From Surf Wiki (app.surf) — the open knowledge base
Acrosin
Mammalian protein found in Homo sapiens
Mammalian protein found in Homo sapiens
| Field | Value |
|---|---|
| Name | Acrosin |
| EC_number | 3.4.21.10 |
| CAS_number | 9068-57-9 |
| GO_code | 0004284 |
| image | 1FIW.png |
Enzyme Mechanism
Acrosin is a typical serine proteinase with trypsin-like specificity.{{Cite journal | doi-access = free
Biological Function
Acrosin is the major proteinase present in the acrosome of mature spermatozoa. It is stored in the acrosome in its precursor form, proacrosin. Upon stimulus, the acrosome releases its contents onto the zona pellucida. After this reaction occurs, the zymogen form of the protease is then processed into its active form, β-acrosin. The active enzyme then functions in the lysis of the zona pellucida, thus facilitating penetration of the sperm through the innermost glycoprotein layers of the ovum.
The importance of acrosin in the acrosome reaction has been contested. It has been found through genetic knockout experiments that mouse spermatozoa lacking β-acrosin (the active protease) still have the ability to penetrate the zona pellucida. Thus, some argue for its role in assisting in the dispersal of acrosomal contents following the acrosome reaction, while others demonstrate evidence for its role as a secondary binding protein between the spermatozoa and zona pellucida. Under the secondary binding protein hypothesis, acrosin could serve a role in binding to molecules on the zona pellucida, tethering the spermatozoa to the egg. This "tethering" would ensure penetration due to the applied motile force of the spermatozoa.
Acrosin regulation has been found to occur through protein C inhibitor (PCI). PCI is present in the male reproductive tract at 40x higher concentrations than in blood plasma.{{Cite journal
Structure
β-acrosin demonstrates a high degree of sequence identity (70-80%) between boar, bull, rat, guinea pig, mouse, and human isoforms. There exists a somewhat similar (27-35%) sequence identity between β-acrosin and other serine proteases such as trypsin and chymotrypsin. While most serine proteases are activated through one cleavage event, proacrosin requires processing at both the N and C-terminal domains. Proacrosin is first cleaved between Arg-22 and adjacent Valine to create a 22 residue light chain, and an active protease termed α-acrosin. This light chain remains associated with the heavy chain, cross-linked through two disulfide bonds to form a heterodimer. Following these N-terminal cleavage events, three cleavages at the C-terminal domain removes 70 residues, yielding β-acrosin. Acrosin has two sites which have been identified as possible N-glycosylation sites: Asn-2 and Asn-169.[[File:1FIW_Active_Site_Surface.png|thumb|450x450px|Acrosin active site surface contour, shown with competitive inhibitor benzamidine. Trp-215 "gatekeeper" residue is shown partially occluding the "top" (as pictured here) entrance to the binding cavity.]]
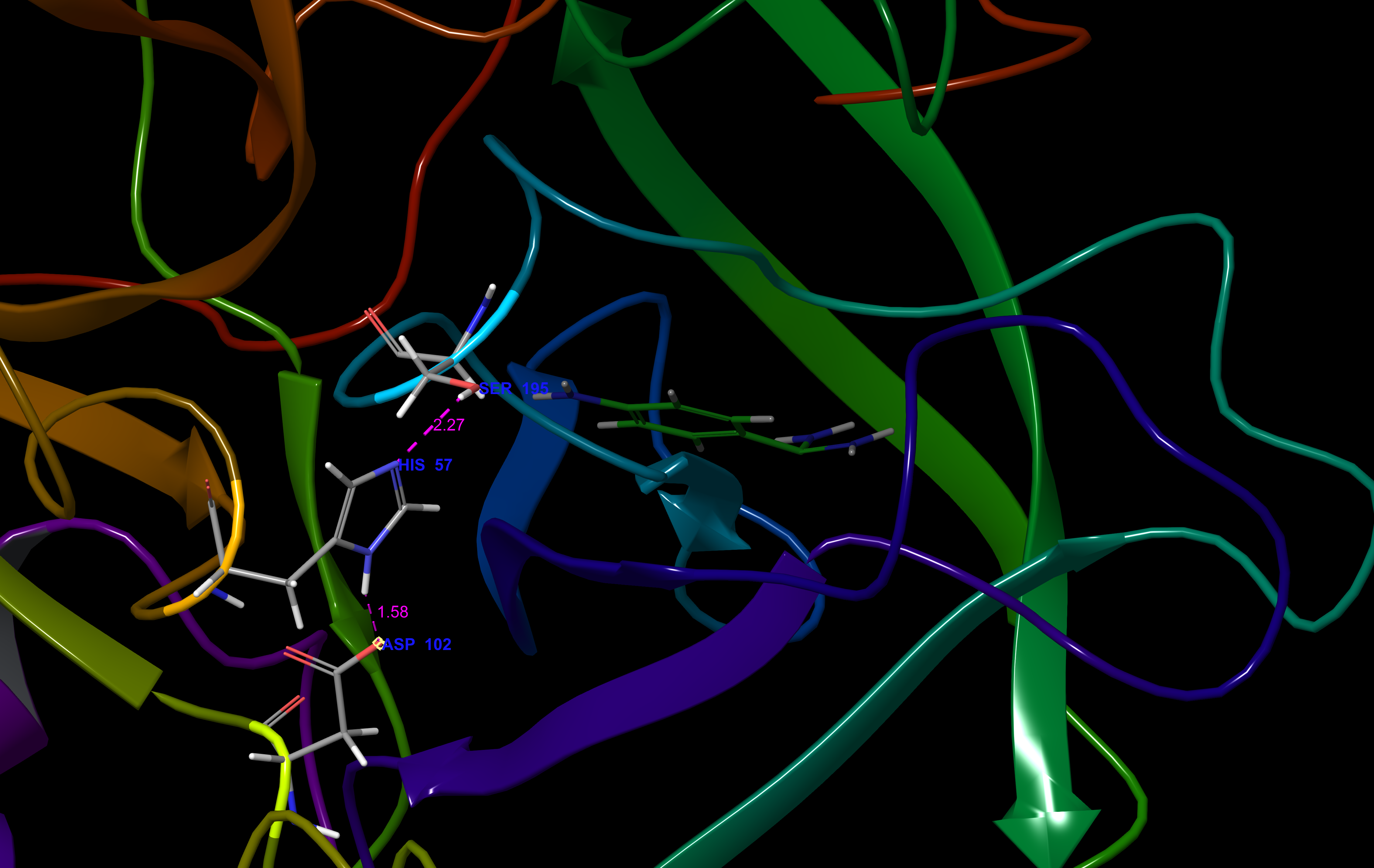
The catalytic triad consists of residues His-57, Asp-102, and Ser-195. These residues are found in a binding pocket that has been termed the "S1" pocket, consistent with the naming scheme that has been adopted for other proteases. The S1 pocket regulates acrosin's specificity for Arg and Lys substrates, with a conserved Trp-215 serving as a "gatekeeper" residue for the binding site entrance.
