From Surf Wiki (app.surf) — the open knowledge base
Bipyridine
Group of chemical compounds
Group of chemical compounds
Bipyridines are a family of organic compounds with the formula (C5H4N)2, consisting of two pyridyl (C5H4N) rings. Pyridine is an aromatic nitrogen-containing heterocycle. The bipyridines are all colourless solids, which are soluble in organic solvents and slightly soluble in water. Bipyridines, especially the 4,4' isomer, are mainly of significance in pesticides.

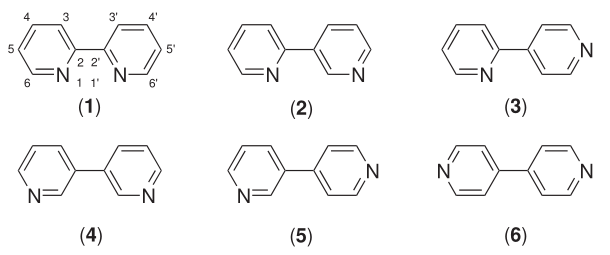
(1) 2,2′-bipyridine; (2) 2,3′-bipyridine; (3) 2,4′-bipyridine; (4) 3,3′-bipyridine; (5) 3,4′-bipyridine; (6) 4,4′-bipyridine]]
Six isomers of bipyridine exist, but two are prominent. 2,2′-bipyridine, also known as bipyridyl, dipyridyl, and dipyridine, is a popular ligand in coordination chemistry
2,2′-Bipyridine
Main article: 2,2'-Bipyridine{{!}}2,2′-Bipyridine
2,2′-Bipyridine (2,2′-bipy) is a chelating ligand that forms complexes with most transition metal ions that are of broad academic interest. Many of these complexes have distinctive optical properties, and some are of interest for analysis. Its complexes are used in studies of electron and energy transfer, supramolecular, and materials chemistry, and catalysis.
2,2′-Bipyridine is used in the manufacture of diquat.
4,4′-Bipyridine
Main article: 4,4'-Bipyridine{{!}}4,4′-Bipyridine
4,4′-Bipyridine (4,4′-bipy) is mainly used as a precursor to the N,N′-dimethyl-4,4′-bipyridinium dication commonly known as paraquat. This species is redox active, and its toxicity arises from its ability to interrupt biological electron transfer processes. Because of its structure, 4,4′-bipyridine can bridge between metal centres to give coordination polymers.
3,4′-Bipyridine
The 3,4′-bipyridine derivatives inamrinone and milrinone are used occasionally for short term treatment of congestive heart failure. They inhibit phosphodiesterase and thus increasing cAMP, exerting positive inotropy and causing vasodilation. Inamrinone causes thrombocytopenia. Milrinone decreases survival in heart failure.
References
References
- {{Ullmann. (2000)
- (2004). "Comprehensive Coordination Chemistry II: from Biology to Nanotechnology". Elsevier Pergamon.
- (2000). "Bipyridine: The Most Widely Used Ligand. A Review of Molecules Comprising at Least Two 2,2'-Bipyridine Units". Chemical Reviews.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Bipyridine — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report