From Surf Wiki (app.surf) — the open knowledge base
Clobazam
Benzodiazepine sedative and anticonvulsant
Benzodiazepine sedative and anticonvulsant
| Field | Value | ||
|---|---|---|---|
| image | Clobazam-2D-skeletal.png | ||
| image_class | skin-invert-image | ||
| width | 200 | ||
| image2 | Clobazam ball-and-stick model.png | ||
| image_class2 | bg-transparent | ||
| alt2 | |||
| tradename | Frisium, Onfi, Sympazan, Urbanyl, among others | ||
| Drugs.com | |||
| MedlinePlus | a612008 | ||
| DailyMedID | Clobazam | ||
| pregnancy_AU | C | ||
| dependency_liability | Physical: Very high | ||
| addiction_liability | High | ||
| routes_of_administration | By mouth | ||
| class | Benzodiazepine | ||
| ATC_prefix | N05 | ||
| ATC_suffix | BA09 | ||
| legal_AU | S4 | ||
| legal_BR | B1 | ||
| legal_BR_comment | |||
| legal_CA | Schedule IV | ||
| legal_DE | Rx-only/Anlage III | ||
| legal_NZ | Class C | ||
| legal_UK | Class C | ||
| legal_UK_comment | |||
| legal_US | Schedule IV | ||
| legal_US_comment | |||
| legal_UN | Psychotropic Schedule IV | ||
| legal_status | Rx-only | ||
| <!-- Pharmacokinetic data --> | bioavailability | 87% (oral) | |
| protein_bound | 80–90% | ||
| metabolism | Liver | ||
| metabolites | {{ubl | ||
| onset | 0.5–4 hours | ||
| elimination_half-life | {{ubl | ||
| *N-desmethylclobazam (norclobazam): 71–82 hours}}<ref>{{cite web | title | Clobazam Monograph for Professionals | url=https://www.drugs.com/monograph/clobazam.html }} |
| excretion | {{ubl | ||
| <!-- Identifiers --> | CAS_number | 22316-47-8 | |
| PubChem | 2789 | ||
| IUPHAR_ligand | 7149 | ||
| DrugBank | DB00349 | ||
| ChemSpiderID | 2687 | ||
| UNII | 2MRO291B4U | ||
| KEGG | D01253 | ||
| ChEBI | 31413 | ||
| ChEMBL | 70418 | ||
| synonyms | |||
| IUPAC_name | 7-chloro-1-methyl-5-phenyl-1,5-benzodiazepine-2,4-dione | ||
| C | 16 | ||
| H | 13 | ||
| Cl | 1 | ||
| N | 2 | ||
| O | 2 | ||
| SMILES | ClC1=CC(N(C2=CC=CC=C2)C(CC(N3C)=O)=O)=C3C=C1 | ||
| StdInChI | 1S/C16H13ClN2O2/c1-18-13-8-7-11(17)9-14(13)19(16(21)10-15(18)20)12-5-3-2-4-6-12/h2-9H,10H2,1H3 | ||
| StdInChIKey | CXOXHMZGEKVPMT-UHFFFAOYSA-N |
| Drugs.com = Psychological: High
- N-desmethylclobazam
- 4-hydroxyclobazam
- norclobazam}} | elimination_half-life = {{ubl|
- clobazam: 36–42 hours
- N-desmethylclobazam (norclobazam): 71–82 hours}}
- Kidney (82%)
- Feces (11%)}}
Clobazam, sold under the brand name Frisium, Onfi, Sympazan, Urbanyl, among others, is a long-acting benzodiazepine derivative used as a sedative–hypnotic, anxiolytic, and anticonvulsant that was patented in 1968. Clobazam was first synthesized in 1966 and first published in 1969. It was approved in Australia in 1970 and France in 1974 for short-term anxiety management. Marketing for clobazam in the treatment of epilepsy began in 1984. Clobazam is a unique 1,5 benzodiazepine which is used in the United States only as an anticonvulsant. It is available in other countries for the therapy of severe and disabling anxiety in addition to epilepsy. Clobazam has shown a distinct profile and addictive potential compared to the more common benzodiazepines.
In October 2011, the US Food and Drug Administration approved clobazam as an adjunctive treatment for seizures associated with Lennox–Gastaut syndrome in adults and children aged two years of age and older. In 2005, clobazam also received approval from Health Canada as an add-on therapy for generalized tonic–clonic, myoclonic, and focal impaired awareness seizures.