Disease and Pharmaceutical Relevance
While one study which utilized mice models indicated that acrosin is not a necessary component of zona pellucida penetration, other studies in humans have shown an association between low acrosomal proteinase activity and infertility. Other research groups have demonstrated a significant correlation between acrosin activity and sperm motility. In rabbit models, an intravaginal contraceptive device that secreted tetradecyl sodium sulfate, a known inhibitor of acrosin and hyaluronidases, had a complete contraceptive effect. Although its exact mechanism of action is not entirely clear, acrosin could thus serve as a novel target for contraceptive agents. Acrosin may represent as a uniquely druggable target due to its location and high cellular specificity.{{Cite journal
Moreover, as serine proteases are important in the potentiation of HIV, research has found that an acrosin inhibitor, 4'-acetamidophenyl 4-guanidinobenzoate, possess the ability to inhibit HIV infection in virus-inoculated lymphocytes. This suggests the further role of acrosin inhibitors as potentially viable agents in the prevention of HIV transmission.
References
References
- (Jan 1990). "Molecular cloning of human preproacrosin cDNA". Human Genetics.
- (2002). "Role of acrosomal matrix proteases in sperm-zona pellucida interactions". Human Reproduction Update.
- T. Baba, S. Azuma, S. Kashiwabara, Y. Toyoda. Sperm from mice carrying a targeted mutation of the acrosin gene can penetrate the oocyte zona pellucida and effect fertilization" ''J. Biol. Chem.'' 1994; 269, pp. 31845–31849
- K. Yamagata, T. Baba, ''et al.'' Acrosin accelerates the dispersal of sperm acrosomal proteins during acrosome reaction" ''J. Biol. Chem.'' 1998; 273, pp. 10470–10474
- R. Jones, C.R. Brown. Identification of a zona-binding protein from boar spermatozoa as proacrosin. Expl" ''Cell Res'' 1987; 171, pp. 505–508
- R. Jones. Interaction of zona pellucida glycoproteins, sulphated carbohydrates and synthetic polymers with proacrosin, the putative egg-binding protein from mammalian spermatozoa" ''Development'' 1991; 111, pp. 1155–1163
- D.P. Green. The head shapes of some mammalian spermatozoa and their possible relationship to the shape of the penetration slit through the zona pellucida. J. Reprod. Fertil., 83 (1988), pp. 377–387
- I. Schechter, A. Berger. On the size of the active site in proteases. I. Papain. Biochim. Biophys. Res. Commun, 27 (1967), pp. 157–162.
- (1990). "Further fractionation of the glycoprotein families of porcine zona pellucida by anion exchange HPLC and some characterization of the separated fractions". J. Biochem..
- S. Shimizu, M. Tsuji, J. Dean. In vitro biosynthesis of three sulphated glycoproteins of murine zonae pellucidae by oocytes grown in follicle culture" ''J. Biol. Chem.'' 1983; 258, pp. 5858–5863
- (1996). "Inhibition of sperm-zona binding by suramin, a potential "lead" compound for design of new anti-fertility agents". Mol. Hum. Rep..
- (Oct 1988). "Acrosomal proteinase activity of human spermatozoa and relation of results to semen quality". Hum Reprod.
- Tummon I.S.; Yuzpe A.A.; Daniel S.A.; Deutsch A. Total acrosin activity correlates with fertility potential after fertilization in vitro" ''Fertil Steril'' 1991 Nov;56(5):933-8.
- (Sep 2000). "Determination of sperm acrosin activity for evaluation of male fertility". Asian J Androl.
- Burck P.J., Zimmerman R.E. An intravaginal contraceptive device for the delivery of an acrosin and hyaluronidase inhibitor" ''Fertil Steril'' 1984 Feb;41(2):314-8.
- (May 1995). "Acrosin inhibitor, 4'-acetamidophenyl 4-guanidinobenzoate, an experimental vaginal contraceptive with anti-HIV activity". Contraception.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Acrosin — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report