From Surf Wiki (app.surf) — the open knowledge base
Dopaminergic pathways
Projection neurons in the brain that synthesize and release dopamine
Projection neurons in the brain that synthesize and release dopamine
Dopaminergic pathways (dopamine pathways, dopaminergic projections) in the human brain are involved in both physiological and behavioral processes including movement, cognition, executive functions, reward, motivation, and neuroendocrine control. Each pathway is a set of projection neurons, consisting of individual dopaminergic neurons.
There are more than 10 dopaminergic cell groups and pathways. The four major dopaminergic pathways are the mesolimbic pathway, the mesocortical pathway, the nigrostriatal pathway, and the tuberoinfundibular pathway. The mesolimbic pathway and the mesocortical pathway form the mesocorticolimbic system. Two other dopaminergic pathways to be considered are the hypothalamospinal tract and the incertohypothalamic pathway.
Parkinson's disease, attention deficit hyperactivity disorder (ADHD), substance use disorders (addiction), and restless legs syndrome (RLS) can be attributed to dysfunction in specific dopaminergic pathways.
The dopamine neurons of the dopaminergic pathways synthesize and release the neurotransmitter dopamine. Enzymes tyrosine hydroxylase and dopa decarboxylase are required for dopamine synthesis. These enzymes are both produced in the cell bodies of dopamine neurons. Dopamine is stored in the cytoplasm and vesicles in axon terminals. Dopamine release from vesicles is triggered by action potential propagation-induced membrane depolarization. The axons of dopamine neurons extend the entire length of their designated pathway.
Pathways {{anchor|Mesocorticolimbic projection}}
Major
Six of the dopaminergic pathways are listed below.
| Pathway name | Description | Associated processes | Associated disorders | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mesocorticolimbic | ||||||||||||
| system | vauthors = Ikemoto S | title = Brain reward circuitry beyond the mesolimbic dopamine system: a neurobiological theory | journal = Neuroscience and Biobehavioral Reviews | volume = 35 | issue = 2 | pages = 129–50 | date = November 2010 | pmid = 20149820 | pmc = 2894302 | doi = 10.1016/j.neubiorev.2010.02.001 | quote = Recent studies on intracranial self-administration of neurochemicals (drugs) found that rats learn to self-administer various drugs into the mesolimbic dopamine structures–the posterior ventral tegmental area, medial shell nucleus accumbens and medial olfactory tubercle. ... In the 1970s it was recognized that the olfactory tubercle contains a striatal component, which is filled with GABAergic medium spiny neurons receiving glutamatergic inputs form cortical regions and dopaminergic inputs from the VTA and projecting to the ventral pallidum just like the nucleus accumbens }} | |
| Figure 3: The ventral striatum and self-administration of amphetamine The "meso" prefix in the word "mesolimbic" refers to the midbrain, or "middle brain", since "meso" means "middle" in Greek. | ||||||||||||
| The mesocortical pathway transmits dopamine from the VTA to the prefrontal cortex. The "meso" prefix in "mesocortical" refers to the VTA, which is located in the midbrain, and "cortical" refers to the cortex. | ||||||||||||
| Nigrostriatal pathway | vauthors = Hull EM, Rodríguez-Manzo G | chapter = Male Sexual Behavior | date=2017 | doi = 10.1016/b978-0-12-803592-4.00001-8 | title = Hormones, Brain and Behavior | pages=1–57 | publisher=Elsevier | isbn = 978-0-12-803608-2 }} to the caudate nucleus and putamen. | ||||
| Tuberoinfundibular pathway | The tuberoinfundibular pathway transmits dopamine from the hypothalamus to the pituitary gland. | |||||||||||
| Hypothalamospinal tract | The hypothalamospinal pathway influences locomotor networks in the brainstem and spinal cord. Modulating motor control and coordination, showcasing the interconnected nature of neural circuits in the brain. | |||||||||||
| Incertohypothalamic pathway | This pathway from the zona incerta influences the hypothalamus and locomotor centers in the brainstem. |
Minor
:Hypothalamospinal :* Hypothalamus → Spinal cord :Incertohypothalamic :* Zona incerta → Hypothalamus :* Zona incerta → Brainstem VTA → Amygdala (mesoamygdaloid pathway) :VTA → Hippocampus :VTA → Cingulate cortex :VTA → Olfactory bulb :SNc → Subthalamic nucleus
Function
Mesocorticolimbic system

The mesocorticolimbic system (mesocorticolimbic circuit) refers to both the mesocortical and mesolimbic pathways. Both pathways originate at the ventral tegmental area (VTA) which is located in the midbrain. Through separate connections to the prefrontal cortex (mesocortical) and ventral striatum (mesolimbic), the mesocorticolimbic projection has a significant role in learning, motivation, reward, memory and movement. Dopamine receptor subtypes, D1 and D2 have been shown to have complementary functions in the mesocorticolimbic projection, facilitating learning in response to both positive and negative feedback. Both pathways of the mesocorticolimbic system are associated with ADHD, schizophrenia and addiction.
