From Surf Wiki (app.surf) — the open knowledge base
Carbon tetrafluoride

Carbon tetrafluoride 0.0038%V at 25 °C | NFPA-H = 1 | NFPA-F = 0 | NFPA-R = 0 | NFPA-S = SA Difluoromethane Fluoroform Tetrabromomethane Tetraiodomethane Germanium tetrafluoride Tin tetrafluoride Lead tetrafluoride
Tetrafluoromethane, also known as carbon tetrafluoride or R-14, is the simplest perfluorocarbon (CF4). As its IUPAC name indicates, tetrafluoromethane is the perfluorinated counterpart to the hydrocarbon methane. It can also be classified as a haloalkane or halomethane. Tetrafluoromethane is a useful refrigerant but also a potent greenhouse gas. Although its atmospheric concentration is small, it persists in the atmosphere for 50,000 years, giving it a very high global warming potential. It has a very high bond strength due to the nature of the carbon–fluorine bond.
Bonding
Because of the multiple carbon–fluorine bonds, and the high electronegativity of fluorine, the carbon in tetrafluoromethane has a significant positive partial charge which strengthens and shortens the four carbon–fluorine bonds by providing additional ionic character. Carbon–fluorine bonds are the strongest single bonds in organic chemistry. Additionally, they strengthen as more carbon–fluorine bonds are added to the same carbon atom. In the one-carbon organofluorine compounds represented by molecules of fluoromethane, difluoromethane, trifluoromethane, and tetrafluoromethane, the carbon–fluorine bonds are strongest in tetrafluoromethane.{{Cite journal
Preparation
Tetrafluoromethane is the product when any carbon compound, including carbon itself, is burned in an atmosphere of fluorine. With hydrocarbons, hydrogen fluoride is a coproduct. It was first reported in 1926. It can also be prepared by the fluorination of carbon dioxide, carbon monoxide or phosgene with sulfur tetrafluoride. Commercially it is manufactured by the reaction of hydrogen fluoride with dichlorodifluoromethane or chlorotrifluoromethane; it is also produced during the electrolysis of metal fluorides MF, MF2 using a carbon electrode.
Although it can be made from a myriad of precursors and fluorine, elemental fluorine is expensive and difficult to handle. Consequently, is prepared on an industrial scale using hydrogen fluoride: :CCl2F2 + 2 HF → CF4 + 2 HCl
Laboratory synthesis
Tetrafluoromethane and silicon tetrafluoride can be prepared in the laboratory by the reaction of silicon carbide with fluorine. : SiC + 4 F2 → CF4 + SiF4
Reactions
Tetrafluoromethane, like other fluorocarbons, is very stable due to the strength of its carbon–fluorine bonds. The bonds in tetrafluoromethane have a bonding energy of 515 kJ⋅mol−1. As a result, it is inert to acids and hydroxides. However, it reacts explosively with alkali metals. Thermal decomposition or combustion of CF4 produces toxic gases (carbonyl fluoride and carbon monoxide) and in the presence of water will also yield hydrogen fluoride.
It is very slightly soluble in water (about 20 mg⋅L−1), but highly soluble in organic solvents. When liquified, it is completely miscible in organic solvents.
Uses
Tetrafluoromethane is sometimes used as a low temperature refrigerant (R-14). It is used in electronics microfabrication alone or in combination with oxygen as a plasma etchant for silicon, silicon dioxide, and silicon nitride. It also has uses in neutron detectors.
