
Ergine interacts with serotonin, dopamine, and adrenergic receptors similarly to but with lower affinity than lysergic acid diethylamide (LSD). It is known to act as an agonist of the serotonin 5-HT2A and 5-HT2B receptors similarly to LSD, albeit much less potently and with reduced activational efficacy. The drug has about 4.3% of the antiserotonergic activity of LSD in the isolated rat uterus in vitro. The psychedelic effects of ergine can be attributed to activation of serotonin 5-HT2A receptors.
Chemistry
Ergine, also known as lysergic acid amide (LSA) or as lysergamide, is a ergoline and lysergamide. It is the simplest lysergamide and is the parent structure of this family of compounds. Hence, all lysergamides are derivatives of ergine. Lysergic acid diethylamide (LSD) is the analogue of ergine with two ethyl groups substituted on its amide moiety.
The extraction of ergine from morning glory seeds has been described.
Natural occurrence
Ergine is not a biosynthetic endpoint itself, but rather a hydrolysis product of lysergic acid hydroxyethylamide (LSH), lysergic acid propanolamide (ergonovine), and ergopeptines or their ergopeptam precursors.
LSH is very vulnerable to this hydrolysis, and many analyses of ergoline-containing products show little to no LSH and substantial amounts of ergine.
An ergine analog, 8-hydroxyergine, has also been found in natural products in two studies. Methylergonovine and methysergide (1-methylmethylergonovine) have also been found in a natural product in one study; these are documented as semisynthetic compounds, so the findings need to be repeated for certainty. The aforementioned chemicals are the only natural lysergamides.
LSH and ergine are predominant in Claviceps paspali, but are only found in trace amounts in the more well-known Claviceps purpurea. Both are ergot-spreading fungi. The major products of C. purpurea are ergopeptines, but C. paspali does not generate ergopeptines. Ergonovine is the only lysergamide in C. purpurea in a substantial amount.
LSH and ergine are also found in the related fungi, Periglandula, which are permanently connected with Ipomoea tricolor, Ipomoea corymbosa, Argyreia nervosa ("morning glory", coaxihuitl, Hawaiian baby woodrose), and an estimated over 440 other Convolvulaceae (ergolines have been identified in 42 of these plants and not all of them contain ergine). Ergonovine is present in Ipomoea tricolor in one-tenth to one-third of the amount of ergine. This variable may account for the varying reports about the psychedelic effect of these seeds.
Other fungi that have been found to contain LSH and/or ergine:
- Unidentified Acremonium species that infects sleepy grass (C. purpurea also infects sleepy grass).
- Unidentified Acremonium species that infects drunken horse grass
- Acremonium coenophialum (infects Festuca arundinacea)
- Epichloë gansuensis var. inebriens (infects drunken horse grass)
- Metarhizium brunneum
- Metarhizium acridum
- Metarhizium anisopliae
- Metarhizium flavoviride
- Metarhizium robertsii
- Aspergillus leporis
- Aspergillus homomorphus
- Aspergillus hancockii
All of these fungi are related to Claviceps fungi. Aspergillus is considered to be a more distant relative of Claviceps.
Other fungi that possibly contain ergine (i.e. they have been found to contain ergonovine and/or ergopeptines):
- Claviceps hirtella
- Neotyphodium lolii
- Unidentified Epichlöe and Neotyphodium (asexual forms of Epichlöe) species
- Aspergillus fumigata
- Aspergillus flavus
- Botritis fabae
- Curvularia lunata
- Geotrichum candidum
- Balansia cyperi
- Balansia claviceps
- Balansia epichloë
- Epichloë amarillans
- Epichloë cabralii (H)
- Epichloë canadensis (H)
- Epichloë coenophiala (H)
- Epichloë festucae
- Epichloë festucae var. lolii
- Epichloë festucae var. lolii x E. typhina (H)
- Epichloë inebriens
- Epichloë glyceriae
- Epichloë mollis
- Epichloë typhina
- Epichloë typhina ssp. poae
- Epichloë typhina ssp. clarkii See table 3 on p. 1290.
- Epichloësp. AroTG-2(H)
- Epichloë sp. FaTG-2(H)
- Epichloë sp. FaTG-4(H)
- Hypomyces aurantius
- Sepedonium sp.
