From Surf Wiki (app.surf) — the open knowledge base
2-succinyl-6-hydroxy-2,4-cyclohexadiene-1-carboxylate synthase
InterPro Family
InterPro Family
| Field | Value |
|---|---|
| Name | 2-succinyl-6-hydroxy-2,4-cyclohexadiene-1-carboxylate synthase |
| EC_number | 4.2.99.20 |
| CAS_number | 122007-88-9 |
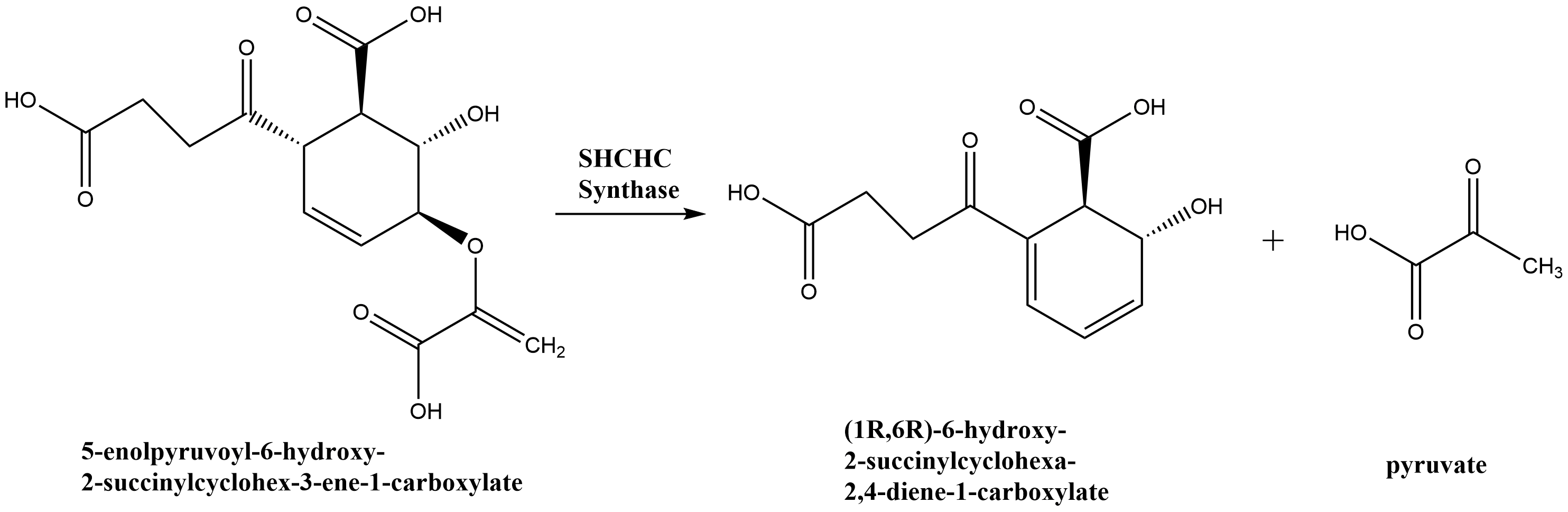
2-Succinyl-6-hydroxy-2,4-cyclohexadiene-1-carboxylate synthase (EC 4.2.99.20), also known as SHCHC synthase is encoded by the menH gene in Escherichia coli and functions in the synthesis of vitamin K. The specific step in the synthetic pathway that SHCHC synthase catalyzes is the conversion of 5-enolpyruvoyl-6-hydroxy-2-succinylcyclohex-3-ene-1-carboxylate to (1R,6R)-6-hydroxy-2-succinylcyclohexa-2,4-diene-1-carboxylate and pyruvate.
Background
Vitamin K is a fat soluble vitamin known to aid in blood clotting. It is recommended that all newborns receive an injection of vitamin K in order to prevent excessive bleeding of the brain after birth. There are two major forms of vitamin K that occur naturally. Phylloquinone, also known as K1, is synthesized by plants and is the major form of vitamin K in the diet. Menaquinone, K2, includes a range of forms that are synthesized by bacteria in the gut.
Vitamin K is synthesized from the molecule chorismate in a nine step conversion process. SHCHC synthase catalyzes the third step in the process.
