From Surf Wiki (app.surf) — the open knowledge base
Dislocation
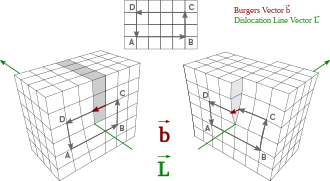
Linear crystallographic defect or irregularity
Rendering article…
Content sourced from wikipedia_dump, available under CC BY-SA 4.0.
This content may have been generated or modified by AI, and may be sourced from third parties. CloudSurf Software LLC makes no warranties as to its accuracy, completeness, or reliability, and accepts no liability for it. Always verify important information against primary sources.
Report