From Surf Wiki (app.surf) — the open knowledge base
Ductal carcinoma in situ
Pre-cancerous breast lesion
.jpg)
Pre-cancerous breast lesion
| Field | Value |
|---|---|
| name | Breast cancer in situ |
| synonyms | Intraductal carcinoma |
| image | Lobules and ducts of the breast.jpg |
| caption | Ducts of the mammary gland, the location of ductal carcinoma |
| field | Oncology |
.jpg)
**Ductal carcinoma ''in situ''''' (DCIS), also known as **intraductal carcinoma''', is a pre-cancerous or non-invasive cancerous lesion of the breast. DCIS is classified as Stage 0. It rarely produces symptoms or a breast lump that can be felt, typically being detected through screening mammography. It has been diagnosed in a significant percentage of men (see male breast cancer).
In DCIS, abnormal cells are found in the lining of one or more milk ducts in the breast. In situ means "in place" and refers to the fact that the abnormal cells have not moved out of the mammary duct and into any of the surrounding tissues in the breast ("pre-cancerous" indicates that it has not yet become an invasive cancer). In some cases, DCIS may become invasive and spread to other tissues, but there is no way of determining which lesions will remain stable without treatment, and which will go on to become invasive. DCIS encompasses a wide spectrum of diseases ranging from low-grade lesions that are not life-threatening to high-grade (i.e., potentially highly aggressive) lesions.
DCIS has been classified according to the architectural pattern of the cells (solid, cribriform, papillary, and micropapillary), tumor grade (high, intermediate, and low grade), or the presence or absence of comedo histology; or, in the case of the apocrine cell-based in situ carcinoma, apocrine ductal carcinoma in situ, it may be classified according to the cell type forming the lesion. DCIS can be detected on mammograms by examining tiny specks of calcium known as microcalcifications. Since suspicious groups of microcalcifications can appear even in the absence of DCIS, a biopsy may be necessary for diagnosis.
About 20–30% of those who do not receive treatment develop breast cancer. DCIS is the most common type of pre-cancer in women. There is some disagreement on its status as cancer; some bodies include DCIS when calculating breast cancer statistics, while others do not.
Terminology
Ductal carcinoma in situ (DCIS) literally means groups of "cancerous" epithelial cells which remain in their normal location (in situ) within the ducts and lobules of the mammary gland. Clinically, DCIS is considered to be a premalignant (i.e. potentially malignant) condition, because the biologically abnormal cells have not yet crossed the basement membrane to invade the surrounding tissue. When multiple lesions (known as "foci" of DCIS) are present in different quadrants of the breast, this is referred to as "multicentric" disease.
For statistical purposes, DCIS is sometimes counted as a "cancer", but this is not always the case. When classified as a cancer, it is referred to as a "non-invasive" or "pre-invasive" form. It is described by the National Cancer Institute as a "noninvasive condition".
Signs and symptoms
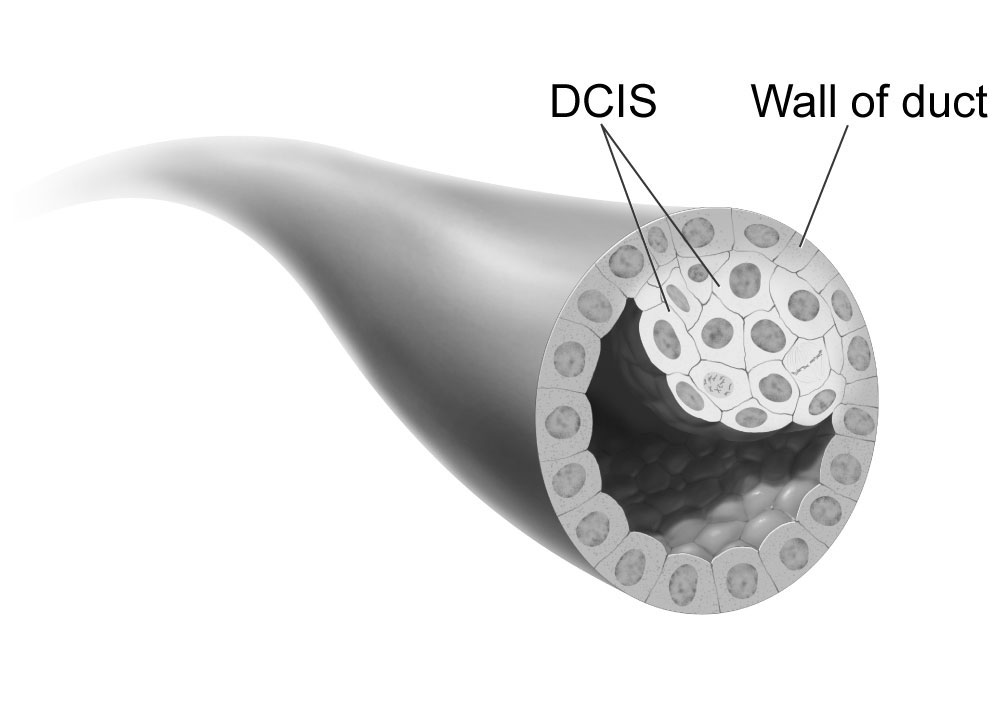
Most of the women who develop DCIS do not experience any symptoms. The majority of cases (80-85%) are detected through screening mammography. The first signs and symptoms may appear if the cancer advances. Because of the lack of early symptoms, DCIS is most often detected at screening mammography.
