From Surf Wiki (app.surf) — the open knowledge base
Acid strength
Measure of the tendency of an acid to dissociate
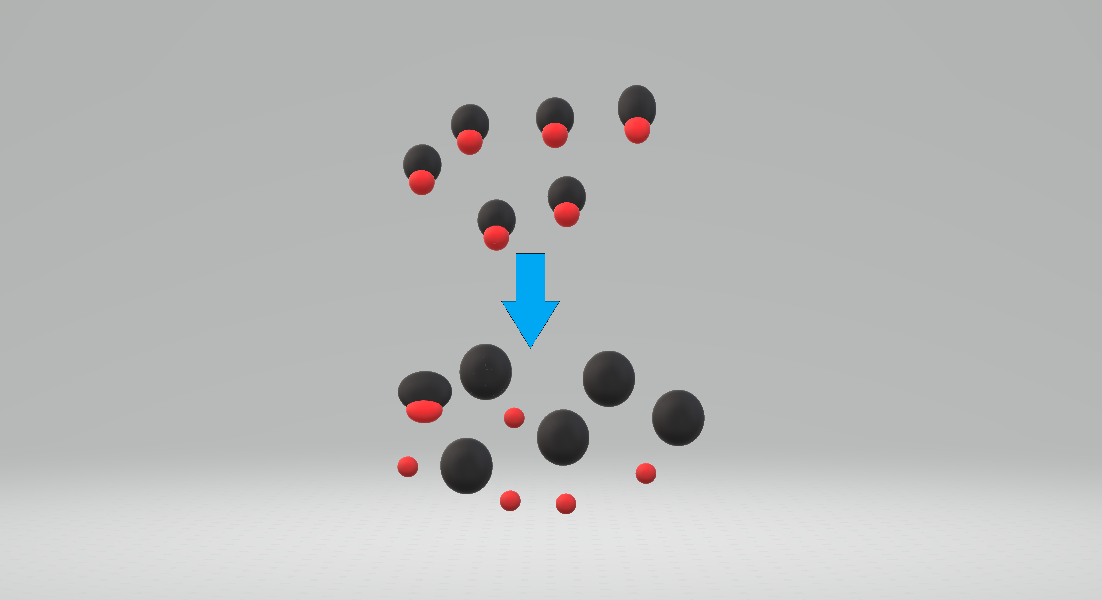
Measure of the tendency of an acid to dissociate
Acid strength is the tendency of an acid, symbolised by the chemical formula , to dissociate into a proton, , and an anion, . The dissociation or ionization of a strong acid in solution is effectively complete, except in its most concentrated solutions.
:
Examples of strong acids are hydrochloric acid (), perchloric acid (), nitric acid () and sulfuric acid ().
A weak acid is only partially dissociated, or is partly ionized in water with both the undissociated acid and its dissociation products being present, in solution, in equilibrium with each other.
:
Acetic acid () is an example of a weak acid. The strength of a weak acid is quantified by its acid dissociation constant, K_a value.
The strength of a weak organic acid may depend on substituent effects. The strength of an inorganic acid is dependent on the oxidation state for the atom to which the proton may be attached. Acid strength is solvent-dependent. For example, hydrogen chloride is a strong acid in aqueous solution, but is a weak acid when dissolved in glacial acetic acid.
Measures of acid strength
The usual measure of the strength of an acid is its acid dissociation constant (K_a), which can be determined experimentally by titration methods. Stronger acids have a larger K_a and a smaller logarithmic constant (\mathrm{p}K_a = - \log K_a) than weaker acids. The stronger an acid is, the more easily it loses a proton, . Two key factors that contribute to the ease of deprotonation are the polarity of the bond and the size of atom , which determine the strength of the bond. Acid strengths also depend on the stability of the conjugate base.