Medical uses
Clobazam belongs to the benzodiazepine class of drugs. As a long-acting benzodiazepine derivative it is primarily used as a sedative-hypnotic, anxiolytic, and as an adjunctive therapy in epilepsy.
As an adjunctive therapy in epilepsy, it is used in patients who have not responded to first-line drugs and in children who are refractory to first-line drugs. It is unclear if there are any benefits to clobazam over other seizure medications for children with Rolandic epilepsy or other epileptic syndromes. It is not recommended for use in children between the ages of six months and three years, unless there is a compelling need.
Clobazam is sometimes used for refractory epilepsies. However, long-term prophylactic treatment of epilepsy may have considerable drawbacks, most importantly decreased antiepileptic effects due to drug tolerance which may render long-term therapy less effective. Other antiepileptic drugs may therefore be preferred for the long-term management of epilepsy. Furthermore, benzodiazepines may have the drawback, particularly after long-term use, of causing rebound seizures upon abrupt or over-rapid discontinuation of therapy forming part of the benzodiazepine withdrawal syndrome.
Clobazam is approved in Canada for add-on use in tonic–clonic, complex partial, and myoclonic seizures. Clobazam is approved for adjunctive therapy in complex partial seizures, certain types of status epilepticus, specifically the myoclonic, myoclonic-absent, simple partial, complex partial, and tonic varieties, and non-status absence seizures. It is also approved for the treatment of anxiety.
In India, clobazam is approved for use as an adjunctive therapy in epilepsy, and in acute and chronic anxiety. In Japan, clobazam is approved for adjunctive therapy in treatment-resistant epilepsy featuring complex partial seizures. In New Zealand, clobazam is marketed as Frisium. In the United Kingdom clobazam (Frisium) is approved for short-term (2–4 weeks) relief of acute anxiety in patients who have not responded to other drugs, with or without insomnia and without uncontrolled clinical depression. It was not approved in the United States until October 2011, when it was approved for the adjunctive treatment of seizures associated with Lennox–Gastaut syndrome in people aged two years of age or older.
In the United States, clobazam is indicated for use in combination with other medicines to control seizures in people aged two years of age and older who have a specific severe form of epilepsy called Lennox–Gastaut syndrome.
Clinical efficacy and tolerability for management of chronic epilepsy and anxiety disorders has been established in multiple studies.
Side effects
In September 2020, the US Food and Drug Administration required the boxed warning be updated for all benzodiazepine medicines to describe the risks of abuse, misuse, addiction, physical dependence, and withdrawal reactions consistently across all the medicines in the class.
Common
Common side effects include fever, drooling, and constipation.
Warnings and precautions
In December 2013, the US Food and Drug Administration added warnings to the label for clobazam, that it can cause serious skin reactions, Stevens–Johnson syndrome, and toxic epidermal necrolysis, especially in the first eight weeks of treatment.
Overdose
Overdose and intoxication with benzodiazepines, including clobazam, may lead to CNS depression, associated with drowsiness, confusion, and lethargy, possibly progressing to ataxia, respiratory depression, hypotension, and coma or death. The risk of a fatal outcome is increased in cases of combined poisoning with other CNS depressants, including alcohol.
Abuse potential and addiction
Classic (non-anxioselective) benzodiazepines in animal studies have been shown to increase reward-seeking behaviours which may suggest an increased risk of addictive behavioural patterns. Clobazam abuse has been reported in some countries, according to a 1983 World Health Organization report.
Dependence and withdrawal
In humans, tolerance to the anticonvulsant effects of clobazam may occur and withdrawal seizures may occur during abrupt or over-rapid withdrawal.
Clobazam as with other benzodiazepine drugs can lead to physical dependence, addiction, and what is known as the benzodiazepine withdrawal syndrome. Withdrawal from clobazam or other benzodiazepines after regular use often leads to withdrawal symptoms which are similar to those seen during alcohol and barbiturate withdrawal. The higher the dosage and the longer the drug is taken, the greater the risk of experiencing unpleasant withdrawal symptoms. Benzodiazepine treatment should only be discontinued via a slow and gradual dose reduction regimen.
Pharmacology
Clobazam is predominantly a positive allosteric modulator at the GABAA receptor to increase GABAergic transmission, particularly chloride conductance in neurons and with some speculated additional activity at sodium channels and voltage-sensitive calcium channels.