In a few cases, DCIS may cause:
- A lump or thickening in or near the breast or under the arm
- A change in the size or shape of the breast
- Nipple discharge or nipple tenderness; the nipple may also be inverted, or pulled back into the breast
- Ridges or pitting of the breast; the skin may look like the skin of an orange
- A change in the way the skin of the breast, areola, or nipple looks or feels such as warmth, swelling, redness or scaliness.
Causes
The specific causes of DCIS are still unknown. The risk factors for developing this condition are similar to those for invasive breast cancer.
Some women are however more prone than others to developing DCIS. Women considered at higher risks are those who have a family history of breast cancer, those who have had their periods at an early age or who have had a late menopause. Also, women who have never had children or had them late in life are also more likely to get this condition.
Long-term use of estrogen-progestin hormone replacement therapy (HRT) for more than five years after menopause, genetic mutations (BRCA1 or BRCA2 genes), atypical hyperplasia, as well as radiation exposure or exposure to certain chemicals may also contribute in the development of the condition. Nonetheless, the risk of developing noninvasive cancer increases with age and it is higher in women older than 45 years.
Diagnosis
80% of cases in the United States are detected by mammography screening. More definitive diagnosis is made by breast biopsy for histopathology. File:Mammogram microcalcifications in carcinoma in situ, CC, details.png|Mammogram microcalcifications in ductal carcinoma in situ File:Histopathology of dystrophic microcalcifications in ductal carcinoma in situ.jpg|Histopathology of dystrophic microcalcifications in DCIS, H&E stain. File:Histopathologic architectural patterns of DCIS.png|Histopathologic architectural patterns of DCIS. File:Histopathology of high-grade DCIS.png|Histopathology of high-grade DCIS. H&E stain. RBC = red blood cell. File:Histopathology of microinvasive ductal carcinoma in situ.png|DCIS with microinvasion, defined as focus of invasive cancer measuring up to 1.0 mm in size. File:Immunohistochemistry with calponin in ductal carcinoma in situ.jpg|Immunohistochemistry for calponin in ductal carcinoma in situ, highlighting myoepithelial cells around all tumor cells, thereby ruling out invasive ductal carcinoma. File:Histopathology of ductal carcinoma in situ with comedo necrosis.jpg|Ductal carcinoma in situ with comedo necrosis spanning 30% of its diameter, which is generally regarded as the minimal size to classify it as comedo.
Treatment
There are different opinions on the best treatment of DCIS. Surgical removal, with or without additional radiation therapy or tamoxifen, is the recommended treatment for DCIS by the National Cancer Institute. Surgery may be either a breast-conserving lumpectomy or a mastectomy (complete or partial removal of the affected breast). If a lumpectomy is used it is often combined with radiation therapy. Chemotherapy is not needed for DCIS since the disease is noninvasive.
While surgery reduces the risk of subsequent cancer, many people never develop cancer even without treatment and the associated side effects. There is no evidence comparing surgery with watchful waiting and some feel watchful waiting may be a reasonable option in certain cases.
Radiation therapy
Use of radiation therapy after lumpectomy provides equivalent survival rates to mastectomy, although there is a slightly higher risk of recurrent disease in the same breast in the form of further DCIS or invasive breast cancer. Systematic reviews (including a Cochrane review) indicate that the addition of radiation therapy to lumpectomy reduces recurrence of DCIS or later onset of invasive breast cancer in comparison with breast-conserving surgery alone, without affecting mortality. The Cochrane review did not find any evidence that the radiation therapy had any long-term toxic effects.
Mastectomy
There is no evidence that mastectomy decreases the risk of death over a lumpectomy.
Mastectomies remain a common recommendation in those with persistent microscopic involvement of margins after local excision or with a diagnosis of DCIS and evidence of suspicious, diffuse microcalcifications.
Sentinel node biopsy
Some institutions that have encountered high rates of recurrent invasive cancers after mastectomy for DCIS have endorsed routine sentinel node biopsy (SNB). However, research indicates that sentinel node biopsy has risks that outweigh the benefits for most women with DCIS. SNB should be considered with tissue diagnosis of high-risk DCIS (grade III with palpable mass or larger size on imaging) as well as in people undergoing mastectomy after a core or excisional biopsy diagnosis of DCIS.