Mesocortical pathway
The mesocortical pathway projects from the ventral tegmental area to the prefrontal cortex (VTA → Prefrontal cortex). This pathway is involved in cognition and the regulation of executive functions (e.g., attention, working memory, inhibitory control, planning, etc.) This intricate neural circuit serves as a crucial communication route within the brain, facilitating the transmission of dopamine, a neurotransmitter associated with reward, motivation, and cognitive control. The prefrontal cortex, being a central hub for executive functions, relies on the input from the mesocortical pathway to modulate and fine-tune cognitive processes essential for goal-directed behavior and decision-making. Dysregulation of the neurons in this pathway has been connected to ADHD.
Mesolimbic pathway
Referred to as the reward pathway, mesolimbic pathway projects from the ventral tegmental area to the ventral striatum (VTA → Ventral striatum [nucleus accumbens and olfactory tubercle]). When a reward is anticipated, the firing rate of dopamine neurons in the mesolimbic pathway increases. The mesolimbic pathway is involved with incentive salience, motivation, reinforcement learning, fear and other cognitive processes. In animal studies, depletion of dopamine in this pathway, or lesions at its site of origin, decrease the extent to which an animal is willing to go to obtain a reward (e.g., the number of lever presses for nicotine or time searching for food). Research is ongoing to determine the role of the mesolimbic pathway in the perception of pleasure.

Nigrostriatal pathway
The nigrostriatal pathway is involved in behaviors relating to movement and motivation. The transmission of dopaminergic neurons to the dorsal striatum particularly plays a role in reward and motivation while movement is influenced by the transmission of dopaminergic neurons to the substantia nigra. The nigrostriatal pathway is associated with conditions such as Huntington's disease, Parkinson's disease, ADHD, Schizophrenia, and Tourette's Syndrome. Huntington's disease, Parkinson's disease, and Tourette's Syndrome are conditions affected by motor functioning while schizophrenia and ADHD are affected by reward and motivation functioning. This pathway also regulates associated learning such as classical conditioning and operant conditioning.