- Cunnigbamella blakesleana
- Mucor biemalis
- Rhizopus nigricans
Biosynthesis
.png)
The biosynthetic pathway to ergine starts like most other ergoline alkaloid- with the formation of the ergoline scaffold. This synthesis starts with the prenylation of L-tryptophan in an SN1 fashion with dimethylallyl diphosphate (DMAPP) as the prenyl donor and catalyzed by prenyltransferase 4-dimethylallyltryptophan synthase (DMATS), to form 4-L-dimethylallyltryptophan (4-L-DMAT). The DMAPP is derived from mevalonic acid. A three strep mechanism is proposed to form 4-L-DMAT: the formation of an allylic carbocation, a nucleophilic attack of the indole nucleus to the cation, followed by deprotonation to restore aromaticity and to generate 4-L-DMAT. 4-Dimethylallyltyptophan N-methyltransferase (EasF) catalyzes the N-methylation of 4-L-DMAT at the amino of the tryptophan backbone, using S-Adenosyl methionine (SAM) as the methyl source, to form 4-dimethylallyl-L-abrine (4-DMA-L-abrine). The conversion of 4-DMA-L-abrine to chanoclavine-I is thought to occur through a decarboxylation and two oxidation steps, catalyzed by the FAD dependent oxidoreductase, EasE, and the catalase, EasC. The chanoclavine intermediate is then oxidized to chanoclavine-l-aldehyde, catalyzed by the short-chain dehydrogenase/reductase (SDR), EasD.

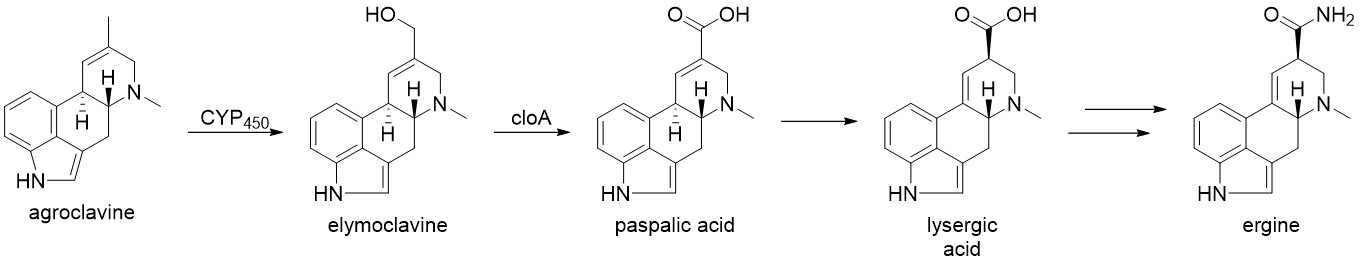
From here, the biosynthesis diverges and the products formed are plant and fungus-specific. The biosynthesis of ergine in Claviceps purpurea will be exemplified, in which agroclavine is produced following the formation of chanoclavine-l-aldehyde, catalyzed by EasA through a keto-enol tautomerization to facilitate rotation about the C-C bond, followed by tautomerization back to the aldehyde and condensation with the proximal secondary amine to form an iminium species, which is subsequently reduced to the tertiary amine and yielding agroclavine. Cytochrome P450 monooxygenases (CYP450) are then thought to catalyze the formation of elymoclavine from argoclavine via a 2 electron oxidation. This is further converted to paspalic acid via a 4 electron oxidation, catalyzed by cloA, a CYP450 monooxygenase. Paspalic acid then undergoes isomerization of the C-C double bond in conjugation with the acid to form D-lysergic acid. While the specifics of the formation of ergine from D-lysergic acid are not known, it is proposed to occur through a nonribosomal peptide synthase (NRPS) with two enzymes primarily involve: D-lysergyl peptide synthase (LPS) 1 and 2.
History
Ergine was first obtained by Sidney Smith and Geoffrey Willward Timmis in 1932.
Albert Hofmann was first to identify ergine as a natural constituent of Turbina corymbosa seeds.
Albert Hofmann describes ergine as "the main constituent of ololiuhqui". Ololiuhqui was used by South American healers in shamanic healing ceremonies. Similarly, ingestion of morning glory seeds by Mazatec tribes to "commune with their gods" was reported by Richard Schultes in 1941 and is still practiced today.
