From Surf Wiki (app.surf) — the open knowledge base
Hofmann–Löffler reaction
Chemical reaction

Chemical reaction
Karl Löffler Curt Freytag In organic chemistry, the Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is a cyclization reaction with remote C–H functionalization.{{cite journal

History
In 1878, the structure of piperidine was still unknown, and A. W. Hofmann believed it was unsaturated.{{cite journal
1bromo-2propylpiperidine (3) and hot sulfuric acid, followed by basic work-up, formed a tertiary amine,{{cite journal

No further examples of the reaction were reported for about 25 years. But in 1909, K. Löffler and C. Freytag extended the transformation to simple secondary amines and applied the process in their elegant synthesis of nicotine (6) from N-bromo-N-methyl-4-(pyridin-3-yl)butan-1-amine (5).{{cite journal

The reaction mechanism only became clear around 1950, when S. Wawzonek investigated various N-haloamine cyclizations.{{cite journal
Reaction mechanism
According to Wawzonek and Thelan's 1949 proposal, an acid first protonates an N-chloroamine, which, in the presence of heat, light, or other initiators, homolyzes to ammonium and chloride free radicals. The ammonium radical intramolecularly abstracts a sterically favored hydrogen atom to afford an alkyl radical which, in a chain reaction, abstracts chlorine from another N-chloroammonium ion to form an alkyl chloride and a new ammonium radical. The alkyl chloride later cyclizes during the basic work-up to the cyclic tertiary amine.{{cite journal

Because the hydrogen abstraction is radical, any chiral configuration at the δ-carbon racemizes.
The reaction also has a quite large hydrogen isotope effect: in the decomposition of 10, the ratio of 1,2-dimethylpyrrolidine 11 and 1,2-dimethylpyrrolidine-2-d 12 (determined by combustion and IR spectra) suggests ≈ 3.42–3.54.

Comparable reactions at a primary carbon also give ≫1, which strongly suggests that the breaking of the C-H bond proceeds to a rather considerable extent in the transition state.
Initiation, inhibition, catalysis
Molecular oxygen inhibits the reaction (trapping the radicals), but Fe2+ salts initiate it.
Further investigations demonstrated that both the rate of the ultraviolet-catalyzed decomposition of dibutylchloroamine and the yield of newly formed pyrrolidine are strongly dependent on the acidity of the reaction medium – faster and higher-yielding reaction was observed with increasing sulfuric acid concentration.
An important question in discussing the role of the acid is whether the N-haloamine reacts in the free base or the salt form in the initiation step. Based on the pKa values of the conjugate acids of 2° alkyl amines (which are generally in the range 10–11), it is evident that N-chloroamines exist largely as salts in a solution of high sulfuric acid concentration. As a result, in the case of chemical or thermal initiation, it is reasonable to assume that it is the N-chloroammonium ion which affords the ammonium free radical. The situation changes, however, when the reaction is initiated upon irradiation with UV light. The radiation must be absorbed and the quantum of the incident light must be large enough to dissociate the N-Cl bond in order for a photochemical reaction to occur. Because the conjugate acids of the N-chloroamines have no appreciable UV absorption above 225 nm, whereas the free N-chloroamine absorb UV light of sufficient energy to cause dissociation (λmax 263 nm, εmax 300),{{cite journal While it was proposed that the higher acid concentration decreases the rate of the initiation step, the acid catalysis involves acceleration of the propagation steps and/or retardation of the chain termination. The influence of certain acidic solvents on the photolytic Hofmann–Löffler–Freytag reaction was also studied by Neale and co-workers.{{cite journal
Intermediates
Isolation of 4-chlorodibutylamine from decomposition of dibutylchloroamine in H2SO4 confirmed the intermediacy of δ–chloroamines. When the acidic solution is made basic, the δ–chloroamine cyclizes to give a cyclic amine and a chloride ion.
Selectivity of hydrogen transfer
In order to determine the structural and geometrical factors affecting the intramolecular hydrogen atom transfer, a number of different N-chloroamines were examined in the Hofmann–Löffler–Freytag reaction. The systems were judiciously chosen in order to obtain data on the following points: relative migration tendencies of primary (1°), secondary (2°) and tertiary (3°) hydrogens; relative rates of 1,5- and 1,6-hydrogen rearrangements; and facility of hydrogen rearrangements in cyclic systems of restricted geometry.
Investigation of the free radical decomposition of N-chlorobutylamylamine 13 allowed to determine 1° vs. 2° hydrogen migration. It was reported that only 1-n-butyl-2-methylpyrrolidine 14 was formed under the reaction conditions, no 1-n-amylpyrrolidine 15 was detected. This observation provided substantial evidence that the radical attack exhibits strong preference for the 2° over 1° hydrogen.

