From Surf Wiki (app.surf) — the open knowledge base
Cope rearrangement
Chemical reaction featuring a sigmatropic rearrangement
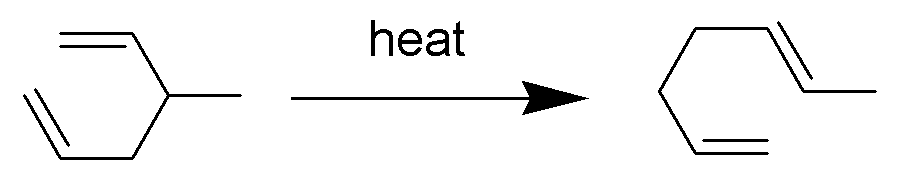
Chemical reaction featuring a sigmatropic rearrangement
The Cope rearrangement (sometimes referred to as the Hardy–Cope rearrangement) is an extensively studied organic reaction involving the [3,3]-sigmatropic rearrangement of 1,5-dienes. It was developed by Arthur C. Cope and Elizabeth Hardy. For example, 3-methyl-hexa-1,5-diene heated to 300 °C yields hepta-1,5-diene.

The Cope rearrangement causes the fluxional states of the molecules in the bullvalene family.
Mechanism
The Cope rearrangement is the prototypical example of a concerted sigmatropic rearrangement. It is classified as a [3,3]-sigmatropic rearrangement with the Woodward–Hoffmann symbol [π2s+σ2s+π2s] and is therefore thermally allowed. It is sometimes useful to think of it as going through a transition state energetically and structurally equivalent to a diradical, although the diradical is not usually a true intermediate (potential energy minimum). The chair transition state illustrated here is preferred in open-chain systems (as shown by the Doering-Roth experiments). However, conformationally constrained systems like cis-1,2-divinyl cyclopropanes can undergo the rearrangement in the boat conformation.

It is currently generally accepted that most Cope rearrangements follow an allowed concerted route through a Hückel aromatic transition state and that a diradical intermediate is not formed. However, the concerted reaction can often be asynchronous and electronically perturbed systems may have considerable diradical character at the transition state. A representative illustration of the transition state of the Cope rearrangement of the electronically neutral hexa-1,5-diene is presented below. Here one can see that the two π-bonds are breaking while two new π-bonds are forming, and simultaneously the σ-bond is breaking while a new σ-bond is forming. In contrast to the Claisen rearrangement, Cope rearrangements without strain release or electronic perturbation are often close to thermally neutral, and may therefore reach only partial conversion due to an insufficiently favorable equilibrium constant. In the case of hexa-1,5-diene, the rearrangement is degenerate (the product is identical to the starting material), so K = 1 by necessity.
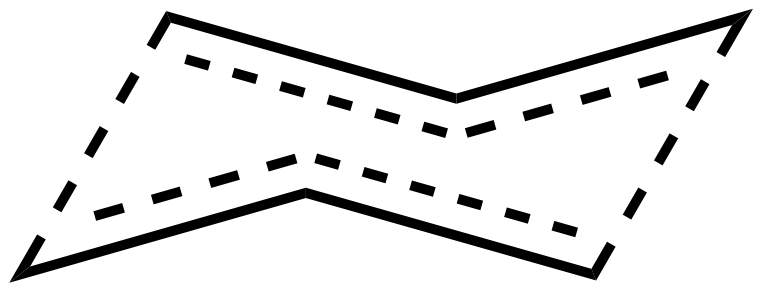
In asymmetric dienes one often needs to consider the stereochemistry, which in the case of pericyclic reactions, such as the Cope rearrangement, can be predicted with the Woodward–Hoffmann rules and consideration of the preference for the chair transition state geometry.
Examples
The rearrangement is widely used in organic synthesis. It is symmetry-allowed when it is suprafacial on all components. The transition state of the molecule passes through a boat or chair like transition state. An example of the Cope rearrangement is the expansion of a cyclobutane ring to a cycloocta-1,5-diene ring:
In this case, the reaction must pass through the boat transition state to produce the two cis double bonds. A trans double bond in the ring would be too strained. The reaction occurs under thermal conditions. The driving force of the reaction is the loss of strain from the cyclobutane ring.
An organocatalytic Cope rearrangement was first reported in 2016. In this process, an aldehyde-substituted 1,5-diene is used, allowing "iminium catalysis" to be achieved using a hydrazide catalyst and moderate levels of enantioselectivity (up to 47% ee) to be achieved.
A number of enzymes catalyze the Cope rearrangement, although its occurrence is rare in nature.
References
References
- [[Arthur C. Cope]]; [[Elizabeth Hardy (chemist). Elizabeth M. Hardy]]; ''[[J. Am. Chem. Soc.]]'' '''1940''', ''62'', 441.
- Rhoads, S. J.; Raulins, N. R.; ''Org. React.'' '''1975''', ''22'', 1–252. (Review)
- Hill, R. K.; ''Compr. Org. Synth.'' '''1991''', ''5'', 785–826.
- Wilson, S. R.; ''Org. React.'' '''1993''', ''43'', 93–250. (Review)
- Michael B. Smith & Jerry March: March's Advanced Organic Chemistry, pp. 1659-1673. John Wiley & Sons, 2007 {{ISBN. 978-0-471-72091-1
- Williams, R. V., Chem. Rev. 2001, 101 (5), 1185–1204.
- (2016). "An Organocatalytic Cope Rearrangement". Angewandte Chemie International Edition.
- (2019). "Structural insights into the calcium dependence of Stig cyclases". RSC Advances.
- (2017-04-26). "Oxidative Cyclization in Natural Product Biosynthesis". Chemical Reviews.
- ''Stepwise Mechanisms in the Oxy-Cope Rearrangement'' Jerome A. Berson and Maitland Jones pp 5017 – 5018; J. Am. Chem. Soc. '''1964'''; {{doi. 10.1021/ja01076a066
- (2007). "Advanced Organic Chemistry: Part B: Reactions and Synthesis". Springer.
- (2007). "Advanced Organic Chemistry: Part B: Reactions and Synthesis". Springer.
- (1975). "[3,3]Sigmatropic rearrangements of 1,5-diene alkoxides. Powerful accelerating effects of the alkoxide substituent". Journal of the American Chemical Society.
- (1984). "Cyclobutene bridgehead olefin route to the American cockroach sex pheromone, periplanone-B". Journal of the American Chemical Society.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Cope rearrangement — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report