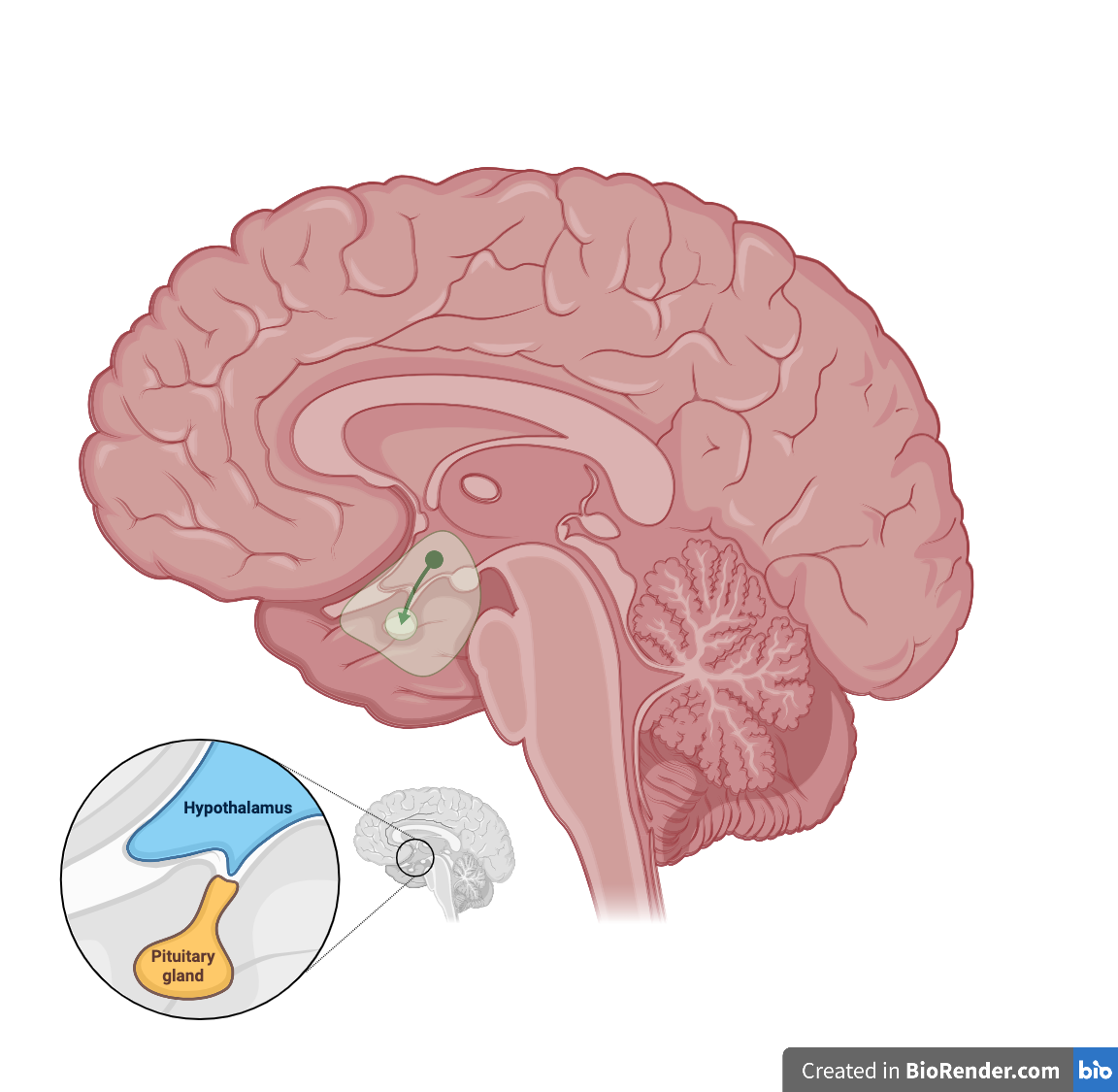
Tuberoinfundibular pathway
The tuberoinfundibular pathway transmits dopamine from the hypothalamus to the pituitary gland. This neural circuit plays a pivotal role in the regulation of hormonal balance and, specifically, in modulating the secretion of prolactin from the pituitary gland, which is responsible for breast milk production in females. Hyperprolactinemia is an associated condition caused by an excessive amount of prolactin production that is common in pregnant women. After childbirth, the tuberoinfundibular pathway resumes its role in regulating prolactin levels. The decline in estrogen levels postpartum contributes to the restoration of dopaminergic inhibition, preventing sustained hyperprolactinemia in non-pregnant and non-nursing individuals.