Prognosis
With treatment, the prognosis is excellent, with greater than 97% long-term survival. If untreated, DCIS progresses to invasive cancer in roughly one-third of cases, usually in the same breast and quadrant as the earlier DCIS. About 2% of women who are diagnosed with this condition and treated died within 10 years.
Epidemiology
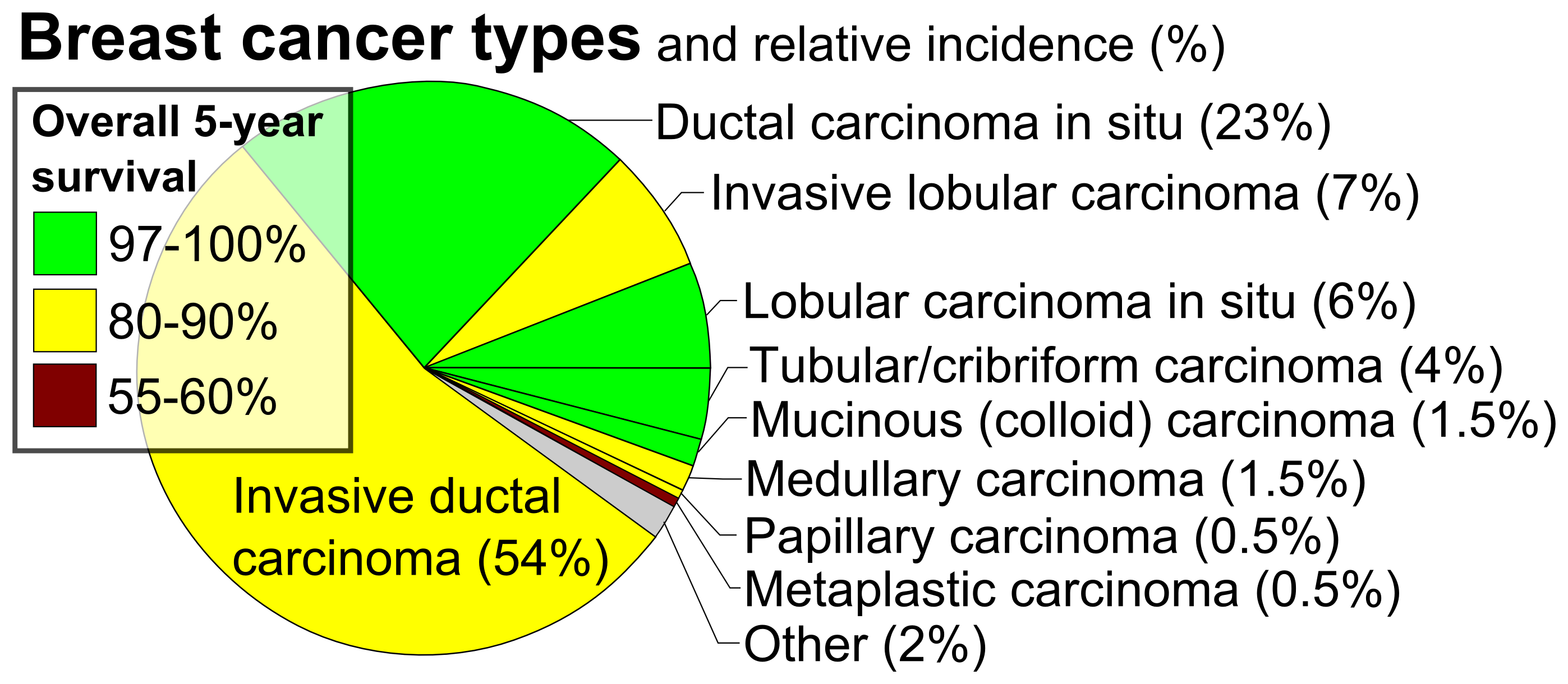
DCIS is often detected with mammographies but can rarely be felt. With the increasing use of screening mammography, noninvasive cancers are more frequently diagnosed and now constitute 15% to 20% of all breast cancers.
Cases of DCIS have increased five-fold between 1983 and 2003 in the United States due to the introduction of screening mammography. In 2009 about 62,000 cases were diagnosed.
References
References
- (May 2013). "A Brief Overview of the WHO Classification of Breast Tumors, 4th Edition, Focusing on Issues and Updates from the 3rd Edition.". Breast Care.
- (1999). "Breast Care: A Clinical Guidebook for Women's Primary Health Care Providers". Springer.
- DePolo, Jamie. (13 October 2023). "Breast Cancer Stages: Stage 0 breast cancer".
- (February 2008). "The sea of uncertainty surrounding ductal carcinoma in situ--the price of screening mammography". J. Natl. Cancer Inst..