Chemistry
Reaction scheme

Enzyme structure
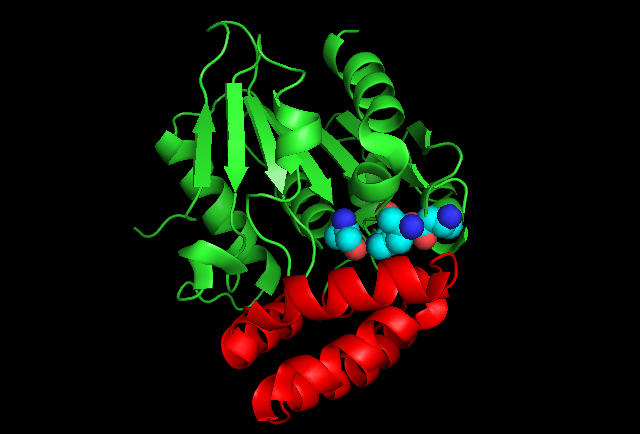
Energetic analysis shows that SHCHC synthase has a low energetic burden for catalytic activity. This means the enzyme is more prone to mutation and is one of the most diverse enzymes in the vitamin K synthetic pathway. Only fifteen amino acid residues are absolutely conserved across mutations of the enzyme.
Catalytic mechanism
The active site contains a catalytic triad of syrine, histine and arginine, which is conserved across all mutants and is proposed to initiate the reaction. The triad residues are located at Ser86, Asp210, and His232. This triad is proposed to catalyze a proton extraction which triggers a transfer of electrons leading to the elimination of pyruvate and formation of SHCHC. Originally, it was proposed that the transition state was stabilized by a nontraditional oxyanion hole. Now a traditional oxyanion hole is favored, but not definitive.
Reaction mechanism

Cofactors and alternate reactions
SHCHC synthase is unaffected by traditional cofactors such as divalent metal ions and EDTA. The enzyme is fairly specific and only acts on SEPHCHC and close derivatives.
Controversy
MenH (SHCHC synthase) was previously thought to be a thioesterase involved in hydrolyzing DHNA-CoA in a later step of menaquinone synthesis. In 2008, it was determined that MenH has poor catalytic activity toward palmitoyl-CoA, casting doubt on its role as a thioesterase. Direct analysis confirmed that MenH is unable to hydrolyze DHNA-CoA. In 2009, it was proposed that a dedicated hotdog fold thioesterase would be needed to catalyze the hydrolysis of DHNA-CoA. A protein was identified in 2013 that could fit this role.
References
References
- (March 2008). "Identification and characterization of (1R,6R)-2-succinyl-6-hydroxy-2,4-cyclohexadiene-1-carboxylate synthase in the menaquinone biosynthesis of ''Escherichia coli''". Biochemistry.
- (2014-07-01). "Information on EC 4.2.99.20 - 2-succinyl-6-hydroxy-2,4-cyclohexadiene-1-carboxylate synthase". TU Braunschweig.
- (2014-11-30). "Micronutrient Information Center". Linus Pauling Institute.
- van Oostende C, Widhalm JR, Furt F, Ducluzeau AL, Basset GJC (2011) Phylloquinone (Vitamin K1): function, enzymes and genes. in Advances in Botanical Research, eds Fabrice Rébeillé and [[Roland Douce]], 59: 229-61, Academic Press (Amsterdam).
- (2013-04-18). "Crystal structures of E. coli native MenH and two active site mutants". PLOS ONE.
- (May 2014). "Molecular basis of the general base catalysis of an α/β-hydrolase catalytic triad". The Journal of Biological Chemistry.
- (July 2009). "Catalytic mechanism of SHCHC synthase in the menaquinone biosynthesis of Escherichia coli: identification and mutational analysis of the active site residues". Biochemistry.
- (April 2009). "A dedicated thioesterase of the Hotdog-fold family is required for the biosynthesis of the naphthoquinone ring of vitamin K1". Proceedings of the National Academy of Sciences of the United States of America.
- (June 2013). "Identification of a hotdog fold thioesterase involved in the biosynthesis of menaquinone in Escherichia coli". Journal of Bacteriology.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about 2-succinyl-6-hydroxy-2,4-cyclohexadiene-1-carboxylate synthase — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report