Cortico-basal ganglia-thalamo-cortical loop
The dopaminergic pathways that project from the substantia nigra pars compacta (SNc) and ventral tegmental area (VTA) into the striatum (i.e., the nigrostriatal and mesolimbic pathways, respectively) form one component of a sequence of pathways known as the cortico-basal ganglia-thalamo-cortical loop. The nigrostriatal component of the loop consists of the SNc, giving rise to both inhibitory and excitatory pathways that run from the striatum into the globus pallidus, before carrying on to the thalamus, or into the subthalamic nucleus before heading into the thalamus. The dopaminergic neurons in this circuit increase the magnitude of phasic firing in response to positive reward error, that is when the reward exceeds the expected reward. These neurons do not decrease phasic firing during a negative reward prediction (less reward than expected), leading to hypothesis that serotonergic, rather than dopaminergic neurons encode reward loss. Dopamine phasic activity also increases during cues that signal negative events, however dopaminergic neuron stimulation still induces place preference, indicating its main role in evaluating a positive stimulus. From these findings, two hypotheses have developed, as to the role of the basal ganglia and nigrostriatal dopamine circuits in action selection. The first model suggests a "critic" which encodes value, and an actor which encodes responses to stimuli based on perceived value. However, the second model proposes that the actions do not originate in the basal ganglia, and instead originate in the cortex and are selected by the basal ganglia. This model proposes that the direct pathway controls appropriate behavior and the indirect suppresses actions not suitable for the situation. This model proposes that tonic dopaminergic firing increases the activity of the direct pathway, causing a bias towards executing actions faster.
These models of the basal ganglia are thought to be relevant to the study of OCD, ADHD, Tourette syndrome, Parkinson's disease, schizophrenia, and addiction. For example, Parkinson's disease is hypothesized to be a result of excessive inhibitory pathway activity, which explains the slow movement and cognitive deficits, while Tourettes is proposed to be a result of excessive excitatory activity resulting in the tics characteristic of Tourettes.
Regulation
The ventral tegmental area and substantia nigra pars compacta receive inputs from other neurotransmitters systems, including glutaminergic inputs, GABAergic inputs, cholinergic inputs, and inputs from other monoaminergic nuclei. The VTA contains 5-HT1A receptors that exert a biphasic effects on firing, with low doses of 5-HT1A receptor agonists eliciting an increase in firing rate, and higher doses suppressing activity. The 5-HT2A receptors expressed on dopaminergic neurons increase activity, while 5-HT2C receptors elicit a decrease in activity. The mesolimbic pathway, which projects from the VTA to the nucleus accumbens, is also regulated by muscarinic acetylcholine receptors. In particular, the activation of muscarinic acetylcholine receptor M2 and muscarinic acetylcholine receptor M4 inhibits dopamine release, while muscarinic acetylcholine receptor M1 activation increases dopamine release. GABAergic inputs from the striatum decrease dopaminergic neuronal activity, and glutaminergic inputs from many cortical and subcortical areas increase the firing rate of dopaminergic neurons. Endocannabinoids also appear to have a modulatory effect on dopamine release from neurons that project out of the VTA and SNc. Noradrenergic inputs deriving from the locus coeruleus have excitatory and inhibitory effects on the dopaminergic neurons that project out of the VTA and SNc. The excitatory orexinergic inputs to the VTA originate in the lateral hypothalamus and may regulate the baseline firing of VTA dopaminergic neurons.
| Neurotransmitter | Origin | Type of Connection | Sources | Glutamate | GABA | Serotonin | Norepinephrine | Endocannabinoids | Acetylcholine | Orexin | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Excitatory projections into the VTA and SNc | |||||||||||||||||||
| Inhibitory projections into the VTA and SNc | |||||||||||||||||||
| Modulatory effect, depending on receptor subtype | |||||||||||||||||||
| Produces a biphasic effect on VTA neurons | |||||||||||||||||||
| Modulatory effect, depending on receptor subtype | |||||||||||||||||||