While the K_a value measures the tendency of an acidic solute to transfer a proton to a standard solvent (most commonly water or DMSO), the tendency of an acidic solvent to transfer a proton to a reference solute (most commonly a weak aniline base) is measured by its Hammett acidity function, the H_0 value. Although these two concepts of acid strength often amount to the same general tendency of a substance to donate a proton, the K_a and H_0 values are measures of distinct properties and may occasionally diverge. For instance, hydrogen fluoride, whether dissolved in water (\mathrm{p}K_a = 3.2) or DMSO (\mathrm{p}K_a = 15), has \mathrm{p}K_a values indicating that it undergoes incomplete dissociation in these solvents, making it a weak acid. However, as the rigorously dried, neat acidic medium, hydrogen fluoride has an H_0 value of –15,{{Cite book
When the acidic medium in question is a dilute aqueous solution, the H_0 is approximately equal to the pH value, which is a negative logarithm of the concentration of aqueous in solution. The pH of a simple solution of an acid in water is determined by both K_a and the acid concentration. For weak acid solutions, it depends on the degree of dissociation, which may be determined by an equilibrium calculation. For concentrated solutions of acids, especially strong acids for which pH H_0 value is a better measure of acidity than the pH.
Strong acids

A strong acid is an acid that dissociates according to the reaction : where S represents a solvent molecule, such as a molecule of water or dimethyl sulfoxide (DMSO), to such an extent that the concentration of the undissociated species is too low to be measured. For practical purposes a strong acid can be said to be completely dissociated. An example of a strong acid is perchloric acid. : (in aqueous solution)
Any acid with a \mathrm{p}K_a value which is less than about −2 behaves as a strong acid. This results from the very high buffer capacity of solutions with a pH value of 1 or less and is known as the leveling effect.
The following are strong acids in aqueous and dimethyl sulfoxide solution. As mentioned above, because the dissociation is so strongly favored, the concentrations of and thus the values of \mathrm{p}K_a cannot be measured experimentally. The values in the following table are average values from as many as 8 different theoretical calculations. :{| class="wikitable"
| +Estimated pKa values |
|---|
| !Acid!!Formula!! in water |
| - |
| Hydrochloric acid |
| - |
| Hydrobromic acid |
| - |
| Hydroiodic acid |
| - |
| Triflic acid |
| - |
| Perchloric acid |
| } |
| Also, in water |
- Nitric acid \mathrm{p}K_a = -1.6
- Sulfuric acid (first dissociation only, \mathrm{p}K_{a1} \approx -3 )
The following can be used as protonators in organic chemistry
- Fluoroantimonic acid
- Magic acid
- Carborane superacid
- Fluorosulfuric acid (\mathrm{p}K_a = -6.4)
Sulfonic acids, such as p-toluenesulfonic acid (tosylic acid) are a class of strong organic oxyacids.{{cite journal |doi-access=free
Weak acids
Main article: Acid dissociation constant
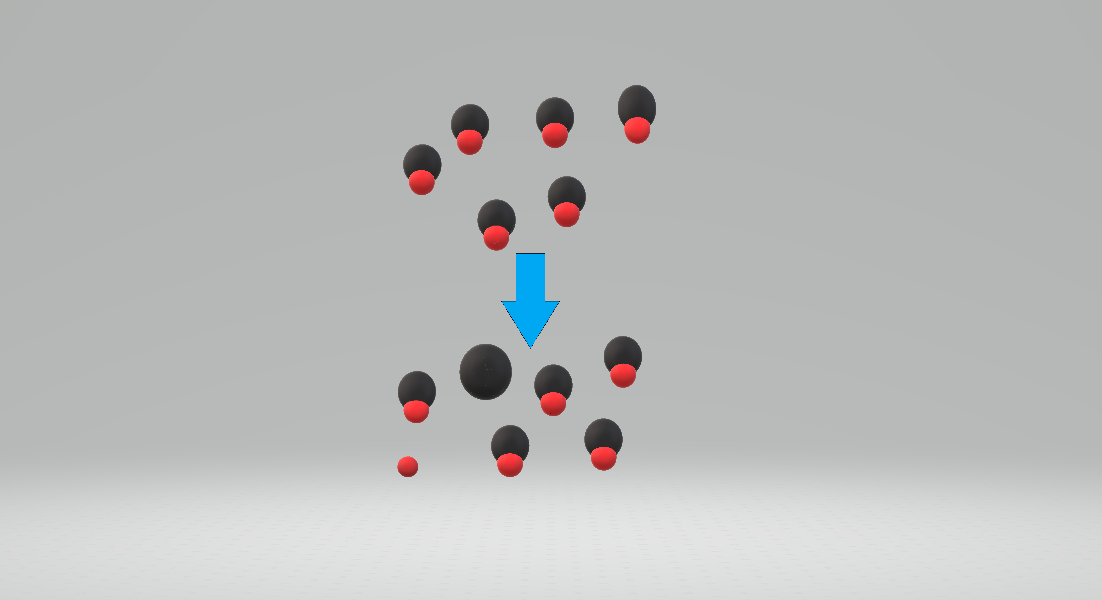
