From Surf Wiki (app.surf) — the open knowledge base
Dihydroorotate dehydrogenase (quinone)
Class of enzymes
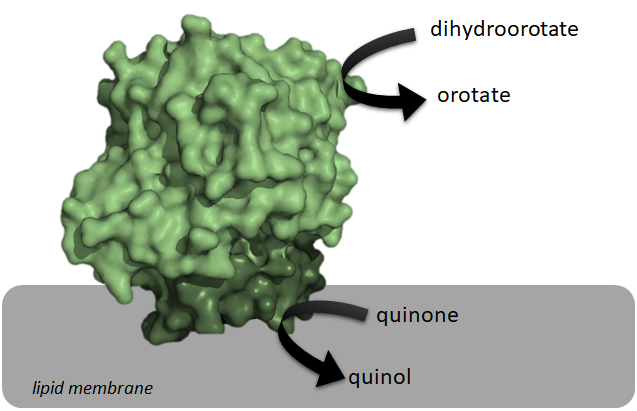
Class of enzymes
| Field | Value |
|---|---|
| Name | Dihydroorotate:quinone oxidoreductase |
| EC_number | 1.3.5.2 |
| CAS_number | 59088-23-2 |
Class 2 dihydroorotate dehydrogenases (DHOQO, ) is an enzyme with systematic name (S)-dihydroorotate:quinone oxidoreductase. This enzyme catalyses the electron transfer from dihydroorotic acid (electron donor) to a quinone (electron acceptor):
These enzymes differ from class 1 dihydroorotate dehydrogenases (DHODH) on the electron acceptor, on their structure, and on their cellular localization. Since the reaction catalyzed by DHOQOs is both part of the electron transport chain and the pyrimidine de novo synthesis, it has been explored as a possible target for cancer treatment, immunological disorders and bacterial/viral infections.

Structure
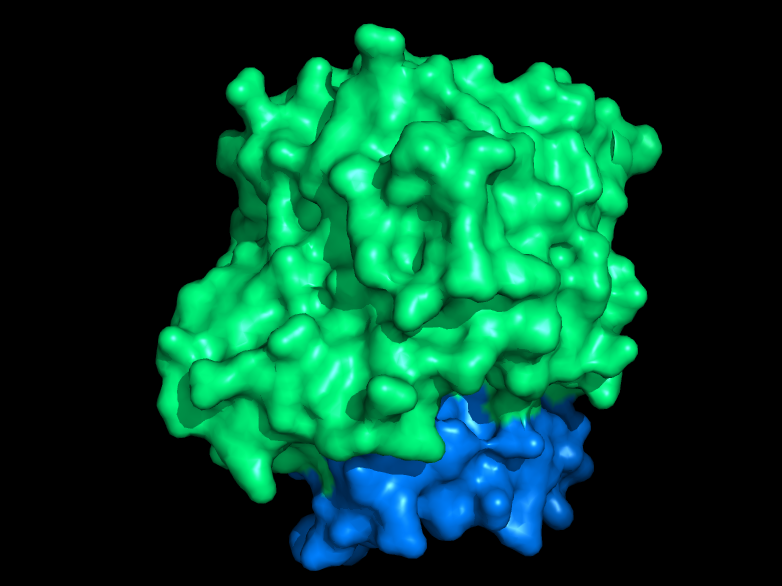
Structurally, DHOQOs are organized in monomers which adopt a (βα)8 (eightfold beta alpha barrel) fold. The enzyme can be separated in its N-terminal domain (blue in the figure) and in its C-terminal domain (green in the figure).
The N-terminal domain is composed of two amphipathic α-helices (αA – αB) which are responsible for the lipid membrane interaction. This region of the protein is also thought to mediate quinone binding.
Regarding the C-terminal domain, much of its structural elements are shared with the soluble counterparts of DHOQOs. This domain is responsible for the binding of the cofactor FMN (making these enzymes part of the Flavoprotein super-family) and the electron donor dihydroorotate, close to the 8 β-strand core.
There are currently crystallographic structures of DHOQOs from 5 different organisms:
- Escherichia coli (example PDB:1F76)
- Mycobacterium tuberculosis (example PDB: 4XQ6)
- Homo sapiens (example PDB:4IGH)
- Rattus norvegicus (example PDB: 4ORI)
- Plasmodium falciparum (example PDB: 1TV5)
References
References
- (November 1978). "Mammalian dihydroorotate dehydrogenase: physical and catalytic properties of the primary enzyme". Archives of Biochemistry and Biophysics.
- (August 1986). "Purification and properties of the bovine liver mitochondrial dihydroorotate dehydrogenase". The Journal of Biological Chemistry.
- (August 1998). "Expression, purification, and characterization of histidine-tagged rat and human flavoenzyme dihydroorotate dehydrogenase". Protein Expression and Purification.
- (December 2006). "Mechanism of flavin reduction in class 2 dihydroorotate dehydrogenases". Biochemistry.
- (March 1999). "The activity of Escherichia coli dihydroorotate dehydrogenase is dependent on a conserved loop identified by sequence homology, mutagenesis, and limited proteolysis". Biochemistry.
- J. Leban, D. Vitt, Human dihydroorotate dehydrogenase inhibitors, a novel approach for the treatment of autoimmune and inflammatory diseases, Arzneimittel-Forschung/Drug Res. (2011). [[doi:10.1055/s-0031-1296169]]
- R.I. Christopherson, S.D. Lyons, P.K. Wilson, Inhibitors of de novo nucleotide biosynthesis as drugs, Acc. Chem. Res. (2002). [[doi:10.1021/ar0000509]]
- M. Löffler, L.D. Fairbanks, E. Zameitat, A.M. Marinaki, H.A. Simmonds, Pyrimidine pathways in health and disease, Trends Mol. Med. (2005). [[doi:10.1016/j.molmed.2005.07.003]]
- D. Lang, R. Thoma, M. Henn-Sax, R. Sterner, M. Wilmanns, Structural evidence for evolution of the β/α barrel scaffold by gene duplication and fusion, Science (80-. ). (2000). [[doi:10.1126/science.289.5484.1546]]
- E. coli Dihydroorotate Dehydrogenase Reveals Structural and Functional Distinction between different classes of dihydroorotate dehydrogenases. [[doi:10.1016/s0969-2126(02)00831-6]]
- Crystal Structure of Dihydroorotate Dehydrogenase from Mycobacterium tuberculosis. [[doi:10.2210/pdb4XQ6/pdb]]
- SAR Based Optimization of a 4-Quinoline Carboxylic Acid Analog with Potent Anti-Viral Activity. [[doi:10.1021/ml300464h]]
- Fluorine Modulates Species Selectivity in the Triazolopyrimidine Class of Plasmodium falciparum Dihydroorotate Dehydrogenase Inhibitors. [[doi:10.1021/jm500481t]]
- Structure of Plasmodium falciparum dihydroorotate dehydrogenase with a bound inhibitor. [[doi:10.1107/S0907444905042642]]
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Dihydroorotate dehydrogenase (quinone) — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report