From Surf Wiki (app.surf) — the open knowledge base
Carbamoyl chloride
Unstable functional group
Unstable functional group

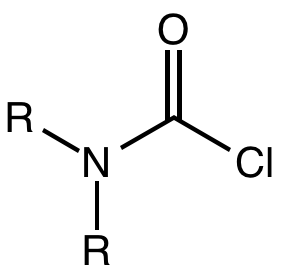
A carbamoyl chloride is the functional group with the formula R2NC(O)Cl. The parent carbamoyl chloride, H2NCOCl, is unstable, but many N-substituted analogues are known. Most examples are moisture sensitive, colourless, and soluble in nonpolar organic solvents. An example is dimethylcarbamoyl chloride (m.p. −90 °C and b.p. 93 °C). Carbamoyl chlorides are used to prepare a number of pesticides, e.g. carbofuran and aldicarb.
Production and examples
Carbamoyl chlorides are prepared by the reaction of an amine with phosgene: :2 R2NH + COCl2 → R2NCOCl + [R2NH2]Cl
They also arise by the addition of hydrogen chloride to isocyanates: :RNCO + HCl → RNHCOCl In this way, carbamoyl chlorides can be prepared with N-H functionality.
Reactions
In a reaction that is typically avoided, hydrolysis of carbamoyl chlorides gives carbamic acids: :R2NCOCl + H2O → R2NC(O)OH + HCl Owing to the influence of the amino group, these compounds are less hydrolytically sensitive than the usual acid chlorides. A related but more useful reaction is the analogous reaction with alcohols: :R2NCOCl + R'OH + C5H5N → R2NC(O)OR' + C5H5NHCl
References
References
- Peter Jäger, Costin N. Rentzea and Heinz Kieczka "Carbamates and Carbamoyl Chlorides" in Ullmann's Encyclopedia of Industrial Chemistry, 2012, Wiley-VCH, Weinheim. {{doi. 10.1002/14356007.a05_051
- (2002). "Nickel-Catalyzed Coupling of Aryl O-Carbamates with Grignard Reagents: 2,7-Dimethylnaphthalene". Org. Synth..
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Carbamoyl chloride — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report