According to the ethnobotanist R. Gordon Wasson, Thomas MacDougall and Francisco Ortega ("Chico"), a Zapotec guide and trader, should be credited for the discovery of the ceremonial use of Ipomoea tricolor seeds in Zapotec towns and villages in the uplands of southern Oaxaca. The seeds of both Ipomoea tricolor and Rivea corymbosa, another species which has a similar chemical profile, are used in some Zapotec towns.
The Central Intelligence Agency conducted research on the psychedelic properties of Rivea corymbosa seeds for MKULTRA.
Hofmann's discovery of ergine and related compounds in morning glory seeds, which are closely structurally related to LSD, was said to have initially been met with "a state of disbelief bordering on accusations of scientific fraud", but was soon confirmed by other researchers.
Society and culture
Legal status
The legality of consuming, cultivating, and possessing ergine varies depending on the country.
Australia
In most Australian states, the consumption of ergine containing materials is prohibited under state legislation.
Canada
In Canada, ergine is not illegal to possess as it is not listed under Canada's Controlled Drugs and Substances Act, though it is likely illegal to sell for human consumption.
New Zealand
In New Zealand, ergine is a controlled drug, however the plants and seeds of the morning glory species are legal to possess, cultivate, buy, and distribute.
United Kingdom
Ergine is considered a Class A substance in the United Kingdom, categorized as a precursor to LSD.
United States
There are no laws against possession of ergine-containing seeds in the United States. However, possession of the pure compound without a prescription or a DEA license would be prosecuted, as ergine, under the name "lysergic acid amide", is listed under Schedule III of the Controlled Substances Act.
References
References
- "Erowid Morning Glory Basics". Erowid.org.
- Anvisa. (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial". [[Diário Oficial da União]].
- (20 May 2021). "Arrêté du 20 mai 2021 modifiant l'arrêté du 22 février 1990 fixant la liste des substances classées comme stupéfiants".
- (January 2015). "Recreational use, analysis and toxicity of tryptamines". Curr Neuropharmacol.
- (August 2012). "Analysis of lysergic acid amide in human serum and urine after ingestion of Argyreia nervosa seeds". Anal Bioanal Chem.
- (October 1993). "Vasoconstriction in bovine vasculature induced by the tall fescue alkaloid lysergamide". Journal of Animal Science.
- (1966). "Alkaloids and Lipids of Ipomoea, Rivea and Convolvulus and Their Application to Chemotaxonomy". Economic Botany.
- (November 1966). "Changes in ergoline alkaloids in seeds during ontogeny of Ipomoea violacea". Journal of Pharmaceutical Sciences.
- (September 1965). "A direct densitometric method on thin-layer plates for the determination of lysergic acid amide, isolysergic acid amide and clavine alkaloids in morning glory seeds". Journal of Chromatography.
- (1978-01-01). ""Legal highs"--constituents, activity, toxicology, and herbal folklore". Clinical Toxicology.
- (August 2021). "Pharmacologic Similarities and Differences Among Hallucinogens". J Clin Pharmacol.
- (July 2018). "Ergot Alkaloids and their Hallucinogenic Potential in Morning Glories". Planta Med.
- (Fall 2001). "Stanislav Grof Interviews Dr. Albert Hofmann". MAPS Bulletin.
- (April 2025). "A new species of Periglandula symbiotic with the morning glory Ipomoea tricolor". Mycologia.
- (1975). "Psychotomimetics of the Convolvulaceae". Prog Med Chem.
- (2013). "International Handbook of Psychiatry". World Scientific.
- (2000). "Mixing the Kykeon". ELEUSIS: Journal of Psychoactive Plants and Compounds.
- (2009). "LSD My Problem Child: Reflections on Sacred Drugs, Mysticism, and Science". MAPS.org.
- (January–March 1971). "Teonanácatl and Ololiuqui, two ancient magic drugs of Mexico". Bulletin on Narcotics.
- (January 1969). "Hallucinogens of plant origin". Science.
- (1963). "The Active Principles of the Seeds of Rivea Corymbosa and Ipomoea Violacea". Harvard University Herbaria.
- (1993). "The Nature of Shamanism: Substance and Function of a Religious Metaphor". State University of New York Press.
- (Winter 1999). "More Stipa Robusta". Entheogen Review.
- (1955). "Amide der stereoisomeren Lysergsäuren und Dihydro-lysergsäuren. 38. Mitteilung über Mutterkornalkaloide". Helvetica Chimica Acta.
- (1967). "The Hallucinogens". Elsevier.