The exact mechanism of action for clobazam, a 1,5-benzodiazepine, which has sedative-hypnotic, anxiolytic, and anticonvulsant effects similar to those produced by other benzodiazepine derivatives, but is a partial agonist at the GABAA receptors. Clobazam is thought to involve the potentiation of GABAergic neurotransmission resulting from binding at the benzodiazepine site of the GABAA receptor. Like other 1,5-benzodiazepines (for example, arfendazam, lofendazam, triflubazam, and CP-1414S), the active metabolite N-desmethylclobazam has less affinity for the α1 subunit of the GABAA receptor compared to the 1,4-benzodiazepines. It has higher affinity for the α2 subunit of the GABAA receptor, which mediates anxiolytic effects, than the α1 subunit. Clobazam also has positive modulatory activity.
Clobazam and its active metabolite, N-desmethylclobazam (norclobazam), work by enhancing GABA-activated chloride influx at GABAA receptors, creating a hyperpolarizing, inhibitory postsynaptic potential. It is the primary active metabolite of clobazam, crucial for its prolonged anti-seizure and anti-anxiety effects, acting similarly on GABAA receptors but with potentially greater importance in long-term therapy, especially for epilepsy, and is largely responsible for the drug's overall clinical action. It was also reported that these effects were inhibited by the GABA antagonist flumazenil, and that clobazam acts more efficiently in GABA-deficient brain tissue.
Metabolism
Clobazam has two major metabolites: N-desmethylclobazam and 4-hydroxyclobazam, the former of which is active. The demethylation is facilitated by CYP2C19, CYP3A4, and CYP2B6 and the 4-hydroxyclobazam by CYP2C18 and CYP2C19.
In children, clobazam half-life values is average 16 hours, while in the elderly, clobazam half-life values are 30 to 48 hours.
Chemistry
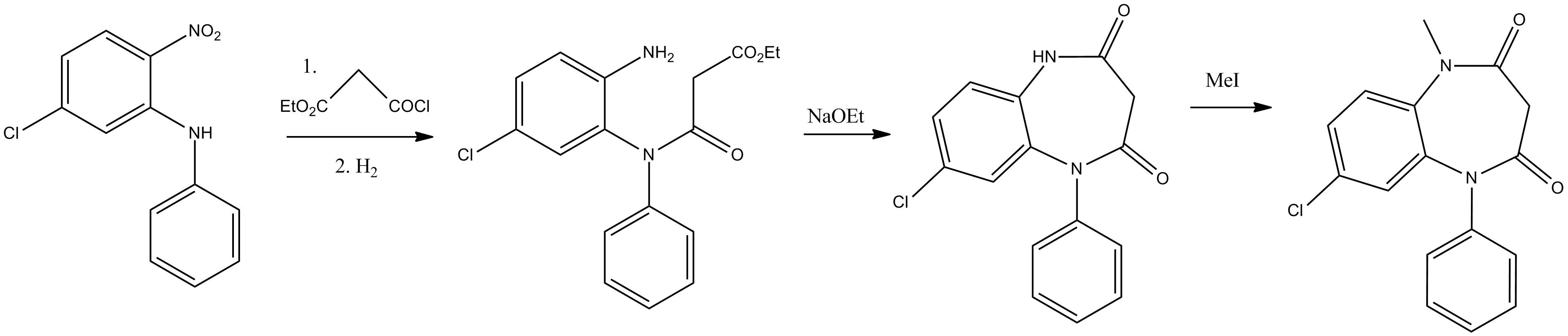
Clobazam is a 1,5-benzodiazepine, meaning that its diazepine ring has nitrogen atoms at the 1 and 5 positions (instead of the usual 1 and 4).
It is not soluble in water and is available in oral form only.
History
Clobazam was discovered at the Maestretti Research Laboratories in Milan and was first published in 1969; Maestretti was acquired by Roussel Uclaf which became part of Sanofi.
References
References
- "Clobazam".
- Anvisa. (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial". [[Diário Oficial da União]].
- (10 July 2025). "Frisium 10 mg Tablet Summary of Product Characteristics (SmPC)".
- (12 March 2024). "Onfi- clobazam tablet; Onfi- clobazam suspension".
- "Clobazam Monograph for Professionals".
- "Urbanyl | Drug Information, Uses, Side Effects, Chemistry".
- "Urbanyl | Drug Information, Uses, Side Effects, Chemistry".
- "Clobazam (T3D4564)". Toxin and Toxin Target Database (T3DB.
- (2020). "StatPearls". StatPearls Publishing.
