From Surf Wiki (app.surf) — the open knowledge base
Acetogenin
Group of chemical compounds
Group of chemical compounds
Acetogenins are a class of polyketide natural products found in plants of the family Annonaceae. They are characterized by linear 32- or 34-carbon chains containing oxygenated functional groups including hydroxyls, ketones, epoxides, tetrahydrofurans and tetrahydropyrans. They are often terminated with a lactone or butenolide. Over 400 members of this family of compounds have been isolated from 51 different species of plants. Many acetogenins are characterized by neurotoxicity.
Examples include:
- Annonacin
- Annonins
- Bullatacin
- Uvaricin
Structure
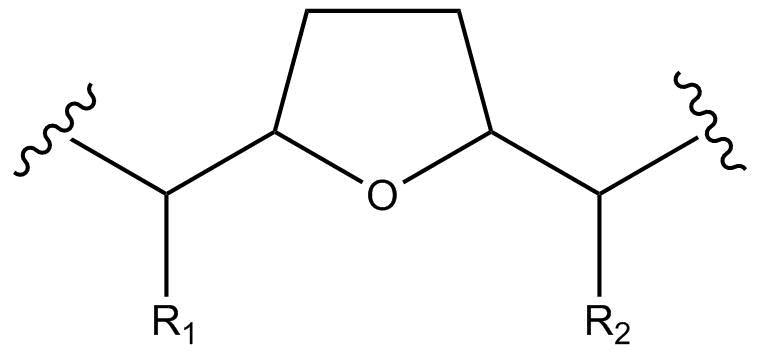

Structurally, acetogenins are a series of C-35/C-37 compounds usually characterized by a long aliphatic chain bearing a terminal methyl-substituted α,β-unsaturated γ-lactone ring, as well as one to three tetrahydrofuran (THF) rings. These THF rings are located along the hydrocarbon chain, along with a number of oxygenated moieties (hydroxyls, acetoxyls, ketones, epoxides) and/or double bonds.
| Compound | R1 | R2 | R3 | R4 | R5 | 4-deoxyannoreticuin | Annonacin | Annopentocin A | Dispalin | Donnaienin C | Goniotetracin | Muricoreacin | Tonkinin A | Uvaribonone |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OH | OH | H | H | H | ||||||||||
| OH | OH | H | OH | H | ||||||||||
| OH | H | H | OH | H | ||||||||||
| OAc | OH | H | OH | H | ||||||||||
| OH | OH | H | OAc | OH | ||||||||||
| OH | OH | H | OH | H | ||||||||||
| OH | H | H | OH | H | ||||||||||
| OH | OH | O | H | H | ||||||||||
| OH | OAc | O | H | H |
Research
Acetogenins have been investigated for their biological properties, but are a concern due to neurotoxicity. Purified acetogenins and crude extracts of the common North American pawpaw (Asimina triloba) or the soursop (Annona muricata) remain under laboratory studies.
Mechanism of action
Acetogenins inhibit NADH dehydrogenase, a key enzyme in energy metabolism.
References
References
- (2008). "Recent Progress on the Total Synthesis of Acetogenins from Annonaceae". Beilstein Journal of Organic Chemistry.
- (2005). "Acetogenins from Annonaceae: Recent Progress in Isolation, Synthesis and Mechanisms of Action". Natural Product Reports.
- (2016). "Acetogenins from Annonaceae".
- (1999). "Annonaceous Acetogenins: Recent Progress". Journal of Natural Products.
- Ana V. Coria-Téllez. (2018). "Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity". Arabian Journal of Chemistry.
- (2011). "Annonaceae fruits and parkinsonism risk: Metabolisation study of annonacin, a model neurotoxin; evaluation of human exposure". Toxicology Letters.
- (2011). "Nuts and Seeds in Health and Disease Prevention". Academic Press.
- Robert A. Levine. (2015). "Determination of Neurotoxic Acetogenins in Pawpaw (Asimina triloba) Fruit by LC-HRMS". J. Agric. Food Chem..
- (2012). "Annonacin in ''Asimina triloba'' fruit: Implication for neurotoxicity". Neurotoxicology.
- (2008). "Paw paw and cancer: annonaceous acetogenins from discovery to commercial products.". Journal of Natural Products.
- (1 July 1994). "Natural substances (acetogenins) from the family Annonaceae are powerful inhibitors of mitochondrial NADH dehydrogenase (Complex I).". Biochemical Journal.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Acetogenin — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report