| The excitatory and inhibitory effects of the LC on the VTA and SNc are time-dependent | vauthors = Chandler DJ, Waterhouse BD, Gao WJ | title = New perspectives on catecholaminergic regulation of executive circuits: evidence for independent modulation of prefrontal functions by midbrain dopaminergic and noradrenergic neurons | journal = Frontiers in Neural Circuits | volume = 8 | page = 53 | year = 2014 | pmid = 24904299 | pmc = 4033238 | doi = 10.3389/fncir.2014.00053 | quote = It has been shown that electrical stimulation of LC results in an excitation followed by a brief inhibition of midbrain dopamine (DA) neurons through an α1 receptor dependent mechanism (Grenhoff et al., 1993). | doi-access = free }} | ||||||||
| Excitatory effect on dopaminergic neurons from inhibiting GABAergic inputs | |||||||||||||||||||
| Inhibitory effect on dopaminergic neurons from inhibiting glutamatergic inputs | |||||||||||||||||||
| May interact with orexins via CB1–OX1 receptor heterodimers to regulate neuronal firing | |||||||||||||||||||
| Modulatory effect, depending on receptor subtype | |||||||||||||||||||
| Excitatory effect on dopaminergic neurons via signaling through orexin receptors (OX1 and OX2) | |||||||||||||||||||
| Increases both tonic and phasic firing of dopaminergic neurons in the VTA | |||||||||||||||||||
| May interact with endocannabinoids via CB1–OX1 receptor heterodimers to regulate neuronal firing | vauthors = Flores A, Maldonado R, Berrendero F | title = Cannabinoid-hypocretin cross-talk in the central nervous system: what we know so far | journal = Frontiers in Neuroscience | volume = 7 | page = 256 | date = December 2013 | pmid = 24391536 | pmc = 3868890 | doi = 10.3389/fnins.2013.00256 | quote = Direct CB1-HcrtR1 interaction was first proposed in 2003 (Hilairet et al., 2003). Indeed, a 100-fold increase in the potency of hypocretin-1 to activate the ERK signaling was observed when CB1 and HcrtR1 were co-expressed ... In this study, a higher potency of hypocretin-1 to regulate CB1-HcrtR1 heteromer compared with the HcrtR1-HcrtR1 homomer was reported (Ward et al., 2011b). These data provide unambiguous identification of CB1-HcrtR1 heteromerization, which has a substantial functional impact. ... The existence of a cross-talk between the hypocretinergic and endocannabinoid systems is strongly supported by their partially overlapping anatomical distribution and common role in several physiological and pathological processes. However, little is known about the mechanisms underlying this interaction. ... Acting as a retrograde messenger, endocannabinoids modulate the glutamatergic excitatory and GABAergic inhibitory synaptic inputs into the dopaminergic neurons of the VTA and the glutamate transmission in the NAc. Thus, the activation of CB1 receptors present on axon terminals of GABAergic neurons in the VTA inhibits GABA transmission, removing this inhibitory input on dopaminergic neurons (Riegel and Lupica, 2004). Glutamate synaptic transmission in the VTA and NAc, mainly from neurons of the PFC, is similarly modulated by the activation of CB1 receptors (Melis et al., 2004). | doi-access = free }} | ||||||||
| Figure 1: Schematic of brain CB1 expression and orexinergic neurons expressing OX1 (HcrtR1) or OX2 (HcrtR2) | |||||||||||||||||||
| Figure 2: Synaptic signaling mechanisms in cannabinoid and orexin systems | |||||||||||||||||||
| Figure 3: Schematic of brain pathways involved in food intake |
Notes
References
References
- (December 2007). "Behavioral functions of the mesolimbic dopaminergic system: an affective neuroethological perspective". Brain Research Reviews.
- "Beyond the Reward Pathway". University of Utah.
- (1995). "Psychopharmacology: the fourth generation of progress". Raven Press.
- (2008). "Handbook of Neurochemistry and Molecular Neurobiology". Springer US.
- (2009). "Molecular Neuropharmacology: A Foundation for Clinical Neuroscience". McGraw-Hill Medical.
- (November 2010). "Brain reward circuitry beyond the mesolimbic dopamine system: a neurobiological theory". Neuroscience and Biobehavioral Reviews.
- (2017). "Hormones, Brain and Behavior". Elsevier.
- (2017). "Reference Module in Neuroscience and Biobehavioral Psychology". Elsevier.
- (2012). "Neurobiology of Psychiatric Disorders". Elsevier.
- (2009). "Molecular Neuropharmacology: A Foundation for Clinical Neuroscience". McGraw-Hill Medical.
- (October 2004). "Synaptic release of dopamine in the subthalamic nucleus". The European Journal of Neuroscience.
- (October 2013). "Potential substrates for nicotine and alcohol interactions: a focus on the mesocorticolimbic dopamine system". Biochemical Pharmacology.
- (June 2011). "Mesocorticolimbic glutamatergic pathway". The Journal of Neuroscience.