Tendency for 3° vs. 1° hydrogen migration was studied with n-butylisohexylamine 16. When 16 was subjected to the standard reaction conditions, rapid disappearance of 16 was observed, but no pyrrolidine product could be isolated. This result suggested that there is a high selectivity for the 3° hydrogen, but the intermediate tertiary chloro compound 17 is rapidly solvolyzed.

Similarly, no cyclic amine was observed with the reaction of n-amylisohexylamine, which demonstrates the selectivity for the 3° vs. 2° hydrogen migration.
A qualitative study of products from the Hofmann–Löffler–Freytag reaction of N-chloromethyl-n-hexylamine 18 was performed in order to evaluate the relative ease of 1,5- and 1,6-hydrogen migration. UV-catalyzed decomposition of 18 followed by basification produced a 9:1 mixture of 1-methyl-2-ethylpyrrolidine 19 and 1,2-dimethylpiperidine 20, which demonstrates that the extent of formation of six-membered rings can be appreciable.

In terms of the geometrical requirements in the intramolecular rearrangement of hydrogen, it was observed that under identical reaction conditions the UV light-catalyzed decomposition of methylcyclohexylchloroamine and N-chloroazacycloheptane proceeds far more slowly than that of dibutylchloroamine. These findings indicate that the prevailing geometries are in these two cases unfavourable for the rearrangement to occur and the Cδ–H–N bond angle required for the intramolecular hydrogen transfer cannot be easily attained.
Generally accepted mechanism
It is generally accepted that the first step in the Hofmann–Löffler–Freytag reaction conducted in acidic medium is the protonation of the N-halogenated amine 21 to form the corresponding N-halogenated ammonium salt 22. In case of thermal or chemical initiation of the free radical chain reaction, the N-halogenated ammonium salt 22 undergoes homolytic cleavage of the nitrogen-halogen bond to generate the nitrogen-centered radical cation 23. In contrast, it has been argued that the UV light-catalyzed initiation involves the free form of the N-haloamine and a rapid protonation of the newly generated neutral nitrogen radical (see the section devoted to mechanistic studies for arguments supporting this statement). Intramolecular 1,5-hydrogen atom transfer produces carbon-centered radical 24, which subsequently abstracts a halogen atom from the N-halogenated ammonium salt 22. This affords the protonated δ-halogenated amine 25 and regenerates the nitrogen-centered radical cation 23, the chain carrier of the reaction. Upon treatment with base, 25 undergoes deprotonation followed by an intramolecular SN2 reaction to yield pyrrolidine 28 via intermediate 27.
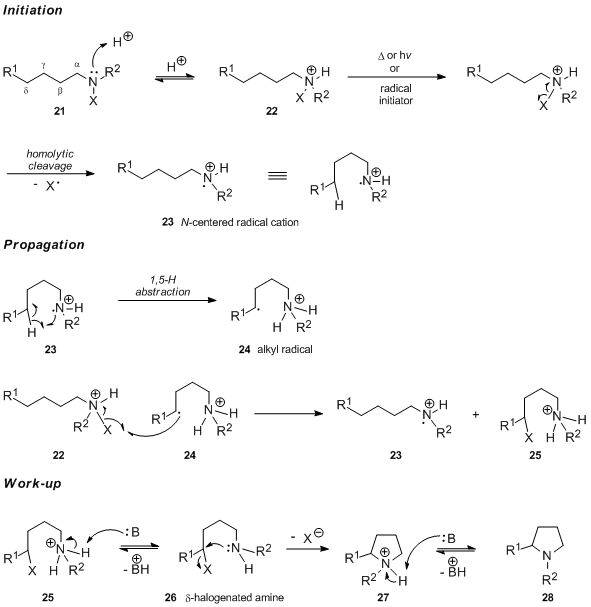
The preferential abstraction of the δ–hydrogen atom corresponds to a six-membered transition state, which can adopt the unstrained cyclohexane chair-type conformation 29.