- (1960). "Isolierung von Lysergsäure-Alkaloiden aus der mexikanischen Zauberdroge Ololiuqui (Rivea corymbosa (L.) Hall. f.)". Experientia.
- (2013-01-01). "Recreational use of D-lysergamide from the seeds of Argyreia nervosa, Ipomoea tricolor, Ipomoea violacea, and Ipomoea purpurea in Poland". Journal of Psychoactive Drugs.
- (2013-09-01). "Drogas emergentes (III): plantas y hongos alucinógenos". Anales del Sistema Sanitario de Navarra.
- (2008). "EMCDDA–Europol 2008 Annual Report on the implementation of Council Decision 2005/387/JHA".
- (May 2004). "Hallucinogens and dissociative agents naturally growing in the United States". Pharmacol Ther.
- (2020). "Risk assessment of Argyreia nervosa".
- "Les Psychodysleptiques". Produits et Problèmes Pharmaceutiques [Prod. Probl. Pharm.].
- [[Albert Hofmann]]. (1970). "Discoveries in Biological Psychiatry". J.B. Lippincott Company.
- (1956). "Relationships between chemical structure and psychoses with the use of psychotoxic substances; comparative pharmacopsychiatric analysis: a new research method". Journal of Clinical and Experimental Psychopathology.
- (August 1956). "Chemische Struktur und Psychose bei Lysergsäure-Derivaten". Praxis.
- (2003). "Hallucinogens: A Forensic Drug Handbook". Elsevier Science.
- (1975). "Hallucinogenic Agents". Wright-Scientechnica.
- {{CiteTiHKAL https://www.erowid.org/library/books_online/tihkal/tihkal26.shtml "LA-111, Ergine, d-Lysergamide. This is an active compound and has been established as a major component in morning glory seeds. It was assayed for human activity, by Albert Hofmann in self-trials back in 1947, well before this was known to be a natural compound. An i.m. administration of a 500 microgram dose led to a tired, dreamy state with an inability to maintain clear thoughts. [...] The epimer, inverted at C-8, is isoergine or d-isolysergamide, and is also a component of morning glory seeds. Hofmann tried a 2 milligram dose of this amide, and as with ergine, he experienced nothing but tiredness, apathy, and a feeling of emptiness. Both compounds are probably correctly dismissed as not being a contributor to the action of these seeds. It is important to note that ergine, as well as lysergic acid itself, is listed as a Schedule III drug in the Controlled Substances Act, as a depressant. This is, in all probability, a stratagem to control them as logical precursors to LSD. [...] Although there are many other chemical treasures in the ergot fungal world, I would like to wrap this commentary up with a return to the topic of morning glory seeds. Four additional alkaloids of the ergot world must be acknowledged as being potentially participating factors in the MGS story. [...] These structures in effect define absolute obscurity, and most probably do not contribute to the morning glory intoxication state. But the others, some present is sizable amounts, may someday help explain why the pharmacology of these seeds is so different than that of the major isolates, the ergines."
- (1972). "Chemical and Biological Aspects of Drug Dependence". CRC Press.
- (1994). "Psychedelic shamanism: the cultivation, preparation and shamanic use of psychotropic plants". Loompanics Unlimited.
- (17 August 2013). "Ergot Wine Revisited - YouTube". Neurosoup.
- (1968). "Die psychische Wirkung der mexikanischen Droge "Ololiuqui" am Menschen". Psychopharmacologia.
- (1980). "LSD, My Problem Child". McGraw-Hill.
- (January 2012). "Variable adverse effects in subjects after ingestion of equal doses of Argyreia nervosa seeds". Forensic Science International.
- (2006). "Ergot alkaloids--biology and molecular biology". Elsevier.
- (January 12, 2008). "Solanaceae and convolvulaceae - secondary metabolites: biosynthesis, chemotaxonomy, biological and economic significance: a handbook". Springer-Verlag.
- (July 2013). "Differential allocation of seed-borne ergot alkaloids during early ontogeny of morning glories (Convolvulaceae)". Journal of Chemical Ecology.
- (April 2015). "Studies on the alkaloid composition of the Hawaiian Baby Woodrose Argyreia nervosa, a common legal high". Forensic Science International.
- (January 1979). "Entheogenic effects of ergonovine". Journal of Psychedelic Drugs.
- (April 1964). "Suicide Following Morning Glory Seed Ingestion". The American Journal of Psychiatry.
