From Surf Wiki (app.surf) — the open knowledge base
Ammonium paratungstate
Ammonium paratungstate (or APT) is a white crystalline salt with the chemical formula . It is described as "the most important raw material for all other tungsten products."
Production
From tungsten ores
Tungsten ores, which are typically oxides, are digested in base to give solutions of tungstate together with many contaminating species. This crude extract is acidified and treated with sulfide to separate molybdenum trisulfide. Upon further acidification APT eventually crystallizes.
Laboratory methods
If a calcined is used, refluxing the ammonia solution is advisable to accelerate its dissolution.
Conversion to tungsten metal
Heating ammonium paratungstate to its decomposition temperature of 600 °C yields tungsten(VI) oxide, as described in this idealized equation :
From there, the trioxide is heated in an atmosphere of hydrogen, yielding elemental tungsten: :
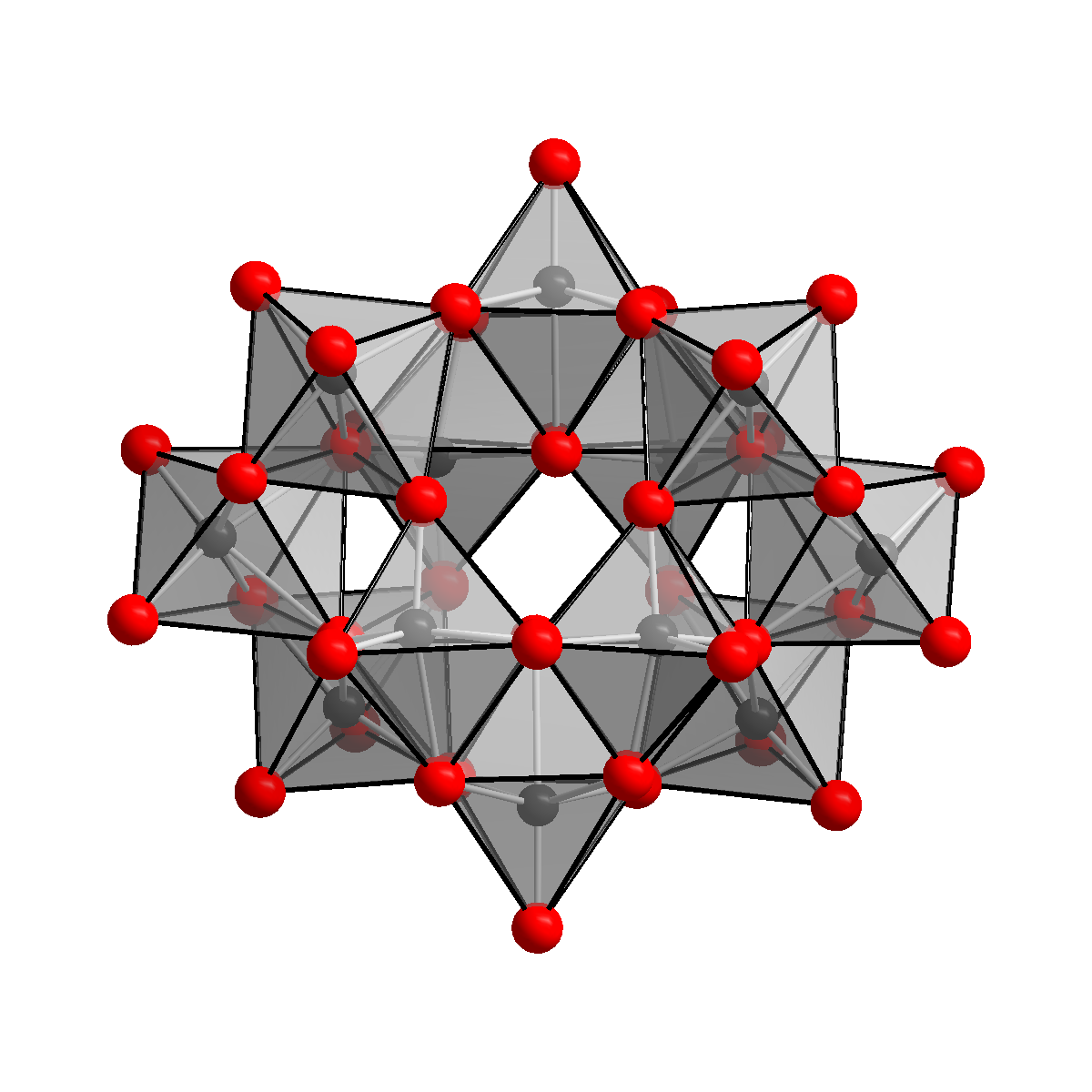
Structure

The anion in has been shown to be , containing two hydrogen atoms, keeping two hydrogen atoms inside the cage. The correct formula notation for ammonium paratungstate is therefore . The ion is known as the paratungstate B ion, as opposed to the paratungstate A ion, which has the formula , similar to the paramolybdate ion. The existence of the paratungstate A ion, however, could not be confirmed by NMR spectroscopy.
Before about 1930, there has been some dispute about the exact composition of the salt, and both and were proposed. O.W. Gibbs remarked about this: :"The alkali tungstates are numerous and unusually complex. Salts of essentially different formulae approach so closely in percentage composition, that the differences lie very near the unavoidable errors of analyses. The analyses are hardly sufficiently close to decide the question upon purely analytical grounds."
Other hydrates
When concentrating an ammoniacal solution of tungstic acid (i.e. hydrous ), the product obtained is ammonium paratungstate. Below 50 °C, the hexahydrate is formed, whereas when the temperature of the solution is above 50 °C, the pentahydrate or heptahydrate is formed. The former crystallizes as triclinic plates or prisms, whereas the latter as pseudorhombic needles. The tetrahydrate is most significant in a commercial sense. The decahydrate and the nonahydrate4)10(H2W12O42) * 9(H2O): anionic "isomorphism"Acta Crystallographica, Section E: Structure Reports Online (2002) 58, (*) p93-p94--are also known.
References
References
- "Tungsten, Tungsten Alloys, and Tungsten Compounds".
- D. J. Jones, "Practical aspects of Sintering Tungsten and Molybdenum" (as referenced in ''Comprehensive Inorganic Chemistry'', J. C. Bailar Jr. et al., p. 744, vol. 3, 1st edition 1973)
- (1972). "Die Kristallstruktur des Ammoniumparawolframat-tetrahydrats (NH4)10[H2W12O42]·4H2O". Zeitschrift für Kristallographie.
- Greenwood & Earnshaw, ''Chemistry of the Elements'', 2nd ed. 1997, pp. 1012–1014
- J. W. Mellor: ''Inorganic and Theoretical Chemistry'', vol. XI, p. 812–813, Longmans Green & Co. 1931
- (1971). "Die Struktur des Ammoniumparawolframates (NH4)10[H2W12O42].10H2O". Acta Crystallographica Section B.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Ammonium paratungstate — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report