From Surf Wiki (app.surf) — the open knowledge base
Cyanohydrin
Functional group in organic chemistry
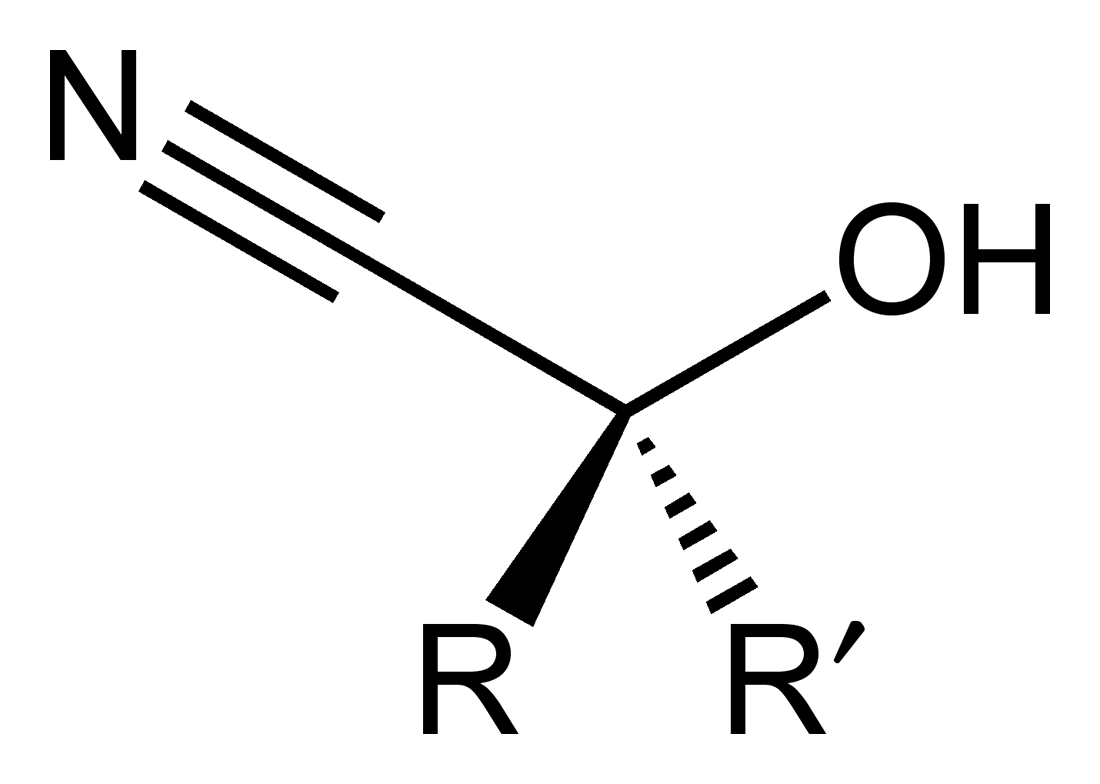
Functional group in organic chemistry

In organic chemistry, a cyanohydrin or hydroxynitrile is a functional group found in organic compounds in which a cyano and a hydroxy group are attached to the same carbon atom. The general formula is , where R is H, alkyl, or aryl. Cyanohydrins are industrially important precursors to carboxylic acids and some amino acids. Cyanohydrins can be formed by the cyanohydrin reaction, which involves treating a ketone or an aldehyde with hydrogen cyanide (HCN) in the presence of excess amounts of sodium cyanide (NaCN) as a catalyst:
:
In this reaction, the nucleophilic ion attacks the electrophilic carbonyl carbon in the ketone, followed by protonation by HCN, thereby regenerating the cyanide anion. Cyanohydrins are also prepared by displacement of sulfite by cyanide salts:
:[[Image:Cyanation of aldehyde with bisulfate.svg|300px]]
Cyanohydrins are intermediates in the Strecker amino acid synthesis. In aqueous acid, they are hydrolyzed to the α-hydroxy acid.
Preparative methods
Cyanohydrins are traditionally prepared by the addition of HCN to the corresponding carbonyl. The reaction is typically catalyzed by base or an enzyme. Because of the hazards with HCN, other less dangerous cyanation reagents are often used.
- Trimethylsilyl cyanide, affording the silyl ether derivative of the cyanohydrin
- Diethylaluminium cyanide, especially for less reactive carbonyls
- Diethyl phosphorocyanidate (DEPC) and lithium cyanide
- Acyl cyanides (RC(O)CN)

Transhydrocyanation
Acetone cyanohydrin, (CH3)2C(OH)CN is the cyanohydrin of acetone. It is generated as an intermediate in the industrial production of methyl methacrylate. In the laboratory, this liquid serves as a source of HCN. The process is called transhydrocyanation, where acetone cyanohydrin, is used as a source of HCN. Thus, acetone cyanohydrin can be used for the preparation of other cyanohydrins, for the transformation of HCN to Michael acceptors, and for the formylation of arenes. Treatment of this cyanohydrin with lithium hydride affords anhydrous lithium cyanide: :[[Image:LiCN-from-acetone-cyanohydrin-2D-skeletal.png|400px]]
Asymmetric cyanohydrin formation
Formation of cyanohydrins introduces a chiral center for aldehydes and for unsymmetrical ketones. The enantioselective hydrocyanation has attracted some attention for the preparation of 2-chloromandelic acid, a drug precursor.
File:Glyconitrile Structural FormulaV1.svg|Glycolonitrile, also called hydroxyacetonitrile or formaldehyde cyanohydrin, is the simplest cyanohydrin. File:Acetone cyanohydrin.svg|Acetone cyanohydrin File:Mandelonitrile-2D-skeletal.svg|Mandelonitrile, occurs in small amounts in the pits of some fruits. File:Amygdalin structure.svg|Amygdalin, a naturally occurring cyanogenic glycoside
References
References
- David T. Mowry. (1948). "The Preparation of Nitriles". [[Chem. Rev.]].
- Corson, B. B.. (1941). "Mandelic Acid".
- (2008). "Lewis Acid Catalyzed Asymmetric Cyanohydrin Synthesis". Chemical Reviews.
- (1999). "Cyanohydrins in Nature and the Laboratory: Biology, Preparations, and Synthetic Applications". Chemical Reviews.
- (2021). "CO2-Enabled Cyanohydrin Synthesis and Facile Iterative Homologation Reactions*". Chemistry – A European Journal.
- William Bauer, Jr. "Methacrylic Acid and Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. {{doi. 10.1002/14356007.a16_441. Article Online Posting Date: June 15, 2000
- 10.1002/047084289X.ra014
- Gaudry, R.. (1955). "Glycolonitrile".
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Cyanohydrin — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report