- (2005). "Breast MRI: Diagnosis and Intervention". Springer.
- (December 2019). "The diagnosis of male breast cancer". The Netherlands Journal of Medicine.
- (27 May 2020). "Invasive breast cancer and breast cancer mortality after ductal carcinoma in situ in women attending for breast screening in England, 1988-2014: population based observational cohort study". BMJ (Clinical Research Ed.).
- (September 2009). "Diagnosis and management of ductal carcinoma in situ (DCIS)". Evidence Report/Technology Assessment.
- (January 2022). "Apocrine lesions of the breast". Virchows Archiv.
- (2008). "Rubin's Pathology: Clinicopathologic Foundations of Medicine". Lippincott Williams and Wilkins.
- (2010). "Overview of the randomized trials of radiotherapy in ductal carcinoma in situ of the breast". Journal of the National Cancer Institute. Monographs.
- (11 April 2014). "Breast Cancer Treatment (PDQ®)".
- (January 1980). "Breast Cancer Treatment (PDQ®)".
- Allred DC. (2010). "Ductal carcinoma in situ: terminology, classification, and natural history". Journal of the National Cancer Institute. Monographs.
- (2011). "Approach to Internal Medicine: A Resource Book for Clinical Practice". Springer.
- (2006). "Textbook of surgery". Blackwell Pub..
- (2007). "Oncology: An Evidence-Based Approach". Springer.
- "Breast Cancer".
- "Signs and Symptoms".
- "After the mammogram".
- "Intraductal Carcinoma of the Breast".
- (9 January 2015). "Ductal Carcinoma In Situ".
- (2022). "Papillary lesions of the breast.". Virchows Arch.
- (27 August 2020). "Ductal Carcinoma in Situ of the Breast". Comput Struct Biotechnol J.
- Moatasim A, Mamoon N. (2022). "Protocol for the Examination of Resection Specimens from Patients with Ductal Carcinoma In Situ (DCIS) of the Breast, Version: 4.4.0.0. Protocol Posting Date: June 2021". Cureus.
- (2019). "Variability in diagnostic threshold for comedo necrosis among breast pathologists: implications for patient eligibility for active surveillance trials of ductal carcinoma in situ.". Mod Pathol.
- (2015). "A Nationwide Cross-Sectional Survey of UK Breast Surgeons' Views on the Management of Ductal Carcinoma In Situ". International Journal of Breast Cancer.
- (11 July 2014). "Ductal Carcinoma ''In Situ'': Treatment Options for Patients With DCIS". National Institutes of Health.
- (Dec 2013). "Treatment of ductal carcinoma in situ: an uncertain harm-benefit balance.". Prescrire Int.
- [http://www.hopkinsmedicine.org/avon_foundation_breast_center/breast_cancers_other_conditions/ductal_carcinoma_in_situ.html Ductal Carcinoma in Situ (DCIS)] {{Webarchive. link. (24 April 2015 , [[Johns Hopkins Medicine]])
- (2013). "Post-operative radiotherapy for ductal carcinoma in situ of the breast". The Cochrane Database of Systematic Reviews.
- (2010). "Ductal carcinoma in situ of the breast: a systematic review of incidence, treatment, and outcomes". Journal of the National Cancer Institute.
- (2010). "Overview of the randomized trials of radiotherapy in ductal carcinoma in situ of the breast". Journal of the National Cancer Institute. Monographs.
- (September 2009). "NIH DCIS Consensus Conference Statement". National Institutes of Health.
- (September 2009). "Diagnosis and management of ductal carcinoma in situ (DCIS).". Evidence Report/Technology Assessment.
- "Intraductal carcinoma".
- (February 2007). "Role of sentinel lymph node biopsy in ductal carcinoma-in-situ treated by mastectomy". Annals of Surgical Oncology.
- (1 December 2019). "Long-Term Outcomes of Sentinel Lymph Node Biopsy for Ductal Carcinoma in Situ". JNCI Cancer Spectrum.
- (November 2019). "Reliability of preoperative breast biopsies showing ductal carcinoma in situ and implications for non-operative treatment: a cohort study.". Breast Cancer Research and Treatment.
- (April 2007). "Is there an indication for sentinel node biopsy in patients with ductal carcinoma in situ of the breast? A review". European Journal of Cancer.
- Basic Pathology, Robbins. (2018). "Breast". Copyright © 2018 by Elsevier Inc.
- (2010). "Biomarker Expression and Risk of Subsequent Tumors After Initial Ductal Carcinoma in Situ Diagnosis". JNCI Journal of the National Cancer Institute.
- (June 2009). "Stromal caveolin-1 levels predict early DCIS progression to invasive breast cancer". Cancer Biology & Therapy.
- (2010). "Epidemiology of ductal carcinoma in situ.". Journal of the National Cancer Institute. Monographs.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Ductal carcinoma in situ — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report