From Surf Wiki (app.surf) — the open knowledge base
Epsomite
Sulfate mineral
Sulfate mineral
| Field | Value |
|---|---|
| name | Epsomite |
| category | Sulfate mineral |
| image | Epsomita 01.jpg |
| caption | Epsomite formation from a Calatayud, Spain cave |
| formula | |
| IMAsymbol | Esm |
| strunz | 7.CB.40 |
| dana | 29.6.11.1 |
| system | Orthorhombic |
| class | Disphenoidal (222) |
| H-M symbol: (2 2 2) | |
| symmetry | *P*2₁2₁2₁ |
| unit cell | a = 11.86, b = 11.99 |
| c = 6.858 [Å]; | |
| color | White, grey, colorless, or pink, greenish |
| habit | Acicular to fibrous encrustations |
| twinning | Rarely observed on {110} |
| cleavage | {010} perfect {101} distinct |
| fracture | Conchoidal |
| mohs | 2 |
| luster | Vitreous, silky when fibrous |
| diaphaneity | Transparent to translucent |
| gravity | 1.67–1.68 |
| opticalprop | Biaxial (−) |
| refractive | n = 1.433 n = 1.455 n = 1.461 |
| birefringence | δ = 0.028 |
| 2V | Measured: 52° |
| solubility | In water |
| alteration | Dehydrates in dry air |
| references |
H-M symbol: (2 2 2) c = 6.858 [Å]; | length fast/slow =
Epsomite, Epsom salt, or magnesium sulfate heptahydrate, is a hydrous magnesium sulfate mineral with formula .
Physical properties
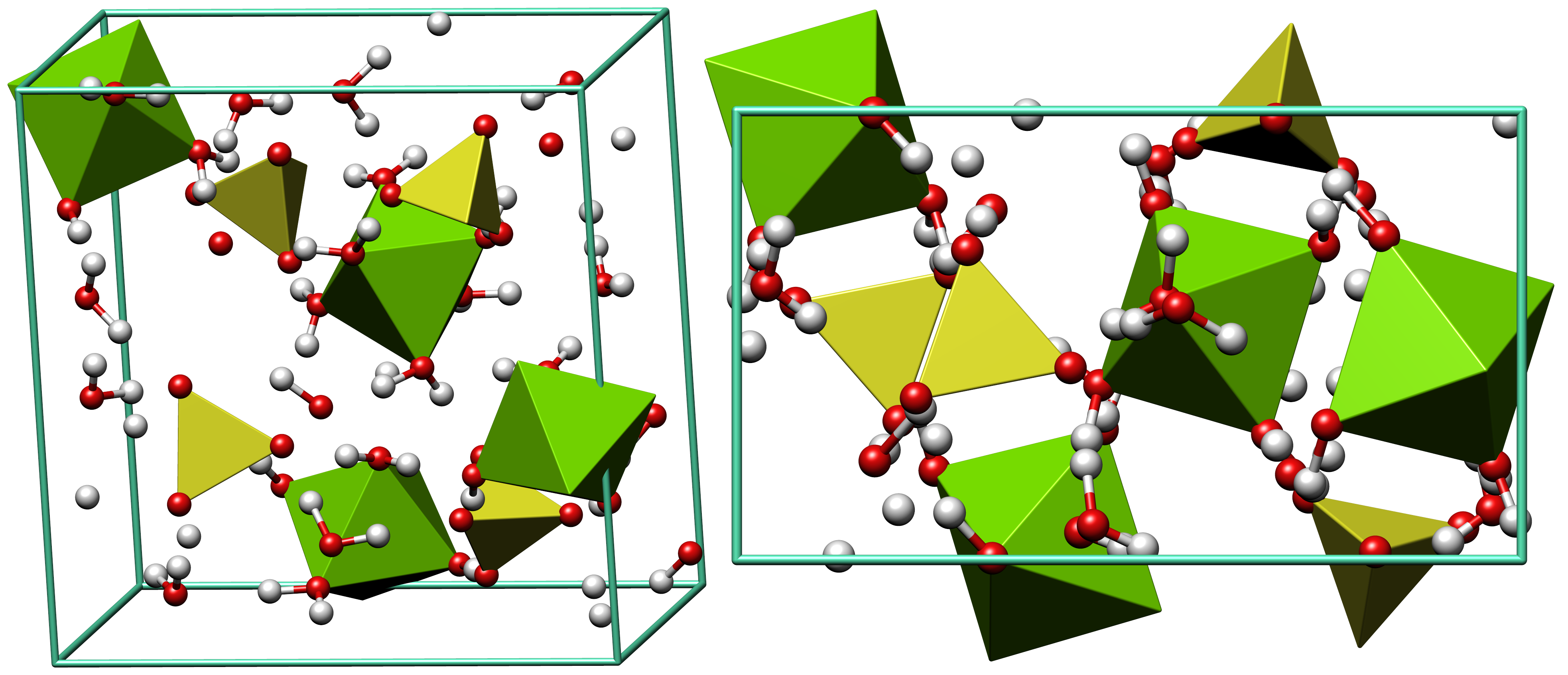
Epsomite crystallizes in the orthorhombic system. The normal form is as massive encrustations, while acicular or fibrous crystals are rarely found. It is colorless to white with tints of yellow, green and pink. It is a soft mineral with variable Mohs hardness around 2.0~2.5, and it has a low specific gravity
It is readily soluble in water, and absorbs water from the air. It converts to hexahydrate with the loss of one water molecule and a switch to monoclinic structure.
The epsomite group includes the solid solution series with morenosite (·) and goslarite (·): these salts have the same structure with the salt-dependent nickel, zinc, or magnesium atoms occupying similar positions. Intermediate salts containing more than one of these elements are also possible.
Etymology
