From Surf Wiki (app.surf) — the open knowledge base
Cadmium sulfate
| | (monohydrate) | (octahydrate) | (anhydrous) | (monohydrate) | (octahydrate) | (anhydrous) | (monohydrate) | (octahydrate) | 1000 C (anhydrous) | 105 C (monohydrate, decomposes) | 40 C (octahydrate, decomposes) | {{ubl |anhydrous: | (0 C) | (25 C) | (99 C) | |{{ubl |monohydrate: | (25 C) | |{{ubl |octahydrate: |very soluble | (20 C) | (40 C) |orthorhombic (anhydrous) |monoclinic (mono & octahydrate) | NFPA-H = 3 | NFPA-F = 1 | NFPA-R = 1 | TLV-TWA = {{ubl | (total dust) | (respirable dust) |Fisher Scientific |Sigma-Aldrich |Cadmium acetate |Cadmium chloride |Cadmium nitrate |Zinc sulfate |Calcium sulfate |Magnesium sulfate
Cadmium sulfate is the name of a series of related inorganic compounds with the formula . The most common form is the monohydrate , but two other forms are known: the octahydrate () and the anhydrous salt (). All salts are colourless and highly soluble in water.
Preparation
Cadmium sulfate hydrate can be prepared by the reaction of cadmium metal or its oxide or hydroxide with dilute sulfuric acid: : :
The anhydrous material can be prepared using sodium persulfate: :
Applications
Cadmium sulfate is used widely for the electroplating of cadmium in electronic circuits. It is also a precursor to cadmium-based pigment such as cadmium sulfide. It is also used for electrolyte in a Weston standard cell as well as a pigment in fluorescent screens.
Structure

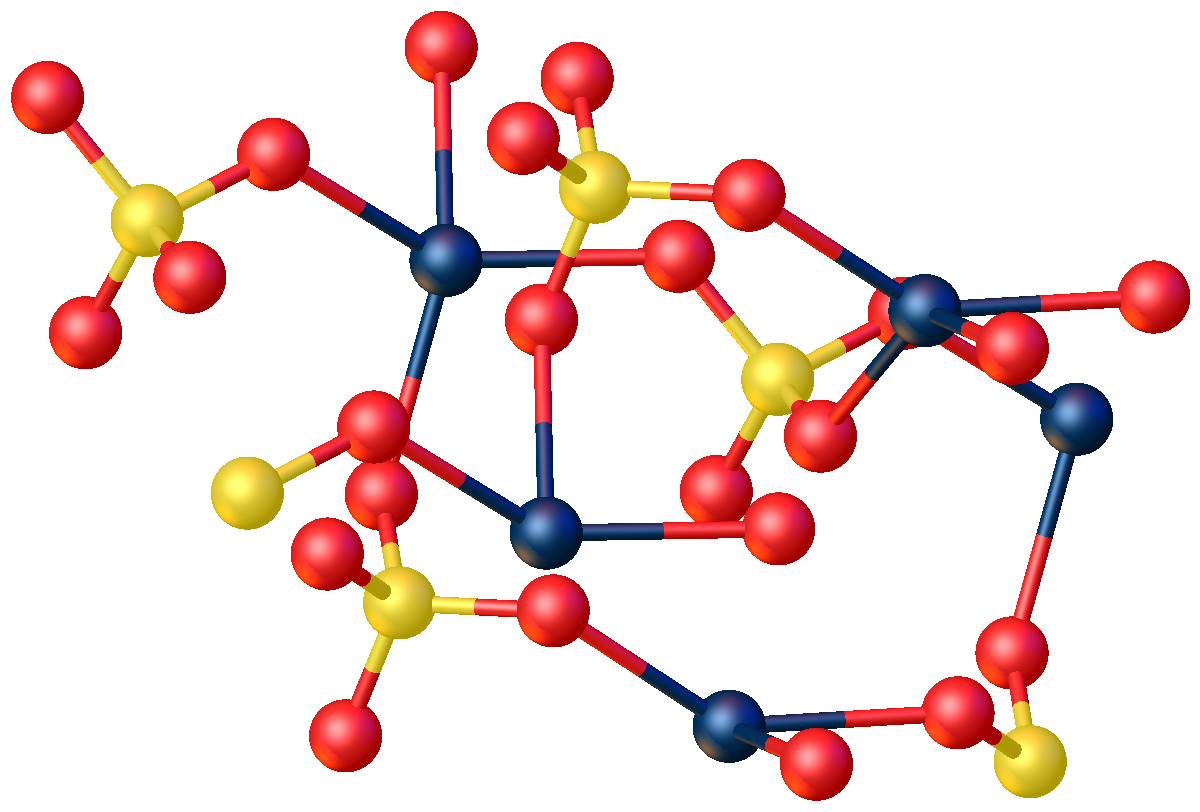
X-ray crystallography shows that is a typical coordination polymer. Each center has octahedral coordination geometry, being surrounded by four oxygen centers provided by four sulfate ligands and two oxygen centers from the bridging water ligands.
Occurrence
Cadmium sulfates occur as the following rare minerals drobecite (), voudourisite (monohydrate), and lazaridisite (the octahydrate).
Safety
Cadmium sulfate (along with cadmium and its compounds) are classified as group 1 (human carcinogens) by IARC and have been identified as causing lung and prostate cancer as well as mutagenic effects in humans.
References
References
- {{RubberBible87th
- Zumdahl, Steven S.. (2009). "Chemical Principles 6th Ed.". Houghton Mifflin Company.
- (20 May 2025). "Safety Data Sheet - Cadmium Sulfate". Sigma-Aldrich.
- (28 December 2021). "Safety Data Sheet - Cadmium Sulfate". ThermoFisher Scientific.
- (1980). "A Reinvestigation of the Crystal Structures of HgSO4 and CdSO4". Zeitschrift für Kristallographie - Crystalline Materials.
- (2015). "Crystal Structure of CdSO4(H2O): A Redetermination". Acta Crystallographica Section E.
- (February 1993). "Beryllium, Cadmium, Mercury, and Exposures in the Glass Manufacturing Industry". World Health Organization - International Agency for Research on Cancer.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Cadmium sulfate — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report