From Surf Wiki (app.surf) — the open knowledge base
Annulyne
Conjugated hydrocarbon ring molecules with at least one triple bond
Conjugated hydrocarbon ring molecules with at least one triple bond
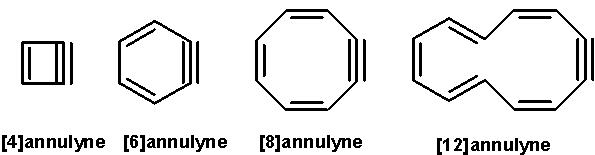
In organic chemistry, annulynes or dehydroannulenes are conjugated monocyclic hydrocarbons with alternating single and double bonds in addition to at least one triple bond.

They are related to annulenes, which only have alternating single and double bonds. The smallest member of this class is [4]annulyne but is never observed because the molecule carries too much angle strain. The next member is [6]annulyne or benzyne which is a reactive intermediate well known in organic chemistry. [8]annulyne is known to exist but quickly dimerizes or trimerizes; the compound has been trapped as its radical anion and observed by EPR spectroscopy. [10]annulyne, like [4]annulyne, only exists in theory.
[12]annulyne has been observed in 2005 by Stevenson et al. in solution by NMR spectroscopy at room temperature. Reaction of 1,5-hexadiyne and potassium tert-butoxide was reported to yield two isomers 5,9-di-trans-[12]-annulyne and 3,11-di-trans-[12]annulyne in a 1:1 ratio. The proposed reaction sequence involved an unspecified electron transfer reaction. A third single isomer 3,9-di-trans-[12]annulyne could be obtained in three steps from hexabromocyclododecane.
Unlike other annulynes, the [12]annulyne isomers were found to be very stable and did not self-condense. These annulynes reacted with potassium metal to form radical anions and dianions. The NMR chemical shifts of two internal protons of the dianion were negative and attributed to an aromatic ring current. The external proton next to the triple bond had a chemical shift of nearly 14, attributed to the positive charge of the potassium cation coordinated to it.
Two new [12]annulyne isomers were reported in 2008 by the same group.
The Stevenson findings were challenged by Christl and Hopf in 2009. They were unable to conceive a plausible mechanism and argued that based on the available spectroscopic data the products formed were in fact two linear 1,3-hexadien-5-yne isomers and not macrocycles. Computational analysis of this compound suggests valence isomerization to biphenyl is very exothermic but also with a high kinetic barrier.
[14]annulyne was reported in 1962 by Jackman et al.
Other annulynes
Dehydroannulenes with more than one triple bond were pioneered by Franz Sondheimer whose research group reported bisdehydro[12]annulene in 1962 and 1,5,9-tridehydro[12]annulene in 1966. A dehydrobenzo[14]annulene was reported in 2001 by Boydston and Haley
Applied research areas
Certain two-dimensional carbon networks containing a repeating dehydroannulene motif have been investigated for potential optoelectronics applications. Building Symmetric Two-Dimensional Two-Photon Materials Ajit Bhaskar, Ramakrishna Guda, Michael M. Haley, and Theodore Goodson III J. Am. Chem. Soc., 2006, 128 (43), pp 13972–13973
References
References
- ''1,2-Didehydro[10]annulenes: Structures, Aromaticity, and Cyclizations'' Armando Navarro-Vázquez and Peter R. Schreiner J. Am. Chem. Soc., 2005, 127 (22), pp 8150–8159 {{doi. 10.1021/ja0507968
- ''[12]Annulynes'' Matthew N. Gard, Matthew K. Kiesewetter, Richard C. Reiter, and Cheryl D. Stevenson [[J. Am. Chem. Soc.]]; '''2005'''; 127(46) pp 16143 - 16150; (Article) {{doi. 10.1021/ja053886l
- ''The Isomers of [12]Annulyne and their Reactive Relationships to Heptalene and Biphenyl'' Brad D. Rose, Richard C. Reiter, and Cheryl D. Stevenson [[Angew. Chem.]] '''2008''', 120, 8842–8846 {{doi. 10.1002/ange.200803863
- ''[12]Annulynes from 1,5-Hexadiyne and Potassium tert-Butoxide? [[Franz Sondheimer]]s Hexadienynes!'' Manfred Christl, Henning Hopf [[Angew. Chem. Int. Ed.]] '''2009''', 48 {{doi. 10.1002/anie.200901741
- ''Dehydro[12]annulenes: Structures, Energetics, and Dynamic Processes'' Lawrence A. Januar, Vivian Huynh, Taylor S. Wood, Claire Castro, and William L. Karney J. Org. Chem., 2011, 76 (2), pp 403–407 {{doi. 10.1021/jo1017537
- ''The Nuclear Magnetic Resonance Spectroscopy of a Series of Annulenes and Dehydro-annulenes'' L. M. Jackman, F. Sondheimer, Y. Amiel, D. A. Ben-Efraim, Y. Gaoni, R. Wolovsky, A. A. Bothner-By J. Am. Chem. Soc., 1962, 84 (22), pp 4307–4312 {{doi. 10.1021/ja00881a022
- ''Unsaturated Macrocyclic Compounds. XXXVI.1 The Synthesis of Two Isomers of Bisdehydro[12]annulene and Biphenylene from 1,5-Hexadiyne'' Reuven Wolovsky, [[Franz Sondheimer]] J. Am. Chem. Soc., 1965, 87 (24), pp 5720–5727 {{doi. 10.1021/ja00952a034
- ''1,5,9-Tridehydro[12]annulene'' F. Sondheimer, R. Wolovsky, P. J. Garratt, I. C. Calder J. Am. Chem. Soc., 1966, 88 (11), p 2610 {{doi. 10.1021/ja00963a057
- ''Diatropicity of Dehydrobenzo[14]annulenes: Comparative Analysis of the Bond-Fixing Ability of Benzene on the Parent 3,4,7,8,9,10,13,14-Octadehydro[14]annulene'' A. J. Boydston and Michael M. Haley Org. Lett., 2001, 3 (22), pp 3599–3601 {{doi. 10.1021/ol016764g
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Annulyne — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report