In 1618, a well was dug by a local farmer on Epsom common, half a mile west of Epsom, Surrey, England, after his cattle refused to drink from surface water in the area. The well water became known and used externally as a treatment for skin diseases and other complaints, and both the well and its water were soon named for Epsom. By the mid-17th century Epsom Water had become popular as far away as continental European countries. To maintain supplies of the water, a second well was dug near the centre of the village, which had become widely known as Epsom Spa. In 1695 Dr. Nehemiah Grew published his study on the water's composition, establishing Epsom salts as a medical remedy. It was later systematically described in 1806. It has been also referred to as "cave cotton" when in its fibrous form.
Occurrence
Epsomite forms as encrustations or efflorescences on limestone cavern walls and mine timbers and walls, rarely as volcanic fumarole deposits, and as rare beds in evaporite layers such as those found in certain bodies of salt water. It occurs in association with melanterite, gypsum, halotrichite, pickeringite, alunogen, rozenite, and mirabilite.
Naturally occurring epsomite in caverns can potentially produce particulates of inhalable size, and may contain significant amounts of toxic impurities.
Uses
Epsom salt is commonly sold as the main ingredient in bath salt, with additives such as glycerin (used as a humectant) and fragrances. The purpose of bath salts is mostly to make the bathing experience more enjoyable and serve as a vehicle for cosmetics, though they are said to improve cleaning and aid in exfoliation.
Research on topical magnesium (for example Epsom salt baths) is very limited. The trade organization 'Epsom Salt Council' recommends bathing 2 or 3 times/week, using 500-600 g Epsom salts each time.
Gallery

References
References
- Warr, L.N.. (2021). "IMA–CNMNC approved mineral symbols".
- "Epsomite".
- "Epsomite".
- "Epsomite".
- "Epsomite".
- "The Waters of Epsom Spa".
- (2007). "McGraw-Hill Encyclopedia of Science & Technology". McGraw-Hill.
- (April 2022). "Could soluble minerals be hazardous to human health? Evidence from fibrous epsomite". Environmental Research.
- Browning, Marie. (1999). "Natural Soapmaking". Sterling.
- "Why take an epsom salts bath?".
- (October 2015). "Report on absorption of magnesium sulfate". Epsom Salt Council.
- (August 2023). "Evaluation of Occupational Exposures and Indoor Environmental Quality in an Underground Cavern Workplace".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Epsomite — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report