From Surf Wiki (app.surf) — the open knowledge base
2,3-sigmatropic rearrangement
Class of chemical reaction
Class of chemical reaction
2,3-Sigmatropic rearrangements are a type of sigmatropic rearrangements and can be classified into two types. Rearrangements of allylic sulfoxides, amine oxides, selenoxides are neutral. Rearrangements of carbanions of allyl ethers are anionic. The general scheme for this kind of rearrangement is:
Atom Y may be sulfur, selenium, or nitrogen. If Y is nitrogen, the reaction is referred to as the Sommelet–Hauser rearrangement if a quaternary ammonium salt is involved or the aza-Wittig reaction if an alpha-metalated tertiary amine is involved; if Y is oxygen, then it is called a 2,3-Wittig rearrangement (not to be confused with the well-known Wittig reaction, which involves a phosphonium ylide). If Y is sulfur, the product can be treated with a thiophil to generate an allylic alcohol in what is known as the Mislow–Evans rearrangement.
A [2,3]-rearrangement may result in carbon-carbon bond formation. It can also be used as a ring-expansion reaction.

Stereoselectivity
2,3-sigmatropic rearrangements can offer high stereoselectivity. At the newly formed double bond there is a strong preference for formation of the E-alkene or trans isomer product. The stereochemistry of the newly formed C-C bond is harder to predict. It can be inferred from the five-membered ring transition state. Generally, the E-alkene will favor the formation of anti product, while Z-alkene will favor formation of syn product.
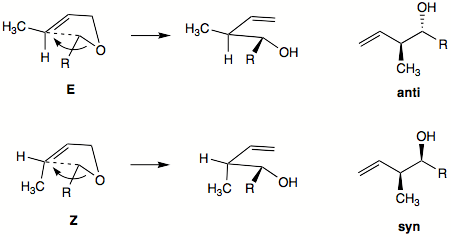
Diastereoselectivity can be high for Z-alkene with alkynyl, alkenyl, or aryl anion-stabilizing group. Diastereoselectivity is usually lower with E-alkenes. Hydrocarbon groups will prefer exo orientation in the envelope-like transition state. Anion-stabilizing group will prefer the endo orientation in transition state.
References
References
- {{JerryMarch
- ''Ring expansion by 2,3-sigmatropic shifts of unstabilized sulfonium ylides. Synthesis of eight- to ten-membered thiacycloalk-4-enes'' V. Cere, C. Paolucci, S. Pollicino, E. Sandri, and A. Fava The Journal of Organic Chemistry 1978 43 (25), 4826-4831 {{doi. 10.1021/jo00419a024
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about 2,3-sigmatropic rearrangement — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report