Environmental effects

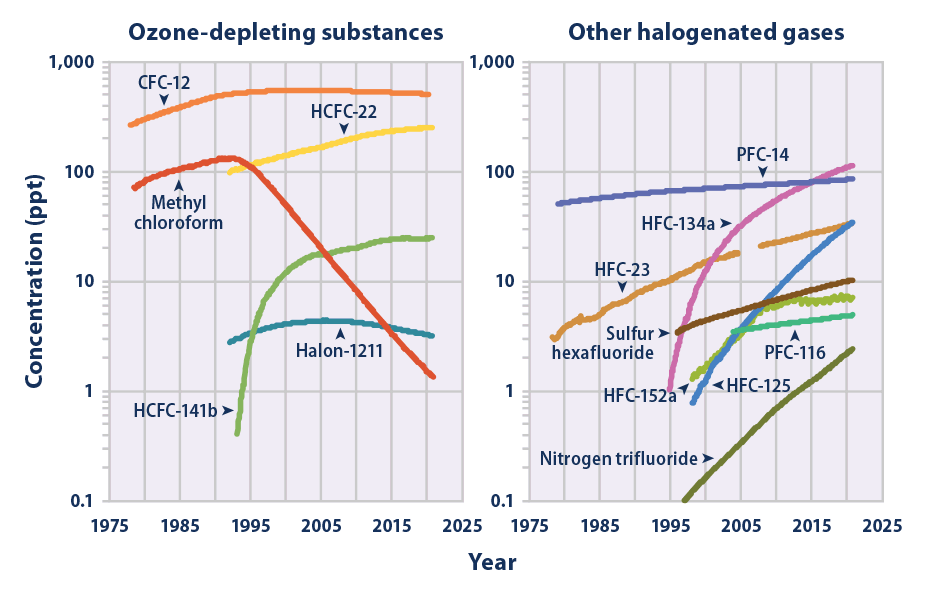
Tetrafluoromethane is a potent greenhouse gas that contributes to the greenhouse effect. It is very stable, has an atmospheric lifetime of 50,000 years, and a high greenhouse warming potential 6,500 times that of CO2.
Tetrafluoromethane is the most abundant perfluorocarbon in the atmosphere, where it is designated as PFC-14. Its atmospheric concentration is growing: its preindustrial concentration produced via radioactive oxidation of fluorite and reaction with organic molecules was less than 40 parts per trillion. As of 2019, the man-made gases CFC-11 and CFC-12 continue to contribute a stronger radiative forcing than PFC-14.
Although structurally similar to chlorofluorocarbons (CFCs), tetrafluoromethane does not deplete the ozone layer because the carbon–fluorine bond is much stronger than that between carbon and chlorine.
Main industrial emissions of tetrafluoromethane besides hexafluoroethane are produced during production of aluminium using Hall-Héroult process. CF4 also is produced as product of the breakdown of more complex compounds such as halocarbons.
Health risks
Due to its density, tetrafluoromethane can displace air, creating an asphyxiation hazard in inadequately ventilated areas. Otherwise, it is normally harmless due to its stability.
Notes
References
References
- (1994). "CRC Handbook of Thermophysical and Themochemical Data". CRC Press.
- Abjean, R.. (15 July 1990). "Refractive index of carbon tetrafluoride (CF4) in the 300-140 nm wavelength range". Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment.
- (1971). "Reference values of the viscosity of twelve gases at 25°C". Transactions of the Faraday Society.
- Harrison, Jeremy J.. (Feb 2021). "New infrared absorption cross sections for the infrared limb sounding of carbon tetrafluoride (CF4)". [[Journal of Quantitative Spectroscopy and Radiative Transfer]].
- O'Hagan D. (February 2008). "Understanding organofluorine chemistry and in cations. An introduction to the C–F bond". Chemical Society Reviews.
- {{Greenwood&Earnshaw
- (2002). "Fluorine Compounds, Organic".
- K. Williams, K. Gupta, M. Wasilik. ''Etch Rates for Micromachining Processing – Part II'' J. Microelectromech. Syst., vol. 12, pp. 761–777, December 2003.
- (2005). "Low efficiency 2-dimensional position-sensitive neutron detector for beam profile measurement". Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment.
- (February 2018). "Changes in Atmospheric Constituents and in Radiative Forcing". [[Intergovernmental Panel on Climate Change]].
- (27 June 2016). "Climate change indicators - Atmospheric concentration of greenhouse gases - Figure 4". [[United States Environmental Protection Agency]].
- (1998). "Natural CF4 and SF6 on Earth". Geophysical Research Letters.
- Butler J. and Montzka S.. (2020). "The NOAA Annual Greenhouse Gas Index (AGGI)". [[NOAA]] Global Monitoring Laboratory/Earth System Research Laboratories.
- Cicerone, Ralph J.. (1979-10-05). "Atmospheric Carbon Tetrafluoride: A Nearly Inert Gas". Science.
- "Bond Energies".
- (2015). "An atmospheric photochemical source of the persistent greenhouse gas CF4". Geophysical Research Letters.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Carbon tetrafluoride — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report