- (April 2010). "Two cases of lysergamide intoxication by ingestion of seeds from Hawaiian Baby Woodrose". Forensic Science International.
- (2013-10-14). "Chemistry in Botanical Classification: Medicine and Natural Sciences: Medicine and Natural Sciences". Elsevier.
- (October 2017). "Resin glycosides from Convolvulaceae plants". Journal of Natural Medicines.
- (December 1964). "Morning Glory Seed Reaction". JAMA.
- (1963). "Pesticide Handbook". College Science Publishers.
- Peter Jordan. Re: Woodrose vs Ipomoea. alt.drugs, UseNet, 10/1/1994 https://erowid.org/plants/hbw/hbw_info1.shtml
- (1973). "The First Book of Sacraments of the Church of the Tree of Life: A Guide for the Religious Use of Legal Mind Alterants". Tree of Life Press.
- (28 September 2010). "Pharmako/Gnosis: Plant Teachers and the Poison Path". North Atlantic Books.
- (July 2013). "Argyreia nervosa (Burm. f.): receptor profiling of lysergic acid amide and other potential psychedelic LSD-like compounds by computational and binding assay approaches". Journal of Ethnopharmacology.
- (January 2017). "Crystal Structure of an LSD-Bound Human Serotonin Receptor". Cell.
- (16 January 2013). "Mapping the binding site of the 5-HT2A receptor using mutagenesis and ligand libraries: Insights into the molecular actions of psychedelics".
- (May 1989). "Stereoselective aspects of hallucinogenic drug action and drug discrimination studies of entactogens". Purdue University.
- (January 1958). "Comparative Study on the Serotonin Antagonism of Amide Derivatives of Lysergic Acid and of Ergot Alkaloids". The Journal of Pharmacology and Experimental Therapeutics.
- (2020). "Handbook of the Behavioral Neurobiology of Serotonin".
- (January 2022). "Psychedelics". Curr Biol.
- (2018). "Chemistry and Structure-Activity Relationships of Psychedelics".
- (April 2001). "Clandestine Extraction of Lysergic Acid Amide (LSA) From Morning Glory Seeds". Journal of the Clandestine Laboratory Investigating Chemists Association.
- Soma Graphics. (January 1996). "Simple Extraction Methods of LA-111 From Argyreia and Ipomoea Species". Soma Graphics.
- (1982-02-19). "Separation of four isomers of lysergic acid α-hydroxyethylamide by liquid chromatography and their spectroscopic identification". Journal of Chromatography A.
- (1968). "Chemistry of alkaloid formation in ergot". Lloydia.
- (September 1973). "Ergot alkaloids. IV. Contribution to the biosynthesis of lysergic acid amides". Folia Microbiologica.
- (1 October 2003). "Biochemical outcome of blocking the ergot alkaloid pathway of a grass endophyte". Journal of Agricultural and Food Chemistry.
- (2010). "The Mycota, Industrial Applications". Springer-Verlag.
- (1976). "Psychopharmacological Agents: Use, Misuse and Abuse". Academic Press.
- (1973). "The Botany and Chemistry of Hallucinogens". Charles Thomas.
- (September 1, 1989). "New Alkaloids of Claviceps paspali". Journal of Natural Products.
- (March–April 1992). "Alkaloids of Stipa robusta (sleepygrass) infected with an Acremonium endophyte". Natural Toxins.
- (July 1960). "Production of lysergic acid derivatives by a strain of Claviceps paspali Stevens and Hall in submerged culture". Nature.
- (April 1970). "Biosynthesis of N-(alpha-hydroxyethyl) lysergamide, a metabolite of Claviceps paspali Stevens and Hall". The Biochemical Journal.
- (1969). "Biosynthesis of ergot alkaloids. Lysergylalanine as precursor of amide-type alkaloids". J. Chem. Soc. D.
- (1973). "The Botany and Chemistry of Hallucinogens". Charles Thomas.
- (November 25, 2008). "The Road to Eleusis: Unveiling the Secret of the Mysteries". North Atlantic Books.
- (February 3, 2018). "Physiology and Genetics". Springer International Publishing.
- (May 2016). "Identification and determination of ergot alkaloids in Morning Glory cultivars". Analytical and Bioanalytical Chemistry.
- (January 2004). "A Reevaluation of the Host Range and Geographical Distribution of Claviceps Species in the United States". Plant Disease.