The Hofmann–Löffler–Freytag reaction is conceptually related to the well-known Barton reaction.
General features of the reaction
- The starting material for the Hofmann–Löffler–Freytag reaction could be N-chloro-, N-bromo-, and N-iodoamines. In case of thermal initiation, the N-chloroamines give better yields for pyrrolidines because N-bromoamines are less stable thermally than the corresponding N-chloroamines.{{cite journal
- The Hofmann–Löffler–Freytag reaction was originally carried out under acidic conditions, but it has been demonstrated that neutral or even weakly basic conditions might also be successfully employed.{{cite journal
- The initially formed nitrogen-centered radical abstracts a H-atom mostly from the δ-position and thus 5-membered rings are formed predominantly.
- Formation of 6-membered rings is also possible, but relatively rare, and in majority of cases is observed in rigid cyclic systems.
- The reaction can be conducted under milder conditions provided that the alkyl radical experiences some form of extra stabilization, e.g. by an adjacent heteroatom.
- The radical process may be initiated by heating, irradiation with light or with radical initiators (e.g. peroxides, metal salts).
Modifications and improvements
Because the original strongly acidic reaction conditions are often not compatible with the sensitive functional and protecting groups of complex substrates, several modifications of the Hofmann–Löffler–Freytag reaction were introduced:
- M. Kimura and Y. Ban demonstrated that adjacent nitrogen atoms can stabilize radical species generated by H-atom abstraction and permit this step to take place under weakly basic conditions{{cite journal

- It has been demonstrated that photolysis of N-haloamides proceeds efficiently under neutral conditions. Irradiation of N-bromoamide 36 (R=tBu) gave rise to bromomethyl-cyclohexane-amide 37 which, upon treatment with base * in situ* afforded iminolactone 38 in 92% yield.{{cite journal

Similarly, S. W. Baldwin and T. J. Doll examined a modification of the Hofmann–Löffler–Freytag reaction during their studies towards the synthesis of the alkaloid gelsemicine 41. The formation of the pyrrolidine ring of 40 was accomplished by irradiation of N-chloroamide 39.

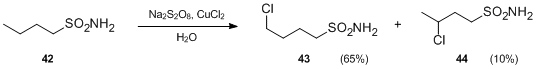
- Another variation of the Hofmann–Löffler–Freytag reaction involves sulfonamides in place of N-haloamines. In the presence of persulphates and metal salts, sulfonamides can undergo intramolecular free-radical funcionalization to produce γ- and δ-chloroalkenylsulfonamides under neutral conditions. For instance, upon treatment with Na2S2O8 and CuCl2, butylsulfonamide 42 was transformed to 4-chlorobutylsulfonamide 43 and 3- chlorobutylsulfonamide 44 in the absence of acid.{{cite journal
- The most important variation of the Hofmann–Löffler–Freytag reaction is the Suárez modification. In 1980, Suárez et al.{{cite journal

The great advantage of the Suárez modification is that the reaction can be performed under very mild neutral conditions compatible with the stability of the protective groups most frequently used in synthetic organic chemistry. Consequently, it permits the use of the Hofmann–Löffler–Freytag reaction with more sensitive molecules. Other notable features of this methodology are the following: (1) the unstable iodoamide intermediates are generated in situ; (2) the iodoamide homolysis proceeds thermally at low temperature (20–40 °C) or by irradiation with visible light, which obviates the need for a UV lamp. The Suárez modification has found numerous applications in synthesis (vide infra).
- Nagib and co-workers have employed a triiodide strategy that expands the scope of the Hofmann–Löffler–Freytag reaction via the Suárez modification to enable the amination of secondary C-H bonds. This approach employs NaI, instead of I2, as a radical precursor to prevent undesired I2-mediated decomposition pathways. Other halide salts (e.g. NaCl and NaBr) afford the postulated intermediates of the interrupted Hofmann–Löffler–Freytag mechanism.
Applications in synthesis
The most prevalent synthetic utility of the Hofmann–Löffler–Freytag reaction is the assembly of the pyrrolidine ring.
The Hofmann–Löffler–Freytag reaction under standard conditions
The procedure for the Hofmann–Löffler–Freytag reaction traditionally requires strongly acidic conditions, which limits its appeal. Nonetheless, it has been successfully applied to functionalization of a wide variety of structurally diverse molecules as exemplified below.
In 1980, J. P. Lavergne. et al.{{cite journal

