From Surf Wiki (app.surf) — the open knowledge base
Adiponitrile
|NFPA-H = 4 |NFPA-F = 2 |NFPA-R = 1 hexanedihydrazide hexanedioyl dichloride hexanediamide 1,4-diisocyanobutane Adiponitrile is an organic compound with the chemical formula (CH2)4(CN)2. This viscous, colourless dinitrile is an important precursor to the polymer nylon 66. In 2005, about one million tonnes of adiponitrile were produced.
Production
Early methods
Because of the industrial value of adiponitrile, many methods have been developed for its synthesis. Early industrial methods started from furfural and later by the chlorination of butadiene to give 1,4-dichloro-2-butene, which with sodium cyanide, converts to 3-hexenedinitrile, which in turn can be hydrogenated to adiponitrile: :ClCH2CH=CHCH2Cl + 2 NaCN → NCCH2CH=CHCH2CN + 2 NaCl :NCCH2CH=CHCH2CN + H2 → NC(CH2)4CN Adiponitrile has also been produced from adipic acid, by dehydration of the diamide, but this is rarely employed.
Modern methods
After patent application in 2004, the majority of adiponitrile is prepared by the nickel-catalysed hydrocyanation of butadiene, as discovered at DuPont, pioneered by William C. Drinkard. The net reaction is: :CH2=CHCH=CH2 + 2 HCN → NC(CH2)4CN
The process involves several stages, the first of which involves monohydrocyanation (the addition of one molecule of HCN), affording isomers of pentenenitriles as well as 2- and 3-methylbutenenitriles. These unsaturated nitriles are subsequently isomerized to the 3-and 4-pentenenitriles. In the final stage, these pentenenitriles are subjected to a second hydrocyanation to produce adiponitrile, the anti-Markovnikov product, as well as 2-methylglutaronitrile, a useful byproduct.
Another side reaction is the alkene metathesis of 3-pentenenitrile to yield dicyanobutenes, which are readily hydrogenated to adiponitrile as described above.
The other major industrial method involves hydrodimerization, starting from acrylonitrile:{{cite journal |publication-date=July 17, 2017 :2 CH2=CHCN + 2 e− + 2 H+ → NCCH2CH2CH2CH2CN
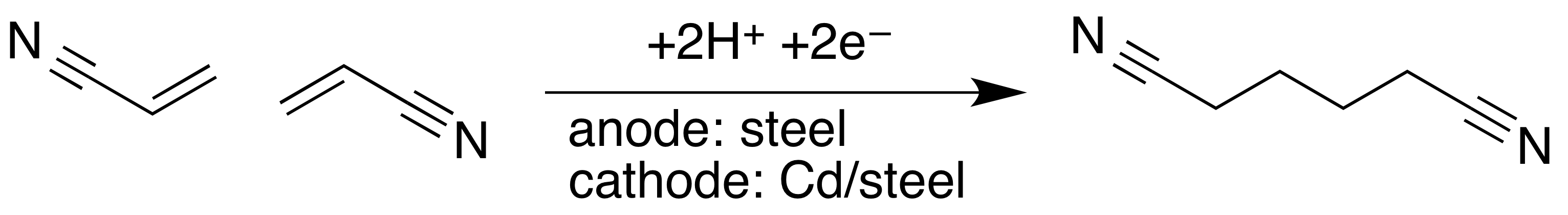
The electrolytic coupling of acrylonitrile was discovered at Monsanto Company.
Applications
Almost all adiponitrile is hydrogenated to hexane-1,6-diamine for the production of nylon: :NC(CH2)4CN + 4 H2 → H2N(CH2)6NH2 Like other nitriles, adiponitrile is susceptible to hydrolysis; however, the resulting adipic acid is less expensively prepared via other routes.
Production
In 2018, there existed approximately 1.5 million metric tons of capacity. The main producers of adiponitrile were:
- Ascend Performance Materials: Decatur, Alabama (US); 400 metric kilotons per year (kt/y), expanded to 580 kt/y by 2022
- Invista: Victoria, Texas and Orange, Texas, (US)
- Invista and BASF "Butachimie ADN plant": Chalampé (France); production to be increased from 100 kt/y in 2020 to 600 kt/y
- Asahi Kasei (Japan)
BASF closed the 128 kt/y ADN plant at Seal Sands in 2009.
In 2015, the Shandong Runxing New Material 100 kt/y plant suffered an explosion and was not reopened. In 2022, Invista plans to open a 300–400 kt/y plant in Shanghai.
Safety
The (median lethal dose) of adiponitrile is 300 mg/kg for oral ingestion by rats.
In 1990, ACGIH adopted a time-weighted average Threshold Limit Value of 2ppm for work-related skin exposure.
The NIOSH recommended skin exposure limit for a work-related time weighted average concentration is 4ppm (18 mg/m3).
Adiponitrile is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.
References
References
- (26 March 2005). "adiponitrile - Compound Summary". National Center for Biotechnology Information.
- {{PGCH. 0015
- "Archived copy".
- M. T. Musser, "Adipic Acid" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. {{doi. 10.1002/14356007.a01_269
- (1964). "Electrolytic Reductive Coupling". Journal of the Electrochemical Society.
- Robert A. Smiley "Hexamethylenediamine" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. {{doi. 10.1002/14356007.a12_629
- (7 Oct 2018). "The chemical industry is bracing for a nylon 6,6 shortage".
- "Ascend Finalizes $175 Million ADN Project in Alabama {{!}} CHEManager".
- (1 April 2009). "End of the line for Seal Sands". KHL Group.
- "INVISTA China ADN project receives final construction permit".
- 2009 TLVs and BEIs, [[American Conference of Governmental Industrial Hygienists]], Signature Publications, page 11 of 254.
- [https://www.cdc.gov/niosh/npg/npgd0015.html NIOSH Pocket Guide] NIOSH Publication 2005-149; September 2005
- "40 C.F.R.: Appendix A to Part 355—The List of Extremely Hazardous Substances and Their Threshold Planning Quantities". [[United States Government Publishing Office.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Adiponitrile — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report