- (1996-05-16). "High Levels of Ergonovine and Lysergic Acid Amide in Toxic Achnatherum inebrians Accompany Infection by an Acremonium -like Endophytic Fungus". Journal of Agricultural and Food Chemistry.
- (1991-01-09). "Naturally Occurring Pest Bioregulators". American Chemical Society.
- (July 2015). "Two distinct Epichloë species symbiotic with Achnatherum inebrians, drunken horse grass". Mycologia.
- (July 2020). "Several Metarhizium Species Produce Ergot Alkaloids in a Condition-Specific Manner". Applied and Environmental Microbiology.
- (November 2021). "Independent Evolution of a Lysergic Acid Amide in Aspergillus Species". Applied and Environmental Microbiology.
- (2009-10-01). "The ergot alkaloid gene cluster: functional analyses and evolutionary aspects". Phytochemistry.
- (2004-06-02). "Symbioses of grasses with seedborne fungal endophytes". Annual Review of Plant Biology.
- (2006). "Ergot: The Genus Claviceps". Harwood Academic Publishers.
- (2013-02-28). "Plant-symbiotic fungi as chemical engineers: multi-genome analysis of the clavicipitaceae reveals dynamics of alkaloid loci". PLOS Genetics.
- (October 2014). "Interspecific hybridization and bioactive alkaloid variation increases diversity in endophytic Epichloë species of Bromus laevipes". FEMS Microbiology Ecology.
- (June 2013). "Currencies of mutualisms: sources of alkaloid genes in vertically transmitted epichloae". Toxins.
- (Sep–Oct 2012). "Epichloe canadensis, a new interspecific epichloid hybrid symbiotic with Canada wildrye (Elymus canadensis)". Mycologia.
- (August 2012). "Genotypic and chemotypic diversity of Neotyphodium endophytes in tall fescue from Greece". Applied and Environmental Microbiology.
- (June 1993). "Extracellular glutathione is a source of cysteine for cells that express gamma-glutamyl transpeptidase". Biochemistry.
- (April 2007). "A complex ergovaline gene cluster in epichloe endophytes of grasses". Applied and Environmental Microbiology.
- (2011-01-01). "Abundant degenerate miniature inverted-repeat transposable elements in genomes of epichloid fungal endophytes of grasses". Genome Biology and Evolution.
- (October 2001). "Elimination of ergovaline from a grass-Neotyphodium endophyte symbiosis by genetic modification of the endophyte". Proceedings of the National Academy of Sciences of the United States of America.
- (January 2015). "Alkaloid variation among epichloid endophytes of sleepygrass (Achnatherum robustum) and consequences for resistance to insect herbivores". Journal of Chemical Ecology.
- (1993-09-01). "Taxonomy of Acremonium endophytes of tall fescue (Festuca arundinacea), meadow fescue (F. pratensis) and perennial ryegrass (Lolium perenne)". Mycological Research.
- (2014-11-04). "Characterization of Epichloë coenophiala within the US: are all tall fescue endophytes created equal?". Frontiers in Chemistry.
- (December 2014). "Biosynthetic pathways of ergot alkaloids". Toxins.
- (1983-07-01). "Unusual Ergot Alkaloid Biosynthesis in Sclerotia of a Claviceps purpurea Mutant". Microbiology.
- (1932). "98. The alkaloids of ergot. Part III. Ergine, a new base obtained by the degradation of ergotoxine and ergotinine". Journal of the Chemical Society (Resumed).
- (November 25, 2008). "The Road to Eleusis: Unveiling the Secret of the Mysteries". North Atlantic Books.
- (2008). "Unauthorized research on cluster headache.". The Entheogen Review.
- (1941). "A Contribution to Our Knowledge of Rivea Corymbosa: The Narcotic Ololinqui of the Aztecs". Botanical Museum of Harvard University.
- (1961). "The Hallucinogenic Fungi Of Mexico: An Inquiry Into The Origins of The Religious Idea Among Primitive Peoples".
- (3 November 1956). "Project Mkultra, Subproject 22 (w/attachments)".
- (1996). "Pharmacotheon: Entheogenic Drugs, Their Plant Sources and History". Natural Products Co..
- "Erowid LSA Vault : Legal Status".
- "Controlled Drugs and Substances Act".
- "Initial schedules of controlled substances (Schedule III), Section 812".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Ergine — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report