- (December 2019). "Differential contributions of striatal dopamine D1 and D2 receptors to component processes of value-based decision making". Neuropsychopharmacology.
- (2009). "Molecular Neuropharmacology: A Foundation for Clinical Neuroscience". McGraw-Hill Medical.
- (December 2008). "Dopaminergic and noradrenergic contributions to functionality in ADHD: the role of methylphenidate". Current Neuropharmacology.
- (December 2010). "New insights into the roles of microRNAs in drug addiction and neuroplasticity". Genome Medicine.
- (October 2011). "Transcriptional and epigenetic mechanisms of addiction". Nature Reviews. Neuroscience.
- Keyser, J. De. (1990). "The mesoneocortical dopamine neuron system". Neurology.
- (2006). "Mesocortical dopamine modulation of executive functions: beyond working memory". Psychopharmacology.
- (November 2012). "The mysterious motivational functions of mesolimbic dopamine". Neuron.
- (December 2004). "Mesolimbic dopaminergic pathways in fear conditioning". Progress in Neurobiology.
- (May 2015). "Pleasure systems in the brain". Neuron.
- (June 2013). "Neuroscience of affect: brain mechanisms of pleasure and displeasure". Current Opinion in Neurobiology.
- (2020). "Molecular neuropharmacology a foundation for clinical neuroscience".
- (May 2015). "Pleasure systems in the brain". Neuron.
- (August 2007). "The role of the dorsal striatum in reward and decision-making". The Journal of Neuroscience.
- (2018). "Physiological and Functional Basis of Dopamine Receptors and Their Role in Neurogenesis: Possible Implication for Parkinson's disease". Journal of Experimental Neuroscience.
- (2016-09-09). "Meta-Analysis of Parkinson's Disease Transcriptome Data Using TRAM Software: Whole Substantia Nigra Tissue and Single Dopamine Neuron Differential Gene Expression". PLOS ONE.
- (2017). "Learning and Memory: A Comprehensive Reference". Elsevier.
- (2021-08-01). "Perioperative pain management and outcomes in patients who -discontinued or continued pre-existing buprenorphine therapy". Journal of Opioid Management.
- (2001). "The Maternal Brain".
- (2013). "The neurocircuitry of illicit psychostimulant addiction: acute and chronic effects in humans". Substance Abuse and Rehabilitation.
- (August 2015). "The ins and outs of the striatum: role in drug addiction". Neuroscience.
- Roberts, Angela C.. (June 2011). "The Importance of Serotonin for Orbitofrontal Function". Biological Psychiatry.
- (February 2011). "From reinforcement learning models to psychiatric and neurological disorders". Nature Neuroscience.
- (June 2013). "Abnormally high degree connectivity of the orbitofrontal cortex in obsessive-compulsive disorder". JAMA Psychiatry.
- (1 January 2008). "The neural bases of obsessive-compulsive disorder in children and adults". Development and Psychopathology.
- (January 2010). "Handbook of Behavioral Neuroscience". Academic.
- (June 2015). "Muscarinic regulation of dopamine and glutamate transmission in the nucleus accumbens". Proceedings of the National Academy of Sciences of the United States of America.
- (December 2007). "Endocannabinoid signaling in midbrain dopamine neurons: more than physiology?". Current Neuropharmacology.
- (December 2011). "Dynamic regulation of midbrain dopamine neuron activity: intrinsic, synaptic, and plasticity mechanisms". Neuroscience.
- (2014). "New perspectives on catecholaminergic regulation of executive circuits: evidence for independent modulation of prefrontal functions by midbrain dopaminergic and noradrenergic neurons". Frontiers in Neural Circuits.
- (December 2013). "Cannabinoid-hypocretin cross-talk in the central nervous system: what we know so far". Frontiers in Neuroscience.
- (February 2010). "Lateral hypothalamic orexin/hypocretin neurons: A role in reward-seeking and addiction". Brain Research.
- (March 2014). "Human orexin/hypocretin receptors form constitutive homo- and heteromeric complexes with each other and with human CB1 cannabinoid receptors". Biochemical and Biophysical Research Communications.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Dopaminergic pathways — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report