P. E. Sonnet and J. E. Oliver{{cite journal

Another example of the construction of a bicyclic amine through the standard Hofmann–Löffler–Freytag methodology is the Waegell's synthesis{{cite journal
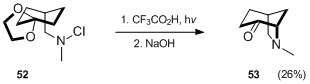
The Hofmann–Löffler–Freytag reaction was employed to synthesize the bridged nitrogen structure of (±)-6,15,16-iminopodocarpane-8,11,13-triene 55, an intermediate useful for the preparation of the kobusine-type alkaloids, from a bicyclic chloroamine 54.{{cite journal

Derivatives of adamantane have also been prepared using the Hofmann–Löffler–Freytag reaction.{{cite journal

Similarly, it has been demonstrated{{cite journal

R. P. Deshpande and U. R. Nayak{{cite journal

An outstanding application of the Hofmann–Löffler–Freytag reaction is found in the preparation of the steroidal alkaloid derivatives. J. Hora{{cite journal

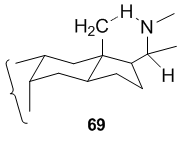
In case of 64 and 66, the five-membered nitrogen ring is formed by attack on the unactivated C-18 methyl group of the precursor (63 or 65, respectively) by a suitably placed nitrogen-centered radical at C-20. The ease of this reaction is due to the fact that in the rigid steroid framework the β-C-18 methyl group and the β-C-20 side chain carrying the nitrogen radical are suitably arranged in space in order to allow the 1,5-hydrogen abstraction to proceed via the six-membered transition state.
The Hofmann–Löffler–Freytag reaction under mild conditions
A number of examples of the Hofmann–Löffler–Freytag reaction under neutral conditions have been presented in the section devoted to modifications and improvements of the original reaction conditions. Hence, the main focus of this section are the applications of the Suárez modification of the Hofmann–Löffler–Freytag reaction.
The Suárez modification of the Hofmann–Löffler–Freytag reaction was the basis of the new synthetic method developed by H. Togo et al.{{cite journal

A very interesting transformation is observed when sulfonamides of primary amides bearing an aromatic ring at the γ-position are treated with various iodanes and iodine under the irradiation with a tungsten lamp.{{cite journal

By the same procedure, 3,4-dihydro-2,1-benzothiazine-2,2-dioxides 81 are obtained from the N-alkyl 2-(aryl)ethanesulfonamides via the sulfonamidyl radical.{{cite journal

E. Suárez et al.{{cite journal

E. Suárez and co-workers also applied their methodology in the synthesis of chiral 8-oxa-6-azabicyclo[3.2.1]-octane 85 and 7-oxa-2-azabicyclo[2.2.1]heptane 87 ring systems. This reaction can be considered to be an intramolecular N-glycosidation that goes through an intramolecular 1,5-hydrogen abstraction promoted by an N-amido radical followed by oxidation of the transient C-radical intermediate to an oxycarbenium ion, which is subsequently trapped by an internal nucleophile.

The utility of the Suárez modification of the Hofmann–Löffler–Freytag reaction was demonstrated by its application in synthesis of a number of steroid and triterpene compounds.{{cite journal

In 2008 P.S. Baran et al.{{cite journal
References
References
- (2016-08-16). "Triiodide-Mediated δ-Amination of Secondary C−H Bonds". Angewandte Chemie International Edition.
- (2017-08-02). "Directed β C–H Amination of Alcohols via Radical Relay Chaperones". Journal of the American Chemical Society.
- (2019-02-27). "Catalytic β C–H amination via an imidate radical relay". Chemical Science.
- (2020-06-22). "Enantioselective radical C–H amination for the synthesis of β-amino alcohols". Nature Chemistry.
This article was imported from Wikipedia and is available under the Creative Commons Attribution-ShareAlike 4.0 License. Content has been adapted to SurfDoc format. Original contributors can be found on the article history page.
Ask Mako anything about Hofmann–Löffler reaction — get instant answers, deeper analysis, and related topics.
Research with MakoFree with your Surf account
Create a free account to save articles, ask Mako questions, and organize your research.
Sign up freeThis content may have been generated or modified by AI. CloudSurf Software LLC is not responsible for the accuracy, completeness, or reliability of AI-generated content. Always verify important information from primary sources.
Report