- (April 1975). "[Study of an anxiolytic, clobazam, in otorhinolaryngology in psychosomatic pharyngeal manifestations]". Semaine des Hopitaux. Therapeutique.
- (1991). "Clobazam in treatment of refractory epilepsy: the Canadian experience. A retrospective study. Canadian Clobazam Cooperative Group". Epilepsia.
- (2015). "Functional Characterization of the 1,5-Benzodiazepine Clobazam and Its Major Active Metabolite N-Desmethylclobazam at Human GABAA Receptors Expressed in Xenopus laevis Oocytes". PLOS ONE.
- "Clobazam".
- (July 2018). "Clobazam monotherapy for focal or generalized seizures". The Cochrane Database of Systematic Reviews.
- "Clobazam 10 mg Tablets - Summary of Product Characteristics (SmPC) - (emc)".
- (December 1998). "Benzodiazepines in the treatment of epilepsy in people with intellectual disability". Journal of Intellectual Disability Research.
- (2020). "Clobazam". Epilepsy Ontario.
- (December 1986). "[Epilepsy with continuous discharges during slow-wave sleep. Treatment with clobazam]". Revue D'electroencephalographie et de Neurophysiologie Clinique.
- (December 1984). "[Treatment of certain forms of status epilepticus by means of a single oral dose of clobazam]". Revue D'electroencephalographie et de Neurophysiologie Clinique.
- "Frisium Press Kit". Aventis Pharma India.
- (July 2003). "Use of clobazam for the treatment of refractory complex partial seizures". Seizure.
- Epilepsy New Zealand. (2000). "Antiepileptic Medication".
- sanofi-aventis. (2002). "Frisium Tablets 10 mg, Summary of Product Characteristics from eMC". Medicines.org.uk.
- "FDA Approves Onfi (clobazam) for the Adjunctive Treatment of Seizures Associated with Lennox-Gastaut Syndrome in Patients Two Years and Older". Lundbeck.
- (2018). "Reference Module in Biomedical Sciences".
- (23 September 2020). "FDA expands Boxed Warning to improve safe use of benzodiazepine drug".
- (3 December 2013). "FDA warns of serious skin reactions with the anti-seizure drug Onfi (clobazam) and has approved label changes". U.S. Food and Drug Administration.
- (July 1997). "Clobazam". International Programme on Chemical Safety (IPCS), World Health Organization.
- (1985). "Benzodiazepines reduce the tolerance to reward delay in rats". Psychopharmacology.
- WHO Review Group. (1983). "Use and abuse of benzodiazepines". Bulletin of the World Health Organization.
- (1983). "[Benzodiazepines in the treatment of epilepsy]". L'Encéphale.
- (1986). "Current status of the 1,4- and 1,5-benzodiazepines in the treatment of epilepsy: the place of clobazam". Epilepsia.
- (1982). "Benzodiazepine withdrawal syndrome: a literature review and evaluation". The American Journal of Drug and Alcohol Abuse.
- {{EMedicine. article. 1187334. Antiepileptic Drugs
- (October 2016). "The clobazam metabolite N-desmethyl clobazam is an α2 preferring benzodiazepine with an improved therapeutic window for antihyperalgesia". Neuropharmacology.
- (August 2001). "[A pharmacological profile of clobazam (Mystan), a new antiepileptic drug]". Nihon Yakurigaku Zasshi. Folia Pharmacologica Japonica.
- (January 2019). "A Comprehensive Overview of the Clinical Pharmacokinetics of Clobazam". Journal of Clinical Pharmacology.
- (August 1996). "Effects of clobazam and its active metabolite on GABA-activated currents in rat cerebral neurons in culture". Epilepsia.
- (December 2002). "Evidence of polymorphic CYP2C19 involvement in the human metabolism of N-desmethylclobazam". Therapeutic Drug Monitoring.
- (November 2004). "In vitro characterization of clobazam metabolism by recombinant cytochrome P450 enzymes: importance of CYP2C19". Drug Metabolism and Disposition.
- (2022). "Antiseizure Medication Interactions: A Clinical Guide". Springer International Publishing.
- (January 2007). "Clobazam". Neurotherapeutics.
- (March 2009). "Drug treatment of epilepsy in the century of the ILAE: the second 50 years, 1959-2009". Epilepsia.
- (1979). "Clobazam: pharmacological and therapeutic profile". British Journal of Clinical Pharmacology.
- (2014). "L'industria delle Medicine.". EDRA LSWR.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Clobazam — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report