A weak acid is a substance that partially dissociates or partly ionizes when it is dissolved in a solvent. In solution, there is an equilibrium between the acid, , and the products of dissociation. : The solvent (e.g. water) is omitted from this expression when its concentration is effectively unchanged by the process of acid dissociation. The strength of a weak acid can be quantified in terms of a dissociation constant, K_a , defined as follows, where [X] signifies the concentration of a chemical moiety, . K_a = \frac{[\mathrm{H}^+][\mathrm{A}^-]}{[\mathrm{HA}]} When a numerical value of K_a is known it can be used to determine the extent of dissociation in a solution with a given concentration of the acid, T_H, by applying the law of conservation of mass. \begin{align}T_H &=[\mathrm{H}^+]+[\mathrm{HA}]\ &=[\mathrm{H}^+]+\frac{[\mathrm{A}^-][\mathrm{H}^+]}{K_a}\ &=[\mathrm{H}^+]+\frac{[\mathrm{H}^+]^2}{K_a} \end{align} where T_H is the value of the analytical concentration of the acid. When all the quantities in this equation are treated as numbers, ionic charges are not shown and this becomes a quadratic equation in the value of the hydrogen ion concentration value, . \frac{[\mathrm{H}^+]^2}{K_a} + [\mathrm{H}^+] - T_H = 0 This equation shows that the pH of a solution of a weak acid depends on both its K_a value and its concentration. Typical examples of weak acids include acetic acid and phosphorous acid. An acid such as oxalic acid () is said to be dibasic because it can lose two protons and react with two molecules of a simple base. Phosphoric acid () is tribasic.
For a more rigorous treatment of acid strength see acid dissociation constant. This includes acids such as the dibasic acid succinic acid, for which the simple method of calculating the pH of a solution, shown above, cannot be used.
Experimental determination
Main article: Acid dissociation constant#Experimental determination
The experimental determination of a \mathrm{p}K_a value is commonly performed by means of a titration.{{cite book : until only the deprotonated species, , remains in solution. At each point in the titration pH is measured using a glass electrode and a pH meter. The equilibrium constant is found by fitting calculated pH values to the observed values, using the method of least squares.
Conjugate acid/base pair
It is sometimes stated that "the conjugate of a weak acid is a strong base". Such a statement is incorrect. For example, acetic acid is a weak acid which has a K_a = 1.75 \times 10^{-5}. Its conjugate base is the acetate ion with K_b = 10^{-14} and K_a = 5.7 \times 10^{-10} (from the relationship K_a \times K_b = 10^{-14}), which certainly does not correspond to a strong base. The conjugate of a weak acid is often a weak base and vice versa.
Acids in non-aqueous solvents
The strength of an acid varies from solvent to solvent. An acid which is strong in water may be weak in a less basic solvent, and an acid which is weak in water may be strong in a more basic solvent. According to Brønsted–Lowry acid–base theory, the solvent S can accept a proton. : For example, hydrochloric acid is a weak acid in solution in pure acetic acid, , which is less basic than water. : The extent of ionization of the hydrohalic acids decreases in the order HI HBr HCl. Acetic acid is said to be a differentiating solvent for the three acids, while water is not.
An important example of a solvent which is more basic than water is dimethyl sulfoxide, DMSO, . A compound which is a weak acid in water may become a strong acid in DMSO. Acetic acid is an example of such a substance. An extensive bibliography of \mathrm{p}K_a values in solution in DMSO and other solvents can be found at Acidity–Basicity Data in Nonaqueous Solvents.
Superacids are strong acids even in solvents of low dielectric constant. Examples of superacids are fluoroantimonic acid and magic acid. Some superacids can be crystallised.{{cite journal |last1=Zhang |first1=Dingliang |last2=Rettig |first2=Stephen J. |last3=Trotter |first3=James |last4=Aubke |first4=Friedhelm |title=Superacid Anions: Crystal and Molecular Structures of Oxonium Undecafluorodiantimonate(V), [H3O][Sb2F11], Cesium Fluorosulfate, CsSO3F, Cesium Hydrogen Bis(fluorosulfate), Cs[H(SO3F)2], Cesium Tetrakis(fluorosulfato)aurate(III), Cs[Au(SO3F)4], Cesium Hexakis(fluorosulfato)platinate(IV), Cs2[Pt(SO3F)6], and Cesium Hexakis(fluorosulfato)antimonate(V), Cs[Sb(SO3F)6]
Lewis acids reacting with Lewis bases in gas phase and non-aqueous solvents have been classified in the ECW model, and it has been shown that there is no one order of acid strengths. The relative acceptor strength of Lewis acids toward a series of bases, versus other Lewis acids, can be illustrated by C-B plots. It has been shown that to define the order of Lewis acid strength at least two properties must be considered. For the qualitative HSAB theory the two properties are hardness and strength while for the quantitative ECW model the two properties are electrostatic and covalent.
Factors determining acid strength
The inductive effect
In organic carboxylic acids, an electronegative substituent can pull electron density out of an acidic bond through the inductive effect, resulting in a smaller \mathrm{p}K_a value. The effect decreases, the further the electronegative element is from the carboxylate group, as illustrated by the following series of halogenated butanoic acids.
| Structure | Name | p*K*a |
|---|---|---|
| [[File:2chlorobutanoic.png]] | 2-chlorobutanoic acid | 2.86 |
| [[File:3chlorobutanoic.png]] | 3-chlorobutanoic acid | 4.0 |
| [[File:4chlorobutanoic.png]] | 4-chlorobutanoic acid | 4.5 |
| [[File:butanoic.png]] | butanoic acid | 4.5 |
Effect of oxidation state
In a set of oxoacids of an element, \mathrm{p}K_a values decrease with the oxidation state of the element. The oxoacids of chlorine illustrate this trend.
| Structure | Name | Oxidation | ||
|---|---|---|---|---|
| state | p*K*a | |||
| 100px | perchloric acid | 7 | −8† | |
| 100px | chloric acid | 5 | −1 | |
| 100px | chlorous acid | 3 | 2.0 | |
| 100px | hypochlorous acid | 1 | 7.53 |
† theoretical
References
References
- Miessler, Gary L.. (1999). "Inorganic chemistry". Prentice-Hall.
- Porterfield, William W.. (1984). "Inorganic chemistry: an unified approach". Addison Wesley Pub. Co.
- (2016). "Acidity of strong acids in water and dimethyl sulfoxide". J. Phys. Chem. A.
- Bell, R. P.. (1973). "The Proton in Chemistry". Cornell University Press.
- {{Housecroft2nd
- Waters, Charlie. (2023-12-21). "Superacid: The Strongest Acids in the World".
- [[George A. Olah]], Schlosberg RH. (1968). "Chemistry in Super Acids. I. Hydrogen Exchange and Polycondensation of Methane and Alkanes in FSO3H–SbF5 ("Magic Acid") Solution. Protonation of Alkanes and the Intermediacy of CH5+ and Related Hydrocarbon Ions. The High Chemical Reactivity of "Paraffins" in Ionic Solution Reactions". Journal of the American Chemical Society.
- (1996). "The ECW Model". Journal of Chemical Education.
- Laurence, C. and Gal, J-F. Lewis Basicity and Affinity Scales, Data and Measurement, (Wiley 2010) pp 50-51 {{ISBN. 978-0-470-74957-9
- (1977). "Graphical display of the enthalpies of adduct formation for Lewis acids and bases". Journal of Chemical Education.
- [https://www.scribd.com/doc/6792576/638478 Section 8: Electrolytes, Electromotive forces and Chemical Equilibrium]
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Acid strength — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report